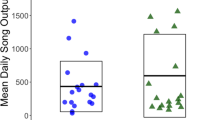
Abstract
The use of songs for mate-attraction is common. Intensive songs may indicate high energetic investment, reflecting an individual’s resource-holding potential and attractiveness as a prospective mate. Consequently, there can be a direct relationship between song metrics and lifetime reproductive success. While singing is held to be energetically costly, quantitative studies in mammals are lacking. Here, we present an exploratory analysis of energetic costs in a singing bat (Mystacina tuberculata). We recorded the songs of 12 male bats and quantified skin temperature (Tsk) responses using temperature telemetry to estimate energy expenditure. We hypothesised that singing would be energetically costly and predicted correlations between Tsk and song duty cycle and between duty cycle and body size. Contrary to our expectations, we found estimated energetic expenditure while singing to be comparatively low. We also found no relationship between estimated energy expenditure and duty cycle, and neither estimated energy expenditure nor duty cycle was correlated with body size. Our results suggest that energetic costs of singing in bats may be lower than previously assumed, and that song output may convey only limited fitness information.
Significance statement
Song is commonly used to communicate information related to mate-attraction or territory defence. Some aspects of song production require more energy to produce, making them an honest signal of a singer’s investment. While our knowledge of bird song and its relationship to mating success is well developed, a similar understanding regarding mammalian song is severely lacking. Numerous bat species produce song, yet we know little about the energetics of song production in this large and diverse order. Using temperature telemetry, we estimate the costs of singing in a free-living lek-breeding bat. To our knowledge, this is the first study to estimate the energetic costs of song production in a mammal.


Similar content being viewed by others
Data availability
All data generated or analysed during this study have been made available as Electronic Supplementary Material.
References
Aschoff J (1982) The circadian rhythm of body temperature as a function of body size. In: Taylor CR, Johansen R, Bolis L (eds) A companion to animal physiology. Cambridge University Press, Cambridge, pp 173–188
Audet D, Thomas DW (1996) Evaluation of the accuracy of body temperature measurement using external radio transmitters. Can J Zool 74:1778–1781
Barclay RM, Kalcounis MC, Crampton LH, Stefan C, Vonhof MJ, Wilkinson L, Brigham RM (1996) Can external radiotransmitters be used to assess body temperature and torpor in bats? J Mammal 77:1102–1106
Barclay RM, Lausen CL, Hollis L (2001) What’s hot and what’s not: defining torpor in free-ranging birds and mammals. Can J Zool 79:1885–1890. https://doi.org/10.1139/cjz-79-10-1885
Barón Birchenall L (2016) Animal communication and human language: an overview. Int J Comp Psychol 29:28000. https://psycnet.apa.org/record/2016-57975-001
Behr O, von Helversen O, Heckel G, Nagy M, Voigt CC, Mayer F (2006) Territorial songs indicate male quality in the sac-winged bat Saccopteryx bilineata (Chiroptera, Emballonuridae). Behav Ecol 17:810–817. https://doi.org/10.1093/beheco/arl013
Boyles JG, Levesque DL, Nowack J, Wojciechowski MS, Stawski C, Fuller A, Smit B, Tattersall GJ (2019) An oversimplification of physiological principles leads to flawed macroecological analyses. Ecol Evol 9:12020–12025. https://doi.org/10.1002/ece3.5721
Boyles JG, Smit B, McKechnie AE (2011) A new comparative metric for estimating heterothermy in endotherms. Physiol Biochem Zool 84:115–123. https://doi.org/10.1086/656724
Catchpole CK, Slater PJB (2008) Bird song: biological themes and variations, 2nd edn. Cambridge University Press, Cambridge
Chaverri G, Iturralde-Polit P, Herrera NIS, Romero A, Chaves-Ramírez S, Sagot M (2021) The energetics of social signaling during roost location in Spix’s disc-winged bats. J Exp Biol 224:jeb238279. https://doi.org/10.1242/jeb.238279
Christie JE, Simpson W (2006) Influence of winter weather conditions on lesser short-tailed bat (Mystacina tuberculata) activity in Nothofagus forest, Fiordland. NZ J Zool 33:133–140. https://doi.org/10.1080/03014223.2006.9518437
Currie SE, Boonman A, Troxell S, Yovel Y, Voigt CC (2020) Echolocation at high intensity imposes metabolic costs on flying bats. Nat Ecol Evol 4:1174–1177. https://doi.org/10.1038/s41559-020-1249-8
Czenze ZJ, Brigham RM, Hickey AJR, Parsons S (2017) Stressful summers? Torpor expression differs between high- and low-latitude populations of bats. J Mammal 98:1249–1255. https://doi.org/10.1093/jmammal/gyx071
Czenze ZJ, Brigham RM, Hickey AJR, Parsons S (2017) Cold and alone? Roost choice and season affect torpor patterns in lesser short-tailed bats. Oecologia 183:1–8. https://doi.org/10.1007/s00442-016-3707-1
Czenze ZJ, Brigham RM, Hickey AJR, Parsons S (2017) Winter climate affects torpor patterns and roost choice in New Zealand lesser short-tailed bats. NZ J Zool 303:236–243. https://doi.org/10.1111/jzo.12486
Czenze ZJ, Thurley T (2018) Weather and demographics affect Dactylanthus flower visitation by New Zealand lesser short-tailed bats. NZ J Ecol 42:80–84. https://doi.org/10.20417/nzjecol.42.8
Davidson SM, Wilkinson GS (2004) Function of male song in the greater white-lined bat, Saccopteryx bilineata. Anim Behav 67:883–891. https://doi.org/10.1016/j.anbehav.2003.06.016
Dechmann DKN, Wikelski M, van Noordwijk HJ, Voigt CC, Voigt-Heucke SL (2013) Metabolic costs of bat echolocation in a non-foraging context support a role in communication. Front Physiol 4:66. https://doi.org/10.3389/fphys.2013.00066
Eckenweber M, Knornschild M (2013) Social influences on territorial signaling in male greater sac-winged bats. Beh Ecol Sociobiol 67:639–648. https://doi.org/10.1007/s00265-013-1483-z
Geiser F (2004) Metabolic rate and body temperature reduction during hibernation and daily torpor. Annu Rev Physiol 66:239–274. https://doi.org/10.1146/annurev.physiol.66.032102.115105
Gil D, Gahr M (2002) The honesty of bird song: multiple constraints for multiple traits. Trends Ecol Evol 17:133–141. https://doi.org/10.1016/S0169-5347(02)02410-2
Hall ML, Kingma SA, Peters A (2013) Male songbird indicates body size with low-pitched advertising songs. PLoS ONE 8:e56717. https://doi.org/10.1371/journal.pone.0056717
Hauser MD, Chomsky N, Fitch WT (2002) The faculty of language: what is it, who has it, and how did it evolve? Science 298:1569–1579. https://doi.org/10.1017/CBO9780511817755.002
Herman LM (2017) The multiple functions of male song within the humpback whale (Megaptera novaeangliae) mating system: review, evaluation, and synthesis. Biol Rev 92:1795–1818. https://doi.org/10.1111/brv.12309
Ilany A, Barocas A, Kam M, Ilany T, Geffen E (2013) The energy cost of singing in wild rock hyrax males: evidence for an index signal. Anim Behav 85:995–1001. https://doi.org/10.1016/j.anbehav.2013.02.023
Jonasson KA, Willis CKR (2012) Hibernation energetics of free-ranging little brown bats. J Exp Biol 215:2141–2149. https://doi.org/10.1242/jeb.066514
Kenagy GJ, Vleck D (1982) Daily temporal organization of metabolism in small mammals: adaptation and diversity. In: Aschoff J, Daan S, Groos GA (eds) Vertebrate circadian systems: structure and physiology. Springer-Verlag, Berlin, pp 322–338
Kerth G, Almasi B, Ribi N, Thiel D, Lüpold S (2003) Social interactions among wild female Bechsteins bats (Myotis bechsteinii) living in a maternity colony. Acta Ethol 5:107–114. https://doi.org/10.1007/s10211-003-0075-8
Kunz TH, Lumsden LF (2003) Ecology of cavity and foliage roosting bats. In: Kunz TH, Fenton MB (eds) Bat ecology. University of Chicago Press, Chicago, pp 3–89
Lenth RV, Buerkner P, Herve M, Love J, Riebl H, Singmann H (2021) Emmeans: estimated marginal means. R package. Version 1.6.1. https://www.rdocumentation.org/packages/emmeans/versions/1.6.1. Accessed 14/07/2021
Linhart P, Fuchs R (2015) Song pitch indicates body size and correlates with males’ response to playback in a songbird. Anim Behav 103:91–98. https://doi.org/10.1016/j.anbehav.2015.01.038
Lloyd BD (2001) Advances in New Zealand mammalogy 1990–2000: short-tailed bats. J Roy Soc New Zeal 31:111–125. https://doi.org/10.1080/03014223.2001.9517642
McNab BK, O’Donnell C (2018) The behavioral energetics of New Zealand’s bats: daily torpor and hibernation, a continuum. Comp Biochem Physiol A 223:18–22. https://doi.org/10.1016/j.cbpa.2018.05.001
Oberweger K, Goller F (2001) The metabolic cost of birdsong production. J Exp Biol 204:3379–3388. https://doi.org/10.1242/jeb.204.19.3379
Ophir AG, Schrader SB, Gillooly JF (2010) Energetic cost of calling: general constraints and species-specific differences. J Evol Biol 23:1564–1569. https://doi.org/10.1111/j.1420-9101.2010.02005.x
Pedersen MB, Fahlman A, Borque-Espinosa A, Madsen PT, Jensen FH (2020) Whistling is metabolically cheap for communicating bottlenose dolphins (Tursiops truncatus). J Exp Biol 223:jeb212498. https://doi.org/10.1242/jeb.212498
Prestwich KN (1994) The energetics of acoustic signaling in anurans and insects. Am Zool 34:625–643
Puechmaille SJ, Borissov IM, Zsebok S, Allegrini B, Hizem M, Kuenzel S, Schuchmann M, Teeling EC, Siemers BM (2014) Female mate choice can drive the evolution of high frequency echolocation in bats: a case study with Rhinolophus mehelyi. PLoS ONE 9:e103452. https://doi.org/10.1371/journal.pone.0103452
Read AF, Weary DM (1992) The evolution of bird song: comparative analyses. Phil Trans R Soc B 338:165–187. https://doi.org/10.1098/rstb.1992.0137
Ruf T, Geiser F (2015) Daily torpor and hibernation in birds and mammals. Biol Rev 90:891–926. https://doi.org/10.1111/brv.12137
Saino N, Galeotti P, Sacchi R, Møller AP (1997) Song and immunological condition in male barn swallows (Hirundo rustica). Behav Ecol 8:364–371. https://doi.org/10.1093/beheco/8.4.364
Scholander P, Hock R, Walters V, Irving L (1950) Adaptation to cold in Arctic and tropical mammals and birds in relation to body temperature, insulation and basal metabolic rate. Biol Bull 99:259–271. https://doi.org/10.2307/1538742
Smarsh GC, Smotherman M (2015) Singing away from home: songs are used on foraging territories in the African megadermatid bat, Cardioderma cor. In: Proceedings of meetings on acoustics. The Acoustical Society of America, Jacksonville, FL, pp 1–11. https://doi.org/10.1121/2.0000131
Smotherman M, Knörnschild M, Smarsh G, Bohn K (2016) The origins and diversity of bat songs. J Comp Physiol A 202:535–554. https://doi.org/10.1007/s00359-016-1105-0
Speakman JR, Racey PA (1991) No cost of echolocation for bats in flight. Nature 350:421–423. https://doi.org/10.1038/350421a0
Toth CA (2016) Lek breeding in the lesser short-tailed bat (Mystacina tuberculata): male courtship, female selection, and the determinants of reproductive strategies. PhD thesis, The University of Auckland
Toth CA, Dennis TE, Pattemore DE, Parsons S (2015) Females as mobile resources: communal roosts promote the adoption of lek breeding in a temperate bat. Behav Ecol 26:1156–1163. https://doi.org/10.1093/beheco/arv070
Toth CA, Parsons S (2018) The high-output singing displays of a lekking bat encode information on body size and individual identity. Behav Ecol Sociobiol 72:102. https://doi.org/10.1007/s00265-018-2496-4
Toth CA, Santure AW, Holwell GI, Pattemore DE, Parsons S (2018) Courtship behaviour and display-site sharing appears conditional on body size in a lekking bat. Anim Behav 136:13–19. https://doi.org/10.1016/j.anbehav.2017.12.007
Vernes SC, Wilkinson GS (2020) Behaviour, biology and evolution of vocal learning in bats. Phil Trans R Soc B 375:20190061. https://doi.org/10.1098/rstb.2019.0061
Voigt CC, Behr O, Caspers B, von Helversen O, Knörnschild M, Mayer F, Nagy M (2008) Songs, scents, and senses: sexual selection in the greater sac-winged bat, Saccopteryx bilineata. J Mammal 89:1401–1410. https://doi.org/10.1644/08-mamm-s-060.1
Voigt CC, Lewanzik D (2012) ‘No cost of echolocation for flying bats’ revisited. J Comp Physiol B 182:831–840. https://doi.org/10.1007/s00360-012-0663-x
Walsberg G, Wolf B (1995) Variation in the respiratory quotient of birds and implications for indirect calorimetry using measurements of carbon dioxide production. J Exp Biol 198:213–219. https://doi.org/10.1242/jeb.198.1.213
Ward S, Lampe HM, Slater PJB (2004) Singing is not energetically demanding for pied flycatchers, Ficedula hypoleuca. Behav Ecol 15:477–484. https://doi.org/10.1093/beheco/arh038
Ward S, Slater PJB (2005) Heat transfer and the energetic cost of singing by canaries Serinus canaria. J Comp Physiol A 191:953–964. https://doi.org/10.1007/s00359-005-0022-4
Wilkinson GS (1986) Social grooming in the common vampire bat, Desmodus rotundus. Anim Behav 34:1880–1889. https://doi.org/10.1016/S0003-3472(86)80274-3
Willis CKR, Brigham RM (2003) Defining torpor in free-ranging bats: experimental evaluation of external temperature-sensitive radiotransmitters and the concept of active temperature. J Comp Physiol B 173:379–389. https://doi.org/10.1007/s00360-003-0343-y
Willis CKR, Cooper CE (2009) Techniques for studying thermoregulation and thermal biology in bats. In: Kunz TH, Parsons S (eds) Ecological and behavioural methods for the study of bats. John Hopkins University Press, Baltimore, MD, pp 646–657
Acknowledgements
The authors would like to thank T. Thurley, A. Baddeley and T. Wright for assistance in the field, the New Zealand Department of Conservation for housing in Pureora and Te Maru o Rereahu Iwi for their support of the project. The authors would also like to thank A.E. McKechnie and M.S. Wojciechowski for helpful discussions about analyses, and two anonymous reviewers whose comments on an earlier version of the manuscript greatly improved its quality.
Funding
KC was supported to undertake this work by a University of Auckland Doctoral Scholarship.
Author information
Authors and Affiliations
Contributions
All authors were involved in conception and design of the experiment. KC conducted the field work. KC and ZJC analysed the data. KC, ZJC and SP wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval
All applicable international, national and institutional guidelines for the use of animals were followed. All procedures were approved by the University of Auckland Animal Ethics Committee (AEC-R1782) and by the New Zealand Department of Conservation (50435-FAU).
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by M. Knoernschild
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Collier, K., Parsons, S. & Czenze, Z.J. Thermal energetics of male courtship song in a lek-breeding bat. Behav Ecol Sociobiol 76, 36 (2022). https://doi.org/10.1007/s00265-022-03141-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03141-5