Abstract
Purpose
The objectives of this study were to evaluate the various branching patterns of segmental bronchi in the right middle lobe (RML) and to survey the anatomical diversity and sex-related differences of these branches in a large sample of the study population.
Materials and methods
In this retrospective board-approved study with informed consent, 10,000 participants (5428 males and 4,572 females, mean age 50 ± 13.5 years [SD]; age range: 3–91 years) who underwent multi-slice CT (MSCT) scans from September 2019 to December 2021 were retrospectively included. The data were applied to generate three-dimensional (3D) and virtual bronchoscopy (VB) simulations of a bronchial tree using the syngo.via post-processing workstation. The reconstructed images were then interpreted to locate and classify distinct bronchial patterns in the RML. Cross-tabulation analysis and the Pearson chi-square test were used to calculate the constituent ratios of bronchial branch types and determine their significance between male and female groups.
Results
Our results revealed that the segmental bronchial ramifications of the RML were classified into two types mainly, i.e., bifurcation (B4, B5, 91.42%) and trifurcation (B4, B5, B*, 8.58%). There were no significant sex-related differences in the proportion of bronchial branches in the RML (P > 0.05).
Conclusion
The current study has confirmed the presence of segmental bronchial variations in the RML lobe using 3D reconstruction and virtual bronchoscopy. These findings may have significant implications for the diagnosis of symptomatic patients and for carrying out specific procedures like bronchoscopy, endotracheal intubation, and lung resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The tracheobronchial tree, which consists of the trachea, segmental bronchi, and bronchioles, originates from the larynx and is responsible for the transport of respiratory gases [18]. We believe that an exact comprehension of the bronchopulmonary segmental anatomy is crucial for precisely interpreting radiological images and air bronchograms [14]. The Boyden classification of bronchi and the international nomenclature of bronchopulmonary anatomy, both based on postmortem sampling and published before the advent of computed tomography in 1950 and 1955, respectively, are extensively used to represent normal bronchial anatomy and variations [3, 4, 24]. The branching patterns of the segmental bronchi, on the other hand, were not addressed. Such nomenclatures not only make it difficult for surgeons to assess lung anatomy accurately, but they can also make resecting a single segmental bronchus more challenging [34].
The anatomy of the tracheobronchial tree, defined in the 41st edition of Gray’s Anatomy [1], focuses only on the most common anatomical pattern of bronchi without any enclosure of anatomical variations, which often confuses anatomists while classifying the bronchi of sectional specimens.
Hence, enhancing the level of expertise in evaluating and classifying segmental bronchial branching patterns is crucial not only for identifying bronchial ramifications on anatomical sectional specimens and clarifying radiological images but also for performing bronchoscopies and pulmonary segmentectomies [19, 34].
To date, several methodologies have been developed to learn about the branching pattern of segmental bronchi including anatomical specimens [22], bronchography [16], transverse CT [15, 20], and bronchoscopy [2]. Unfortunately, there is no general unanimity on the best strategy for evaluating and categorizing segmental bronchial variations. Currently, with the application of a series of new surgical methods such as segmental pneumonectomy in clinical practice, clinicians need to have solid anatomical knowledge to better understand the branching pattern of the segmental bronchi. A distinct bronchial division of each lung segment may provide more precise instructions for medical professionals [28]. Therefore, further anatomical research is required to not only analyze and identify typical bronchial patterns but also to distinguish any other possible variations.
In recent years, with the advances in CT imaging methods, multi-slice Somatom Definition Flash CT has been applied for radiological assessments, ushering in a new era of medical imaging and broadening the potential use of imaging for studying the anatomy of the airway by offering various image reconstruction techniques [9]. Dual-source Flash CT achieves a real sense of rapid scanning and can greatly reduce dynamic changes in image density due to cardiopulmonary activity. At the same time, flash scanning can attain single-sector reconstruction (semi-scan) and enhance diagnostic accuracy by displaying the finest bronchial patterns with unprecedented clarity, thereby providing an accurate, new, and reliable method for the anatomical exploration of bronchial segments.
Notably, high-quality three-dimensional (3D) images and virtual bronchoscopy (VB) images can be generated using the post-processing workstation of Siemens syngo.via [5, 10, 13].
At present, there is also a lack of statistical analysis data on the composition ratio of bronchial branch types, and no studies on sex-related differences based on large sample sizes have been reported. Due to the lack of previous clinical studies on this specific part of the lung and the limited size of the study samples, we mainly focused on the right middle lobe (RML) to identify the prevailing pattern and the variations of segmental bronchi, which might improve the clinical diagnostic and treatment efficacy.
Furthermore, to our knowledge, this is the first study to explore bronchial segmental variations and assess if there are any sex-related differences regarding these branching types in the general population on a large scale.
In recent times, the coronavirus pandemic (COVID-19) impacted the whole world and caused a substantial increase in the rates of morbidity and mortality. Although most of the commonly used clinical investigation modalities, such as chest X-ray, CT [17, 25], and bronchoscopy [30], are helpful, there is still an urgent need for an efficient tool with comprehensive knowledge to assess and precisely pinpoint the fibrotic changes in pulmonary tissues. Thus, the current investigation using multi-slice Somatom Definition Flash CT may provide health professionals with a novel opportunity to estimate the magnitude and dispersion of the disease at a glance. Moreover, it can provide a roadmap for procedures like bronchoscopy, bronchoalveolar lavage, and segmental resection.
In this regard, this study aimed to use 3D and VB images acquired from MSCT scans to reveal various branching types of segmental bronchi in the RML and survey the anatomical diversity and sex-related differences of these branches in a large sample of the study population.
Materials and methods
Study sample
The current study was approved by our institutional ethics review board, which followed the principles of the World Medical Association Declaration of Helsinki and written informed consent was obtained for the retrospective review of patient records and images. The study participants were chosen randomly from patients who presented to our hospital with complaints of cough, dyspnea, and/or hemoptysis during the period from October 2020 to September 2022. Our retrospective analysis was based on the Somatom Definition Flash CT images of 10,000 patients without endobronchial or pulmonary lesions. The study sample included 5428 males (3–91 years of age; mean age: 49.57 years; standard deviation [SD]: 13.9 years) and 4572 females (3–91 years of age; mean age: 49.67 years; SD: 13.3 years).
Imaging technique
All patients underwent conventional thoracic scanning with dual-source multi-slice Somatom Definition Flash CT (Siemens, Forchheim, Germany) with the following scanning parameters: collimation 64 × 0.6 mm, frame rotation time 0.33 s/r, scanning time 4.32 s, interval 5 s, tube voltage 120 kV, reference tube current 137Mas, reconstruction slice thickness 1.0 mm, interval 1.0 mm and standardized mediastinal window. All participants were scanned in a supine position. The scanning range was from cranial to caudal (i.e. from the larynx to the lower edge of the diaphragm).
Image analysis
Two experienced radiologists who majored in sectional radiological anatomy reviewed and interpreted all the 3D and VB images, and reached their conclusion by consensus. The actual data collected were uploaded to Siemens syngo.via client software for image post-processing, which then, was reconstructed to generate a 3D bronchial tree and VB images.
The identification of segmental bronchi was based on bronchial lumen diameter, branching direction, extension orientation, and the number of segmental branches in a 3D bronchial tree. Since the bronchial tree could rotate around in 3D space, the viewing angle of the 3D image was altered periodically in case the region of interest was blurred or the line of sight was blocked by another bronchus.
To reconstruct the VB images, first, we identified the specific bronchus and placed a virtual endoscope on a sectional image of the CT lung window. Then, the chosen bronchoscope was converted into a simulated bronchoscope image. Finally, the virtual bronchoscope passed through the bronchus to the segmental bronchi of each lung segment.
The segmental bronchi were named and numbered according to their location and anatomical configuration, which further aided in classifying various bronchial patterns precisely.
In addition, the categorization of segmental bronchial patterns was based on the bifurcation or trifurcation of lobar bronchi of the 3D bronchial tree and the shape of the carina in VB images.
Statistical analysis
Data on the medical record number, sex, age, and branch types of the segmental bronchus in the right middle lobe were collected. IBM SPSS Statistics 22.0 software (IBM Corp., Armonk, NY, USA) was used to analyze the data. First, cross-tabulation was used to calculate the constituent ratio of the bronchial branch types in the right middle lobe. Then, we compared the overall composition ratio of bronchial branch types between males and females to assess the significance of a relationship between sex and the branching types using the Pearson chi-square test.
Results
To reveal the segmental bronchial branching types in the RML, CT scans of the thorax were carefully examined. The study population contained 5428 (54.28%) males and 4572 (45.72%) females (in total 10,000 participants).
The classification of the bronchial branching patterns appears difficult when the radiologist manually evaluates each 2D segment since this interpretation necessitates a 3D conceptual anatomical reconstruction from many 2D sections. Hence, the branching patterns of bronchi could be fully understood at a glance with one 3D image of a bronchial tree or VB.
Bronchial tree of the right middle lobe (RML)
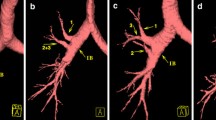
There were two main anatomical types of the bronchial tree of the RML (Figs. 1, 2, 3 and Table 1). In 9142 cases (type I, 91.42%), the bronchus of the RML bifurcated into lateral (B4) and medial (B5) segments (Fig. 1a, b). In the remaining 858 cases (type II, 8.58%), the bronchus was trifurcated into lateral (B4), medial (B5), and extra bronchus (B*) (Fig. 2a, b).
Discussion
The advancement of interventional respiratory medicine necessitates the acquisition of bronchial anatomical knowledge, whose diversity in branching types has become increasingly substantial in recent years. These branches may differ from person to person in terms of their origin and number, making it more challenging for medical experts to comprehend the exact anatomy in diagnosing and applying a precise treatment approach. The current study demonstrated for the first time the presence of structural anatomical variations of segmental bronchi in a large number of study participants, which were analyzed using 3D reconstruction and virtual bronchoscopy acquired using somatom definition flash CT. Our results revealed mainly two types of bronchial branching patterns in the right middle lobe. To fully acknowledge these branching patterns, an optimal nomenclature is essential for tackling this issue. Nevertheless, it is only possible if we first understand the bronchial tree variations embryologically and the different research methodologies that have been implemented previously [2, 4].
Normal tracheobronchial growth commences by week 4 of gestation when some endodermal cells from the cranial portion of the foregut start budding off the foregut and creating a tiny bud called the respiratory diverticulum, which then gives rise to the right and left lung buds at 26–28 embryonic days. These lung buds elongate into primary bronchi, and the five lobar bronchi appear as a sympodial outgrowth of the primary bronchi at 30–32 embryonic days. The lobar bronchi then elongate and promptly branch off to form all segmental bronchi at 32–34 embryonic days, at which time bronchial variations may also occur [8].
Numerous imaging methodologies, such as dissection [22], bronchography [16, 27], transverse CT [15, 20], spiral CT [10, 21], and conventional fiberoptic bronchoscopy [2] have been developed to examine the course and distribution of bronchial branches and, as a result, to ensure the correct diagnostic approach and surgical strategy.
Among these methods, dissection is the most basic and direct method to study the branching course and distribution rules of segmental bronchi. Although it is considered the most accurate modality for studying bronchial ramifications, the method and process of creating a bronchial tree casting exhibit some limitations and complications, such as the limited number of specimens and the need for contrasting colors, rendering this method unhelpful in research with large sample sizes. Bronchography is another method that can be used to observe the anatomy of a live human being. However, this technique uses 2D imaging, which can cause an overlap between bronchi running in 3D space, thus causing imprecision. CT scanning technology (transverse CT and spiral CT) [2, 19] can achieve relatively better results, but it requires spatial integration of fractional data accumulated from multiple sections, a procedure that can be strenuous. Lastly, fiberoptic bronchoscopy can only provide endobronchial views and is invasive, which may cause a slight risk of breathing discomfort, bleeding, infection, or even lung collapse.
In the current study, a high-resolution Somatom Definition Flash CT assisted with the Siemens Syngo.via post- processing workstation was used for 3D bronchial reconstruction and VB perspectives. The bronchial anatomy could, thus, be exhibited through extra bronchial and endobronchial views using the 3D bronchial tree and VB images, respectively, allowing the bronchial branching configurations to be interpreted at a glimpse [11, 31]. Furthermore, by rotating the bronchial tree in 3D space, it was possible to present the composite bronchial structures in a way that was more anatomically accurate while overcoming the confines of overlapping and hazy segmental bronchial display seen in 2D imaging techniques like bronchography and transverse CT [7, 11, 25, 31, 34]. Importantly, due to advances in multi-detector CT that allow the acquisition of isotropic data, the reconstruction data of 3D images could be obtained from the standard thorax examination of Somatom Flash CT for clinical diagnosis. As a result, the 3D and VB images acquired in our study were easier to construct than anatomical specimens.
Compared with the previous studies, the current study was able to improve the quality of the data by increasing the sample size and providing more accurate information on the anatomical types of segmental bronchi [2, 11, 15, 16, 20, 22]. Most of the variations found in our study were extreme and had not been documented by predecessors. Such as, the bronchi of the right middle lobe showed trifurcation types with an incidence of 8.58%. In addition, the use of virtual bronchoscopes and 3D reconstruction images allowed the researchers to collect more detailed information on the sex-related changes in the segmental bronchi. Hence, we found no significant sex-related difference in the proportion of bronchial branches in the RML (P > 0.05).
The probable reason for the differences might be the different sample sizes or modalities used in previous studies. A distinctive benefit of the current study was that the 3D bronchial tree images could be interpreted by visualizing in a panoramic view. Our documentation of the various anatomical configurations of the bronchial tree may be of assistance in distinguishing all bronchopulmonary segments and, hence, precisely accounting for the locality of focal lesions in the lungs [19, 23, 26]. Thus, a preoperative understanding of the bronchosegmental anatomy can help surgeons in performing precise surgical procedures like sublobar resection or pulmonary segmentectomy [29, 32, 33]. Moreover, a thorough preoperative understanding of the bronchosegmental anatomy may aid surgeons in performing surgical procedures like sublobar resection or pulmonary segmentectomy and can also offer precise instructions for conducting fiberoptic bronchoscopy [6, 29, 32, 33]. In the absence of such knowledge, the implementation of thoracic surgery or fiberoptic bronchoscopy intervention may lead to incomplete treatment or failure. Regarding the implications of our study, it can also be very useful for patients with COVID-19 who typically require a noninvasive investigation to reach a correct imaging diagnosis, making it easier to implement bronchoalveolar lavage (BAL), segmentectomy, lobectomy, or other medical approaches accurately and smoothly [12, 13, 17]. However, one probable limitation of this study was that the subjects were selected from patients who had some prevalence of airway diseases and were not healthy volunteers. In conclusion, employing MSCT, our study has successfully identified and displayed simplified bronchial branching patterns in the RML using 3D and VB images. Besides, this is also the first study examining sex-related differences in the types of segmental bronchi based on a large sample size. Thus, the results obtained in this study may allow medical professionals to understand the course and distribution of segmental bronchial branches and their variations pre-operatively to articulate a precise surgical and interventional treatment.
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Abbreviations
- 3D:
-
Three dimensional
- 2D:
-
Two dimensional
- VB:
-
Virtual bronchoscopy
- RML:
-
Right middle lobe
- B4:
-
Lateral segmental bronchus
- B5:
-
Medial segmental bronchus
- B*:
-
Extra segmental bronchus
References
Anthony VD (2016) Gray’s anatomy, the anatomical basis of clinical practice. Clin Anat 29:264–265. https://doi.org/10.1002/ca.22677
Beder S, Küpeli E, Karnak D, Kayacan O (2008) Tracheobronchial variations in turkish population. Clin Anat 21:531–538. https://doi.org/10.1002/ca.20667
Boyden EA (1955) Segmental anatomy of the lungs a study of the patterns of the segmental bronchi and related pulmonary vessels. McGraw-Hill, New York
Chassagnon G, Morel B, Carpentier E, Le Pointe HD, Sirinelli D (2016) Tracheobronchial branching abnormalities: lobe-based classification scheme. Radiographics 36:358–373. https://doi.org/10.1148/rg.2016150115
Dalrymple NC, Prasad SR, Freckleton MW, Chintapalli KN (2005) Introduction to the language of three-dimensional imaging with multidetector CT. Radiographics 25:1409–1428. https://doi.org/10.1148/rg.255055044
Fetita CI, Prêteux F, Beigelman AC, Grenier P (2004) Pulmonary airways: 3-D reconstruction from multislice CT and clinical investigation. IEEE Trans Med Imaging 23:1353–1364. https://doi.org/10.1109/TMI.2004.826945
Gariani J, Martin SP, Botsikas D, Becker CD, Montet X (2018) Evaluating the effect of increased pitch, iterative reconstruction and dual source CT on dose reduction and image quality. Br J Radiol 91(1088):20170443. https://doi.org/10.1259/bjr.20170443
Ghaye B, Szapiro D, Fanchamps JM, Dondelinger RF (2001) Congenital bronchial abnormalities revisited. Radiographics 21:105–119. https://doi.org/10.1148/radiographics.21.1.g01ja06105
Hammond E, Chan KS, Ames JC, Stoyles N, Sloan CM, Guo J, Newell JD, Hoffman EA, Sieren JC (2018) Impact of advanced detector technology and iterative reconstruction on low-dose quantitative assessment of lung computed tomography density in a biological lung model. Med Phys 45:3657–3670. https://doi.org/10.1002/mp.13057
Horton KM, Horton MR, Fishman EK (2007) Advanced visualization of airways with 64-MDCT: 3D mapping and virtual bronchoscopy. Am J Roentgenol 189:1387–1396. https://doi.org/10.2214/AJR.07.2824
Huang M, Wang T, Wang X, Zhao X (2019) An anatomical study of the right bronchial tree using multi-detector computed tomography. Surg Radiol Anat 41:335–338. https://doi.org/10.1007/s00276-019-02199-7
Keenan RJ, Landreneau RJ, Maley RH, Singh D, Macherey R, Bartley S, Santucci T (2004) Segmental resection spares pulmonary function in patients with stage I lung cancer. Ann Thorac Surg 78:228–233. https://doi.org/10.1016/j.athoracsur.2004.01.024
Kiraly AP, Helferty JP, Hoffman EA, McLennan G, Higgins WE (2004) Three-dimensional path planning for virtual bronchoscopy. IEEE Trans Med Imaging 23:1365–1379. https://doi.org/10.1109/TMI.2004.829332
Krause GR, Lubert M (1951) The anatomy of the bronchopulmonary segments: clinical applications. Radiology 56:333–354. https://doi.org/10.1148/56.3.333
Lee KS, Bae WK, Lee BH, Kim IY, Choi EW, Lee BH (1991) Bronchovascular anatomy of the upper lobes: evaluation with thin-section CT. Radiology 181:765–772. https://doi.org/10.1148/radiology.181.3.1947094
Leroux BT (1962) The bronchial anatomy of the left upper lobe. J Thorac Cardiovasc Surg 44:216–224. https://doi.org/10.1016/s0022-5223(19)32975-710.1016/j.compmedimag.2013.07.003
Li Y, Xia L (2020) Coronavirus disease 2019 (COVID-19): role of chest CT in diagnosis and management. Am J Roentgenol 214:1280–1286. https://doi.org/10.2214/AJR.20.22954
Mehran RJ (2018) Fundamental and practical aspects of airway anatomy: from glottis to segmental bronchus. Thorac Cardiovasc Surg 28:117–125. https://doi.org/10.1016/j.thorsurg.2018.02.003
Mori K, Ema S, Kitasaka T, Mekada Y, Ide I, Murase H, Suenaga Y, Takabatake H, Mori M, Natori H (2005) Automated nomenclature of bronchial branches extracted from CT images and its application to biopsy path planning in virtual bronchoscopy. Lect Notes Comput Sci 3750:854–861. https://doi.org/10.1007/11566489_105
Naidich DP, Zinn WL, Ettenger NA, Garay MDI, SM, (1988) Basilar segmental bronchi: thin-section CT evaluation. Radiology 169:11–16. https://doi.org/10.1148/radiology.169.1.3420245
Perhomaa M, Lähde S, Rossi O, Suramo I (1997) Helical CT in evaluation of the bronchial tree. Acta Radiol 38:83–91. https://doi.org/10.3109/02841859709171247
Pitel M, Boyden EA (1953) Variations in the bronchovascular patterns of the left lower lobe of fifty lungs. J Thorac Surg 26:633–653. https://doi.org/10.1016/s0096-5588(20)30791-1
Reck M, Rabe KF (2017) Precision diagnosis and treatment for advanced non–small-cell lung cancer. N Engl J Med 377:849–861. https://doi.org/10.1056/nejmra1703413
Room C (1950) Nomenclature of broncho-pulmonary anatomy; an international nomenclature accepted by the thoracic society. Thorax 5:222–228. https://doi.org/10.1136/thx.5.3.222
Rosell J, Cabras P (2013) A three-stage method for the 3D reconstruction of the tracheobronchial tree from CT scans. Comput Med Imaging Graph 37:430–437. https://doi.org/10.1016/j.compmedimag.2013.07.003
Salvolini L, Bichi SE, Costarelli L, DeNicola M (2000) Clinical applications of 2D and 3D CT imaging of the airways—a review. Eur J Radiol 34:9–25. https://doi.org/10.1016/S0720-048X(00)00155-8
Scannell JG (1949) Bronchographic anatomy of the lungs. Surg Clin North Am 29(2):573–581. https://doi.org/10.1016/S0039-6109(16)32701-3
Sealy WC, Connally SR, Dalton ML (1993) Naming the bronchopulmonary segments and the development of pulmonary surgery. Ann Thorac Surg 55:184–188. https://doi.org/10.1016/0003-4975(93)90507-E
Ugalde P, Camargo J, de J, Deslauriers J, (2007) Lobes, fissures, and bronchopulmonary segments. Thorac Cardiovasc Surg 17:587–599. https://doi.org/10.1016/j.thorsurg.2006.12.008
Wahidi MM, Shojaee S, Lamb CR, Ost D, Maldonado F, Eapen G, Caroff DA, Stevens MP, Ouellette DR, Lilly C, Gardner DD, Glisinski K, Pennington K, Alalawi R (2020) The use of bronchoscopy during the coronavirus disease 2019 pandemic: CHEST/AABIP guideline and expert panel report. Chest 158:1268–1281. https://doi.org/10.1016/j.chest.2020.04.036
Wang T, Meng M, Huang M, Zhao X (2018) Variations of right bronchial tree: a study with multi-detector CT. Surg Radiol Anat 40:955–958. https://doi.org/10.1007/s00276-018-2033-1
Wu WB, Xu XF, Wen W, Xu J, Zhu Q, Chen L (2016) Thoracoscopic pulmonary sub-subsegmentectomy based on three-dimensional images. Ann Thorac Surg 102:389–391. https://doi.org/10.1016/j.athoracsur.2016.04.048
Yang Q, Xie B, Hu M, Sun X, Huang X, Guo M (2016) Thoracoscopic anatomic pulmonary segmentectomy: a 3-dimensional guided imaging system for lung operations. Interact Cardiovasc Thorac Surg 23:183–189. https://doi.org/10.1093/icvts/ivw085
Zhao X, Ju Y, Liu C, Li J, Huang M, Sun J, Wang T (2009) Bronchial anatomy of left lung: a study of multi-detector row CT. Surg Radiol Anat 31:85–91. https://doi.org/10.1007/s00276-008-0404-8
Acknowledgements
This study was supported by Major Scientific and Technological Innovation Projects in Shandong Province, China (2019JZZY020106, 2015ZDXX0201A02).
Author information
Authors and Affiliations
Contributions
SJ: Project development, study concepts and design, literature research, clinical studies, data collection, data analysis, manuscript preparation, manuscript editing. YZ: project development, literature research, clinical studies, manuscript editing. DW: study concepts and design, clinical studies, data collection, data analysis. YM: study concepts and design, clinical studies, data collection, data analysis, statistical analysis. HL: study concepts and design, clinical studies, data collection, data analysis. CL: project development, study concepts and design, literature research, manuscript editing. SL: project development, study concepts and design, literature research, manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
All procedures followed were per the protocol of the work center and ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. This work was approved by the Ethics Committee of the School of Basic Medicine of Shandong University (No. ECSBMSSDU2018-1-050).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Javed, S., Zhang, Y., Wan, D. et al. Identification of anatomical types of segmental bronchi in right middle lobe using multi-slice CT. Surg Radiol Anat 45, 819–825 (2023). https://doi.org/10.1007/s00276-023-03159-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-023-03159-y