Abstract
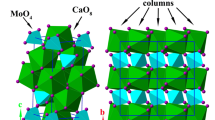
A Cs-bearing polyphase aggregate with composition (in wt%): 76(1)CsAlSi5O12 + 7(1)CsAlSi2O6 + 17(1)amorphous, was obtained from a clinoptilolite-rich epiclastic rock after a beneficiation process of the starting material (aimed to increase the fraction of zeolite to 90 wt%), cation exchange and then thermal treatment. CsAlSi5O12 is an open-framework compound with CAS topology; CsAlSi2O6 is a pollucite-like material with ANA topology. The thermal stability of this polyphase material was investigated by in situ high-T X-ray powder diffraction, the combined P–T effects by a series of runs with a single-stage piston cylinder apparatus, and its chemical stability following the “availability test” (“AVA test”) protocol. A series of additional investigations were performed by WDS–electron microprobe analysis in order to describe the P–T-induced modification of the material texture, and to chemically characterize the starting material and the run products. The “AVA tests” of the polyphase aggregate show an extremely modest release of Cs+: 0.05 mg/g. In response to applied temperature and at room P, CsAlSi5O12 experiences an unquenchable and displacive Ama2-to-Amam phase transition at about 770 K, and the Amam polymorph is stable in its crystalline form up to 1600 K; a crystalline-to-amorphous phase transition occurs between 1600 and 1650 K. In response to the applied P = 0.5 GPa, the crystalline-to-amorphous transition of CsAlSi5O12 occurs between 1670 and 1770 K. This leads to a positive Clapeyron slope (i.e., dP/dT > 0) of the crystalline-to-amorphous transition. When the polyphase aggregate is subjected at P = 0.5 GPa and T > 1770 K, CsAlSi5O12 melts and only CsAlSi2O6 (pollucite-like; dominant) and Cs-rich glass (subordinate) are observed in the quenched sample. Based on its thermo-elastic behavior, P–T phase stability fields, and Cs+ retention capacity, CsAlSi5O12 is a possible candidate for use in the immobilization of radioactive isotopes of Cs, or as potential solid hosts for 137Cs γ-radiation source in sterilization applications. More in general, even the CsAlSi5O12-rich aggregate obtained by a clinoptilolite-rich epiclastic rock appears to be suitable for this type of utilizations.






Similar content being viewed by others
References
Adl T, Vance ER (1982) CsAlSi5O12: a possible host for 137Cs immobilization. J Mater Sci 17:849–855
Annehed H, Fälth L (1984) The crystal structure of Cs0.35Al0.35Si2.65O6, a cesium-aluminosilicate with the bikitaite framework. Z Kristallogr 166:301–306
Araki T (1980) Crystal structure of a cesium aluminosilicate, Cs[AlSi5O12]. Z Kristallogr 152:207–213
Bearlocher Ch, Meier WM, Olson DH (2001) Atlas of zeolite framework types, fifth revised version. Elsevier, Amsterdam
Bellatreccia F, Della Ventura G, Gatta GD, Cestelli Guidi M, Harley S (2012) Carbon dioxide in pollucite, a feldspathoid with the ideal composition (Cs,Na)16Al16Si32O96·nH2O. Min Mag 76:903–911
Brundu A, Cerri G (2015) Thermal transformation of Cs-clinoptilolite to CsAlSi5O12. Micropor Mesopor Mat 208:44–49
Bubnova RS, Krzhizhanovskaya MG, Filatov SK, Ugolkov VL, Paufler P (2007) XRD and DSC study of the formation and the melting of a new zeolite-like borosilicate CsBSi5O12 and (Cs,Rb)BSi5O12 solid solutions. Z Kristallogr 222:83–88
Cappelletti P, Rapisardo G, de’ Gennaro B, Colella A, Langella A, Graziano SF, Bish DL, de’ Gennaro M (2011) Immobilization of Cs and Sr in aluminosilicate matrices derived from natural zeolites. J Nucl Mater 414:451–457
Cerri G, Cappelletti P, Langella A, de’ Gennaro M (2001) Zeolitization of Oligo-Miocene volcaniclastic rocks from Logudoro (northern Sardinia, Italy). Contrib Mineral Petrol 140:404–421
Cerri G, Farina M, Brundu A, Daković A, Giunchedi P, Gavini E, Rassu G (2016) Natural zeolites for pharmaceutical formulations: preparation and evaluation of a clinoptilolite-based material. Micropor Mesopor Mat 223:58–67
Drábek M, Rieder M, Viti C, Weiss Z, Frýda J (1998) Hydrothermal synthesis of a Cs ferruginous trioctahedral mica. Can Mineral 36:755–761
Firor RL, Seff K (1977) Zero-coordinate K+. Crystal structure of dehydrated cesium and potassium exchanged zeolite A, Cs7K5-A. J Am Chem Soc 99:6249–6253
Fisch M (2007) Synthesis and temperature dependent structure characterization of microporous CsAlSi5O12. MSc Thesis, Universität Bern (Switzerland), p 83
Fisch M, Armbruster Th, Kolesov B (2008) Temperature-dependent structural study of microporous CsAlSi5O12. J Solid State Chem 181:423–431
Fisch M, Armbruster Th, Libowitzky E (2010) Microporous CsAlSi5O12 at non-ambient conditions: partial Na exchange at 100 °C. In: Hadjiivanov K, Valtchev V, Mintova S, Vayssilov G (eds) Topics in chemistry and materials science: advanced micro- and mesoporous materials—09, vol 4. Heron Press, Sofia, pp 61–70
Fumagalli P, Zanchetta S, Poli S (2009) Alkali in phlogopite and amphibole and their effects on phase relations in metasomatized peridotites: a high-pressure study. Contrib Mineral Petrol 158:723–737
Gallagher SA, McCarthy GJ, Smith DK (1977) Preparation and X-ray characterization of CsAlSiO4. Mat Res Bull 12:1183–1190
Gatta GD, Rotiroti N, Zanazzi PF, Rieder M, Drabek M, Weiss Z, Klaska R (2008a) Synthesis and crystal structure of the feldspathoid CsAlSiO4: an open-framework silicate and potential nuclear waste disposal phase. Am Mineral 93:988–995
Gatta GD, Rotiroti N, Fisch M, Kadiyski M, Armbruster T (2008b) Stability at high-pressure, elastic behaviour and pressure-induced structural evolution of CsAlSi5O12, a potential nuclear waste disposal phase. Phys Chem Minerals 35:521–533
Gatta GD, Rinaldi R, McIntyre GJ, Nénert G, Bellatreccia F, Guastoni A, Della Ventura G (2009a) On the crystal structure and crystal chemistry of pollucite, (Cs,Na)16Al16Si32O96·nH2O: a natural microporous material of interest in nuclear technology. Am Mineral 94:1560–1568
Gatta GD, Rotiroti N, Boffa Ballaran T, Sanchez-Valle C, Pavese A (2009b) Elastic behavior and phase-stability of pollucite, a potential host for nuclear waste. Am Mineral 94:1137–1143
Gatta GD, Vignola P, McIntyre GJ, Diella V (2010) On the crystal chemistry of londonite [(Cs,K,Rb)Al4Be5B11O28]: a single-crystal neutron diffraction study at 300 and 20 K. Am Mineral 95:1467–1472
Gatta GD, Vignola P, Lee Y (2011) Stability of (Cs,K)Al4Be5B11O28 (londonite) at high pressure and high temperature: a potential neutron absorber material. Phys Chem Minerals 38:429–434
Gatta GD, Adamo I, Meven M, Lambruschi E (2012a) A single-crystal neutron and X-ray diffraction study of pezzottaite, Cs(Be2Li)Al2Si6O18. Phys Chem Minerals 39:829–840
Gatta GD, Merlini M, Lotti P, Lausi A, Rieder M (2012b) Phase stability and thermo-elastic behavior of CsAlSiO4 (ABW): a potential nuclear waste disposal material. Micropor Mesopor Mat 163:147–152
Gualtieri AF (2000) Accuracy of XRPD QPA using the combined Rietveld-RIR method. J Appl Cryst 33:267–278
Hammersley AP, Svensson SO, Hanfland M, Fitch AN, Häusermann D (1996) Two-dimensional detector software: from real detector to idealized image or two-theta scan. High Press Res 14:235–245
Hughes RW, Weller MT (2002) The structure of the CAS type zeolite, Cs4[Al4Si20O48] by high-resolution powder neutron diffraction and 29Si MAS NMR. Micropor Mesopor Mater 51:189–196
Hwang H, Seoung D, Gatta GD, Blom DA, Vogt T, Lee Y (2015) Topotactic and reconstructive changes at high pressures and temperatures from Cs-natrolite to Cs-hexacelsian. Am Mineral 100:1562–1567
Ishizawa N, Miyata T, Minato I, Marumo F, Iwai S (1980) A structural investigation of alpha-Al2O3 at 2170 K. Acta Crystallogr B 36:228–230
Ito J (1976) Crystal synthesis of a new cesium alumosilicate, CsAlSi5O12. Am Mineral 61:170–171
Klika Z, Weiss Z, Mellini M, Drábek M (2006) Water leaching of cesium from selected cesium mineral analogues. Appl Geochem 21:405–418
Kobayashi H, Yanase I, Mitamura T (1997) A new model for the pollucite thermal expansion mechanism. J Am Ceramic Soc 80:2161–2164
Kobayashi H, Sumino S, Tamai S, Yanase I (2006) Phase transition and lattice thermal expansion of Cs-deficient pollucite, Cs1−x Al1−x Si2+x O6 (x ≤ 0.25), compounds. J Am Ceramic Soc 89:3157–3161
Komarneni S, Roy R (1983) Hydrothermal reaction and dissolution studies of CsAlSi5O12 in water and brines. J Am Ceramic Soc 66:471–474
Komarneni S, White WB (1981) Stability of pollucite in hydrothermal fluids. Sci Basis Nucl Waste Manag 3:387–396
Lambruschi E, Gatta GD, Adamo I, Bersani D, Salvioli-Mariani E, Lottici PP (2014) Raman and structural comparison between the new gemstone pezzottaite Cs(Be2Li)Al2Si6O18 and Cs-beryl. J Raman Spectrosc 45:993–999
Larson AC, Von Dreele RB (1994) General structure analysis system (GSAS). Los Alamos National Laboratory Report LAUR, pp 86–748
Liguori B, Caputo D, Iucolano F, Aprea P, de' Gennaro B (2013) Entrapping of Cs and Sr in heat-treated zeolite matrices. J Nucl Mat 435:196–201
Mellini M, Weiss Z, Rieder M, Drábek M (1996) Cs-ferriannite as a possible host for waste cesium: crystal structure and synthesis. Eur J Mineral 8:1265–1271
Pabalan RT, Bertetti FP (2001) Cation-exchange properties of natural zeolites. Rev Mineral Geochem 45:453–518
Rampf M, Fisch M, Dittmer M, Ritzberger C, Schweiger M, Höland W (2015) Tailoring the thermal expansion of glass-ceramics by controlled twofold crystallization of Li2Si2O5 and CsAlSi5O12. Inter J Appl Glass Sci (in press). doi:10.1111/ijag.12180
Sanchez-Valle V, Chi-Hong C, Gatta GD (2010) Single-crystal elastic properties of (Cs,Na)AlSi2O6·H2O pollucite: with potential use for long-term storage of Cs radioisotopes. J Appl Phys 108(1–7):093509
Sovacool BK (2010) A critical evaluation of nuclear power and renewable electricity in Asia. J Contemp Asia 40:393–400
Taylor P, DeVaal SD, Derrek G (1989) Owen Stability relationships between solid cesium aluminosilicates in aqueous solutions at 200 C. Can J Chem 67:76–81
Thomson P, Cox DE, Hastings JB (1987) Rietveld refinement of Debye-Scherrer synchrotron X-ray data from Al2O3. J Appl Crystallogr 20:79–83
Van Der Sloot HA, Kosson DS (1995) ECN-Report-94-029. Netherlands Energy Res. Foundation, Petten
Vance TB, Seff K (1975) Hydrated and dehydrated crystal structure of seven-twelfths cesium-exchanged zeolites A. J Phys Chem 79:2163–2166
Vance ER, Cartz L, Karioris FG (1984) X-ray diffraction and leaching of CsAlSi5O12 and CsZr2(PO4)3 irradiated by argon (3 MeV) ions. J Mater Sci 19:2943–2947
Acknowledgments
GDG acknowledges the Italian Ministry of Education, MIUR-Project: “Futuro in Ricerca 2012—ImPACT-RBFR12CLQD”. The authors thank Marco Merlini for the data collected at ESRF. GC acknowledges the support of Fondazione Banco di Sardegna, 2011, Project: “Synthesis of ceramic materials of environmental and industrial relevance from zeolitic precursors”. Martin Fisch (Bern) and an anonymous reviewer are thanked for the revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gatta, G.D., Brundu, A., Cappelletti, P. et al. New insights on pressure, temperature, and chemical stability of CsAlSi5O12, a potential host for nuclear waste. Phys Chem Minerals 43, 639–647 (2016). https://doi.org/10.1007/s00269-016-0823-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-016-0823-8