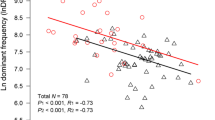
Abstract
Ambotuy
Favoring males’ specific sexual signals, female preferences play a major role in frogs’ evolutionary process by selecting traits linked to those signals. However, the factors constraining and determining those preferences are scarcely explored in an evolutionary background. Here, through a phylogenetic comparative approach we check whether anuran species phylogenetic proximity and calling site predicts female preferences for dominant frequency and whether those preferences influence species sexual size dimorphism. Our hypotheses are as follows: 1) closer species have similar females’ preferences related to the dominant frequency of the partners’ calls; 2) the calling site influences sound propagation and consequently the preference of females for the dominant frequency of the males’ calls; and 3) the preference for calls with low dominant frequency influences the size of the males and consequent reduction of the biSased dimorphism for females. We did not find support for our hypotheses, neither for the influence of phylogenetic proximity nor for calling site determining these preferences. Moreover, female preferences did not impact on species sexual size dimorphism. Besides shedding light into our hypotheses, this study represents a considerable advance on evolutionary studies of female preferences in anura, which still lacks broad species comparative approaches. Furthermore, we suggest future studies to expand knowledge regarding frogs’ female preferences.
Significance statement
This study advances our comprehension of female preferences in frogs by investigating the factors that shape these preferences and their implications for species sexual size dimorphism. Utilizing phylogenetic comparative methods, an approach rarely used in the context of anuran female preferences, this study represents a significant step in applying broad comparative approaches in this field. Highlighting the complex nature of mate choice and its relationship to morphology, soundscape, and phylogeny, we present important insights into evolutionary hypotheses related to female preferences. Lastly, we provide advice on how future studies could further explore this topic in a broader comparative framework while also discussing the limitations of available data on anuran mating preferences.



Similar content being viewed by others
Data availability
Data generated or analyzed during this study comes from a literature survey; therefore, in addition to the data used, we provide complete references for the studies from which they are derived in the supplementary data.
References
Andersson M, Iwasa Y (1996) Sexual selection. Trends Ecol Evol 11:53–58. https://doi.org/10.1016/0169-5347(96)81042-1
Andersson M, Simmons LW (2006) Sexual selection and mate choice. Trends Ecol Evol 21:296–302. https://doi.org/10.1016/j.tree.2006.03.015
Arato J, Fitch WT (2021) Phylogenetic signal in the vocalizations of vocal learning and vocal non-learning birds. Phil Trans R Soc B 376:20200241
Backwell PRY, Passmore NI (1990) Suitable approach perches affect female phonotaxis in an arboreal frog. Herpetologica 46:11–14
Baugh AT, Ryan MJ (2011) The relative value of call embellishment in túngara frogs. Behav Ecol Sociobiol 65:359–367. https://doi.org/10.1007/s00265-010-1053-6
Baugh AT, Akre KL, Ryan MJ (2008) Categorical perception of a natural, multivariate signal: mating call recognition in túngara frogs. P Natl Acad Sci USA 105:8985–8988
Bee MA (2008) Parallel female preferences for call duration in a diploid ancestor of an allotetraploid treefrog. Anim Behav 76:845–853. https://doi.org/10.1016/j.anbehav.2008.01.029
Bee MA, Micheyl C (2008) The cocktail party problem: what is it? How can it be solved? And why should animal behaviorists study it? J Comp Psychol 122:235–251. https://doi.org/10.1037/0735-7036.122.3.235
Bosch J, Márquez R (2001) Call timing in male-male acoustical interactions and female choice in the midwife toad Alytes obstetricans. Copeia 2001:169–177. https://doi.org/10.1643/0045-8511(2001)001[0169:ctimma]2.0.co;2
Bosch J, Márquez R (2005) Female preference intensities on different call characteristics and symmetry of preference above and below the mean in the Iberian midwife toad Alytes cisternasii. Ethology 111:323–333. https://doi.org/10.1111/j.1439-0310.2004.01058.x
Brumm H, Slater PJB (2006) Animals can vary signal amplitude with receiver distance: evidence from zebra finch song. Anim Behav 72:699–705. https://doi.org/10.1016/j.anbehav.2006.01.020
Candolin U, Heuschele J (2008) Is sexual selection beneficial during adaptation to environmental change? Trends Ecol Evol 23:446–452. https://doi.org/10.1016/j.tree.2008.04.008
Colleye O, Vandewalle P, Lanterbecq D, Lecchini D, Parmentier E (2011) Interspecific variation of calls in clownfishes: degree of similarity in closely related species. BMC Evol Biol 11:365. https://doi.org/10.1186/1471-2148-11-365
Cope ED (1864) Contributions to the herpetology of tropical America. Proc Acad Natl Sci Phila 16:166–181
Cope ED (1874) Description of some species of reptiles obtained by Dr. John F. Bransford, Assistant Surgeon United States Navy, while attached to the Nicaraguan Surveying Expedition in 1873. Proceedings of the Academy of Natural Sciences of Philadelphia 26:64–72
da Rocha SMC, Lima AP, Kaefer IL (2018) Territory size as a main driver of male-mating success in an Amazonian nurse frog (Allobates paleovarzensis, Dendrobatoidea). Acta Ethol 21:51–57. https://doi.org/10.1007/s10211-017-0280-5
Dawson B, Ryan MJ (2009) Early experience leads to changes in the advertisement calls of male Physalaemus pustulosus. Copeia 2009:221–226. https://doi.org/10.1643/CE-07-254
Ey E, Rahn C, Hammerschmidt K, Fischer J (2009) Wild female olive baboons adapt their grunt vocalizations to environmental conditions. Ethology 115:493–503
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15
Fisher RA (1930) The genetical theory of natural selection. Clarendon Press, Oxford
Fitch WT, Hauser MD (2003) Unpacking “honesty”: vertebrate vocal production and evolution of acoustic signals. Acoust Commun 65–137. https://doi.org/10.1007/0-387-22762-8_3
Forrest TG (1994) From sender to receiver: propogation and environmental effects on acoustic signals. Am Zool 34:644–654. https://doi.org/10.1093/icb/34.6.644
Frost DR (2020) Amphibian species of the world: an online reference, version 6.0. In: American Museum of Natural History, New York, USA, http://research.amnh.org/vz/herpetology/amphibia/%5Cnhttp://research.amnh.org/herpetology/amphibia/index.html
Gerhardt HC (1978) Temperature coupling in the vocal communication system of the gray tree frog, Hyla versicolor. Science 199:992–994. https://doi.org/10.1126/science.199.4332.992
Gerhardt HC, Huber F (2002) Acoustic communication in insects and anurans: common problems and diverse solutions. The University of Chicago Press, Chicago
Gerhardt HC, Mudry KM (1980) Temperature effects on frequency preferences and mating call frequencies in the green treefrog, Hyla cinerea (Anura: Hylidae). J Comp Physiol 137:1–6. https://doi.org/10.1007/BF00656911
Gingras B, Boeckle M, Herbst CT, Fitch WT (2013a) Call acoustics reflect body size across four clades of anurans. J Zool 289(2):143–150. https://doi.org/10.1111/j.1469-7998.2012.00973.x
Gingras B, Mohandesan E, Boko D, Fitch WT (2013b) Phylogenetic signal in the acoustic parameters of the advertisement calls of four clades of anurans. BMC Evol Biol 13:134. https://doi.org/10.1186/1471-2148-13-134
Godin J-GJ, Briggs SE (1996) Female mate choice under predation risk in the guppy. Anim Behav 51:117–130. https://doi.org/10.1006/anbe.1996.0010
Goicoechea N, De La Riva I, Padial JM (2010) Recovering phylogenetic signal from frog mating calls. Zool Scr 39:141–154. https://doi.org/10.1111/j.1463-6409.2009.00413.x
Goutte S, Dubois A, Howard SD, Marquez R, Rowley JJL, Dehling JM, Grandcolas P, Rongchuan X, Legendre F (2016) Environmental constraints and call evolution in torrent-dwelling frogs. Evolution 70:811–826. https://doi.org/10.1111/evo.12903
Goutte S, Dubois A, Howard SD, Márquez R, Rowley JJL, Dehling JM, Grandcolas P, Xiong RC, Legendre F (2018) How the environment shapes animal signals: a test of the acoustic adaptation hypothesis in frogs. J Evol Biol 31:148–158. https://doi.org/10.1111/jeb.13210
Halfwerk W, Smit JAH, Loning H, Lea AM, Geipel I, Ellers J, Ryan MJ (2017) Environmental conditions limit attractiveness of a complex sexual signal in the túngara frog. Nat Commun 8:1891. https://doi.org/10.1038/s41467-017-02067-1
Han X, Fu J (2013) Does life history shape sexual size dimorphism in anurans? A Comparative Analysis BMC Evol Biol 13:27. https://doi.org/10.1186/1471-2148-13-27
Hardt B, Benedict L (2020) Can you hear me now? A review of signal transmission and experimental evidence for the acoustic adaptation hypothesis. Bioacoustics 30:716–742. https://doi.org/10.1080/09524622.2020.1858448
Harmon LJ, Weir JT, Brock CD, Glor RE, Challenger W (2008) GEIGER: investigating evolutionary radiations. Bioinformatics 24:129–131. https://doi.org/10.1093/bioinformatics/btm538
Hepp F, Lourenço ACC, Pombal JP (2017) Bioacoustics of four Scinax species and a review of acoustic traits in the Scinax catharinae species group (Amphibia: Anura: Hylidae). Salamandra 53:212–230
Höbel G (2000) Reproductive ecology of Hyla rosenbergi in Costa Rica. Herpetologica 56:446–454
Höbel G, Gerhardt HC (2003) Reproductive character displacement in the acoustic communication system of green tree frogs (Hyla cinerea). Evolution 57:894–904. https://doi.org/10.1111/j.0014-3820.2003.tb00300.x
Houde AE, Endler JA (1990) Correlated evolution of female mating preferences and male color patterns in the guppy Poecilia reticulata. Science 248:1405–1408. https://www.jstor.org/stable/2874454
Howard RD (1978) The evolution of mating strategies in bullfrogs, Rana catesbeiana. Evolution 32:850–871. https://doi.org/10.2307/2407499
Jetz W, Pyron RA (2018) The interplay of past diversification and evolutionary isolation with present imperilment across the amphibian tree of life. Nat Ecol Evol 2:850–858. https://doi.org/10.1038/s41559-018-0515-5
Köhler J, Jansen M, Rodríguez A, Kok PJR, Toledo LF, Emmrich M, Glaw F, Haddad CFB, Rödel M-O, Vences M (2017) The use of bioacoustics in anuran taxonomy: theory, terminology, methods and recommendations for best practice. Zootaxa 4251:1–124
Laird KL, Clements P, Hunter KL, Taylor RC (2016) Multimodal signaling improves mating success in the green tree frog (Hyla cinerea), but may not help small males. Behav Ecol Sociobiol 70:1517–1525. https://doi.org/10.1007/s00265-016-2160-9
Lardner B, Lakim MB (2004) Female call preferences in tree-hole frogs: why are there so many unattractive males? Anim Behav 68:265–272. https://doi.org/10.1016/j.anbehav.2004.05.003
Levy DL, Heald R (2015) Biological scaling problems and solutions in amphibians. CSH Perspect Biol 8:a019166. https://doi.org/10.1101/cshperspect.a019166
Maan ME, Cummings ME (2009) Sexual dimorphism and directional sexual selection on aposematic signals in a poison frog. P Natl Acad Sci USA 106:19072–19077. https://doi.org/10.1073/pnas.0903327106
Márquez R (1993) Male reproductive success in two midwife toads, Alytes obstetricans and A. cisternasii. Behav Ecol Sociobiol 32:283–291. https://doi.org/10.1007/BF00166518
Márquez R, Bosch J (1997) Male advertisement call and female preference in sympatric and allopatric midwife toads. Anim Behav 54:1333–1345. https://doi.org/10.1006/anbe.1997.0529
Marquez R (1995) Female choice in the midwife toads (Alytes obstetricans and A. cisternasii). Behaviour 91:1689–1699. https://doi.org/10.1017/CBO9781107415324.004
Marten K, Marler P (1977) Sound transmission and its significance for animal vocalization. I Temperate Habitats Behav Ecol Sociobiol 2:271–290. https://doi.org/10.1007/BF00299740
McLean MJ, Bishop PJ, Nakagawa S (2012) Male quality, signal reliability and female choice: assessing the expectations of inter-sexual selection. J Evol Biol 25:1513–1520. https://doi.org/10.1111/j.1420-9101.2012.02533.x
Milinski M, Bakker TCM (1992) Costs influence sequential mate choice in sticklebacks, Gasterosteus aculeatus. Proc R Soc Lond B 250:229–233. https://doi.org/10.1098/rspb.1992.0153
Monnet J-M, Cherry MI (2002) Sexual size dimorphism in anurans. Proc R Soc Lond B 269:2301–2307. https://doi.org/10.1098/rspb.2002.2170
Moreno-Gómez FN, Bacigalupe LD, Silva-Escobar AA, Soto-Gamboa M (2015) Female and male phonotactic responses and the potential effect of sexual selection on the advertisement calls of a frog. Anim Behav 104:79–86. https://doi.org/10.1016/j.anbehav.2015.03.010
Morton ES (1975) Ecological sources of selection on avian sounds. Am Nat 109:17–34
Muñoz MI, Goutte S, Ellers J, Halfwerk W (2020) Environmental and morphological constraints interact to drive the evolution of communication signals in frogs. bioRxiv. https://doi.org/10.1101/2020.04.18.047936
Naguib M, Wiley RH (2001) Estimating the distance to a source of sound: mechanisms and adaptations for long-range communication. Anim Behav 62:825–837. https://doi.org/10.1006/anbe.2001.1860
Nali RC, Zamudio KR, Haddad CFB, Prado CPA (2014) Size-dependent selective mechanisms on males and females and the evolution of sexual size dimorphism in frogs. Am Nat 184:727–740. https://doi.org/10.1086/678455
Peters WCH (1867) Herpetologische Notizen. Monatsberichte der Königlichen Preussische Akademie des Wissenschaften zu Berlin 1867:13–37
Pincheira-Donoso D, Harvey LP, Grattarola F, Jara M, Cotter SC, Tregenza T, Hodgson DJ (2020) The multiple origins of sexual size dimorphism in global amphibians. Glob Ecol Biogeogr 30:443–458. https://doi.org/10.1111/geb.13230
Ptacek MB (2000) The role of mating preferences in shaping interspecific divergence in mating signals in vertebrates. Behav Process 51:111–134. https://doi.org/10.1016/S0376-6357(00)00123-6
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org
Reby D, McComb K (2003) Anatomical constraints generate honesty: acoustic cues to age and weight in the roars of red deer stags. Anim Behav 65:519–530. https://doi.org/10.1006/anbe.2003.2078
Revell LJ (2012) phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol Evol 3:217–223. https://doi.org/10.1111/j.2041-210X.2011.00169.x
Richards DG, Wiley RH (1980) Reverberations and amplitude fluctuations in the propagation of sound in a forest: implications for animal communication. Am Nat 115:381–399
Richardson C, Joly P, Léna J-P, Plénet S, Lengagne T (2010) The challenge of finding a high-quality male: a treefrog solution based on female assessment of male calls. Behaviour 147:1737–1752. https://doi.org/10.1163/000579510X530221
Richards-Zawacki CL, Wang IJ, Summers K (2012) Mate choice and the genetic basis for colour variation in a polymorphic dart frog: inferences from a wild pedigree. Mol Ecol 21:3879–3892. https://doi.org/10.1111/j.1365-294X.2012.05644.x
Robertson JGM (1986) Female choice, male strategies and the role of vocalizations in the Australian frog Uperoleia rugosa. Anim Behav 34:773–784. https://doi.org/10.1016/S0003-3472(86)80061-6
Römer H (1992) Ecological constraints for the evolution of hearing and sound communication in insects. In: Webster DB, Popper AN, Fay RR (eds) The Evolutionary Biology of Hearing. Springer, New York
Rosso A, Castellano S, Giacoma C (2006) Preferences for call spectral properties in Hyla intermedia. Ethology 112:599–607. https://doi.org/10.1111/j.1439-0310.2005.01186.x
Ryan MJ, Rand AS (1993) Sexual selection and signal evolution: the ghost of biases past. Phil Trans R Soc B 340:187–195. https://doi.org/10.1098/rstb.1993.0057
Ryan MJ, Rand AS (2003) Sexual selection in female perceptual space: how female túngara frogs perceive and respond to complex population variation in acoustic mating signals. Evolution 57:2608–2618. https://doi.org/10.1111/j.0014-3820.2003.tb01503.x
Schmidt O (1857) Diagnosen neuer Frösche des zoologischen Cabinets zu Krakau. Sitzungsberichte der Kaiserlichen Akademie der Wissenschaften, Mathematisch-Naturwissenschaftliche Classe 24:10–15
Schneider JG (1799) Historia Amphibiorum Naturalis et Literarariae. Fasciculus Primus. Continens Ranas, Calamitas, Bufones, Salamandras et Hydros in Genera et Species Descriptos Notisque suis Distinctos. Jena: Friederici Frommanni
Schrode KM, Ward JL, Vélez A, Bee MA (2012) Female preferences for spectral call properties in the western genetic lineage of Cope’s gray treefrog (Hyla chrysoscelis). Behav Ecol Sociobiol 66:1595–1606. https://doi.org/10.1007/s00265-012-1413-5
Shine R (1979) Sexual selection and sexual dimorphism in the amphibia. Copeia 1979:297–306. https://doi.org/10.2307/1443418
Smith RJ (1999) Statistics of sexual size dimorphism. J Hum Evol 36:423–458. https://doi.org/10.1006/jhev.1998.0281
Summers K, Symula R, Clough M, Cronin T (1999) Visual mate choice in poison frogs. Proc R Soc Lond B 266:2141–2145
Tanner JC, Bee MA (2019) Within-individual variation in sexual displays: signal or noise? Behav Ecol 30:80–91. https://doi.org/10.1093/beheco/ary165
Tonini JFR, Provete DB, Maciel NM, Morais AR, Goutte S, Toledo LF, Pyron RA (2020) Allometric escape from acoustic constraints is rare for frog calls. Ecol Evol 10:3686–3695. https://doi.org/10.1002/ece3.6155
Tung Ho LS, Ané C (2014) A linear-time algorithm for Gaussian and non-Gaussian trait evolution models. Syst Biol 63:397–408. https://doi.org/10.1093/sysbio/syu005
Wells KD (1977) Territoriality and male mating success in the green frog (Rana clamitans). Ecology 58:750–762
Wiley HR (1994) Errors, exaggeration and deception in animal communication. In: Real L (ed) Behavioral mechanisms in ecology. University of Chicago Press, Chicago, pp 157–189
Wiley RH, Richards DG (1978) Physical constraints on acoustic communication in the atmosphere: implications for the evolution of animal vocalizations. Behav Ecol Sociobiol 3:69–94
Williams GC (1966) Adaptation and natural selection. Princeton University Press, Princeton, NJ
Wollerman L, Wiley RH (2002) Background noise from a natural chorus alters female discrimination of male calls in a neotropical frog. Animal Behav 63:15–22. https://doi.org/10.1006/anbe.2001.1885
Wollerman L, Carolina N, Hill C (1998) Stabilizing and directional preferences of female Hyla ebraccata for calls. Anim Behav 55:1619–1630
Yu Y, Hu Y, Zhang Q, Zheng R, Shen B, Kong S, Li K (2020) Female preferences for call properties of giant spiny frog (Quasipaa spinosa). Pak J Zool 52:825–834. https://doi.org/10.17582/journal.pjz/20180503100508
Zahavi A (1975) Mate selection—a selection for a handicap. J Theor Biol 53:205–214. https://doi.org/10.1016/0022-5193(75)90111-3
Zhu B, Wang J, Zhao L, Sun Z, Brauth SE, Tang Y, Cui J (2016) Bigger is not always better: females prefer males of mean body size in Philautus odontotarsus. PLoS ONE 11:e0149879
Zimmerman BL (1983) A comparison of structural features of calls of open and forest habitat frog species in the Central Amazon. Herpetologica 39:235–246
Acknowledgements
We are thankful for the reviewers’ comments and feedback during the submission of this manuscript.
Funding
This work was supported by a Coordenação de Aperfeiçoamento de Pessoal e Nível Superior (CAPES) PhD scholarship to JVB and IM; Global Marie S. Curie Fellowship (EAVESTROP-661408 to DL) granted by the European Commission (Program H2020); Conselho Nacional de Desenvolvimento Científico (CNPq grant number 309894/2017–4 to RPB and 303467/2021–5 to JAFD-F); and Fundação de Âmparo a Pesquisa do Estação de Goiás/Conselho Nacional de Desenvolvimento Científico/Programa de Apoio aos Núcleos de Excelência (FAPEG/CNPq/PRONEX 201710267000507 to RPB). This work was developed in the context of the National Institutes for Science and Technology (INCT) in Ecology, Evolution, and Biodiversity Conservation (CNPq proc. 465610/2014–5 and FAPEG proc. 201810267000023).
Author information
Authors and Affiliations
Contributions
Conceptualization: JVB, IM, DL, and RPB; methodology: JVB, IM, and JAFD-F; formal analysis and investigation: JVB and IM; writing—original draft preparation: JVB and IM; writing—review and editing: JVB, IM, DL, JAFD-F, and RPB.
Corresponding author
Ethics declarations
Ethics approval
This study uses data from a literature survey and, therefore, does not involve any direct observation or manipulation of animals by the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by A. Taylor Baugh.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bernardy, J.V., Melo, I., Llusia, D. et al. Female preferences for dominant frequency in frogs: constraints and impact on sexual size dimorphism. Behav Ecol Sociobiol 78, 4 (2024). https://doi.org/10.1007/s00265-023-03418-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-023-03418-3