Abstract
Background
PECS type-1 block, a US-guided superficial interfacial block, provides effective analgesia after breast surgery. Aesthetic breast augmentation is one of the most common surgical procedures in plastic surgery. Subpectoral prostheses cause severe pain. The aim of this study was to investigate the effect of different volumes of the solution on the efficacy of PECS type-I block for postoperative analgesia after breast augmentation surgery.
Methods
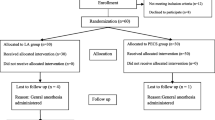
Ninety ASA status I–II female patients aged between 18 and 65 years who scheduled breast augmentation surgery under general anesthesia were included in this study. The patients were randomly divided into three groups of 30 patients each (Group 20 = 20 ml of anaesthetic solution, Group 30 = 30 ml anaesthetic solution, and Group K = Control group). Postoperative assessment was performed using the VAS score. The VAS scores were recorded postoperatively at 1, 2, 4, 8, 16 and 24 h.
Results
Fentanyl consumption was statistically significantly lower in Group 20 and Group 30 compared to the Control group (p < 0.05). There was no statistically significant difference in fentanyl consumption between Group 20 and Group 30. The right and left VAS scores were statistically significantly lower in Groups 20 and 30 than in the Control group (p < 0.05). There was no statistical difference in terms of VAS scores between Group 20 and Group 30. The use of rescue analgesia was statistically lower in Groups 20 and 30.
Conclusions
PECS type-1 block using 20 ml of 0.25% bupivacaine can provide effective analgesia after breast augmentation surgery.
Level of Evidence IV
This journal requires that authors assign a level of evidence to each article. For a full description of these evidence-based medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.




Similar content being viewed by others
References
McCarthy CM et al (2012) The magnitude of effect of cosmetic breast augmentation on patient satisfaction and health-related quality of life. Plast Reconstr Surg 130(1):218–223
Von Sperlingl ML, Høimyrl H, Finnerupl K, Jensenl TS, Finnerupl NB (2011) Persistent pain and sensory changes following cosmetic breast augmentation. Eur J Pain 15(3):328–332
Buenaventura R, Adlaka R, Sehgal N (2008) Opioid complications and side effects. Pain Phys 11:105–120
Stanley SS, Hoppe IC, Ciminello FS (2012) Pain control following breast augmentation: a qualitative systematic review. Aesthet Surg J 32(8):964–972
Blanco R (2011) The ‘pecs block’: a novel technique for providing analgesia after breast surgery. Anaesthesia 66(9):847–848
Kang CM, Kim WJ, Yoon SH, Cho CB, Shim JS (2007) Postoperative pain control by intercostal nerve block after augmentation mammoplasty. Aesthet Plast Surg 41(5):1031–1036
Fayman M, Beeton A, Potgieter E, Becker PJ (2003) Comparative analysis of bupivacaine and ropivacaine for infiltration analgesia for bilateral breast surgery. Aesthet Plast Surg 27(2):100–103
Tan P et al (2017) A comparison of 4 analgesic regimens for acute postoperative pain control in breast augmentation patients. Ann Plast Surg 78(6S):299–304
Gardiner S, Rudkin G, Cooter R, Field J, Bond M (2012) Paravertebral blockade for day-case breast augmentation: a randomized clinical trial. Anesth Analg 115(5):1053–1059
Wahba SS, Kamal SM (2014) Thoracic paravertebral block versus pectoral nerve block for analgesia after breast surgery. Egypt J Anaesth 30(2):129–135
Kulhari S, Bharti N, Bala I, Arora S, Singh G (2016) Efficacy of pectoral nerve block versus thoracic paravertebral block for postoperative analgesia after radical mastectomy: a randomized controlled trial. BJA Br J Anaesth 117(3):382–386
Bashandy GMN, Abbas DN (2015) Pectoral nerves I and II blocks in multimodal analgesia for breast cancer surgery: a randomized clinical trial. Reg Anesth Pain Med 40(1):68–74
Morioka H, Kamiya Y, Yoshida T, Baba H (2015) Pectoral nerve block combined with general anesthesia for breast cancer surgery: a retrospective comparison. JA Clin Rep 1(1):15
Karaca O, Pınar HU, Arpacı E, Dogan R, Cok OY, Ahiskalioglu A (2018) The efficacy of ultrasound-guided type-I and type-II pectoral nerve blocks for postoperative analgesia after breast augmentation: a prospective, randomised study. Anaesth Crit Care Pain Med 38:47–52
Benyamin R, Trescot AM, Datta S et al (2008) Opioid complications and side effects. Pain Phys 11(2 Suppl l):S105
Gago Martinez A, Escontrela Rodriguez B, Planas Roca A et al (2016) Intravenous ibuprofen for treatment of post-operative pain: a multicenter, double blind, placebo-controlled, randomized clinical trial. PLoS ONE 11:e0154004
Moore DC (1998) Intercostal nerve block. Int Anesthesiol Clin 36(4):29–42
Giebler RM, Scherer RU, Peters J (1997) Incidence of neurologic complications related to thoracic epidural catheterization. Anesthesiol J Am Soc Anesthesiol 86(1):55–63
Stone MB, Carnell J, Fischer JW, Herring AA, Nagdev A (2011) Ultrasound-guided intercostal nerve block for traumatic pneumothorax requiring tube thoracostomy. Am J Emerg Med 29:697
Hardy PAJ (1988) Anatomical variation in the position of the proximal intercostal nerve. BJA Br J Anaesth 61(3):338–339
Nasr M, Jabbour H, Habre SB (2017) Intercostal nerve block versus bupivacaine pectoralis major infiltration in subpectoral breast augmentation: a randomized controlled trial. Leban Med J 103(4152):1–5
Butterworth JF, Mackey DC, Wasnick JD (2013) Morgan and Mikhail’s clinical anesthesiology, vol 272, 5th edn. McGraw-Hill, New York
Freysz M, Beal JL, Timour Q, Bertrix L, Faucon G (1988) Systemic toxicity of local anesthetics. Pharmacokinetic and pharmacodynamic factors. In: Annales francaises d’anesthesie et de reanimation, vol 7, no 3, pp 181–188
Cros J et al (2018) Pectoral I block does not improve postoperative analgesia after breast cancer surgery: a randomized, double-blind, dual-centered controlled trial. Reg Anesth Pain Med 43(6):596–604
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Ethical Approval
The study received approval from the local ethics committee.
Informed Consent
Informed consent was obtained from all patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ekinci, M., Ciftci, B., Celik, E.C. et al. The Efficacy of Different Volumes on Ultrasound-Guided Type-I Pectoral Nerve Block for Postoperative Analgesia After Subpectoral Breast Augmentation: A Prospective, Randomized, Controlled Study. Aesth Plast Surg 43, 297–304 (2019). https://doi.org/10.1007/s00266-019-01322-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-019-01322-8