Abstract
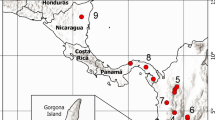
Signal divergence and sensory preferences may lead to sexual isolation and eventually promote speciation between animal populations. However, few studies have quantified the degree of chemical signal divergence and scent-mediated sexual isolation in lizard populations. Geographic and ecological variations among populations of the whiptail lizard Aspidoscelis lineattissimus suggest that there might be chemical signal divergence among these populations. Here, we used gas chromatography-mass spectrometry to characterize and compare the chemical composition of the femoral gland secretions of male whiptail lizards of four populations from a western region of Mexico and, through behavioral experiments, explored the effects of lizard scents on precopulatory behaviors and intrasexual male-male chemical recognition among populations. Our results showed that males of each population contain a divergent mixture of compounds in their femoral gland secretions. Differential chemosensory behavior indicated that male and female lizards discriminated and were more attracted to scents of lizards from their same population. Although females also seem to discriminate male scents between populations, their associated preference to territories scent-marked by males of their own population is different between regions (eastern vs western) and not between populations. We suggest that between some populations of A. lineattissimus there may be partial premating isolation mediated by chemical signals and behavioral divergence.
Significance statement
Geographic variation in sexual signals can strongly affect discrimination and recognition abilities among reproductive individuals from divergent populations, resulting in sexual isolation and speciation. Studies suggest that reproductive isolation and speciation in lizard systems may be mediated by chemical signals, male mate preferences, and male-male interactions but not by female mate preferences. Using gas chromatography-mass spectrometry techniques and behavioral experiments, we found that chemical divergence in femoral gland secretions of male Aspidoscelis lineattissimus influences behavioral discrimination among four distinct populations. Males and females recognized and responded more toward lizard scents from their own population. In addition, some female populations were able to discriminate between territories scent-marked by males from different populations. We suggest that chemical and behavioral differences between populations may influence partial premating isolation, which can be mediated by inter and intrasexual interactions.





Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aitchison J (1986) The statistical analysis of compositional data: monographs in statistics and applied probability. Chapman and Hall, London
Alberts AC (1992) Constraints on the design of chemical communication systems in terrestrial vertebrates. Am Nat 139:62–89. https://doi.org/10.1086/285305
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x
Anderson MJ, Willis TJ (2003) Canonical analysis of principal coordinates: a useful method of constrained ordination for ecology. Ecology 84:511–525. https://doi.org/10.1890/0012-9658(2003)084[0511:CAOPCA]2.0.CO;2
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA for PRIMER: guide to software and statistical methods. PRIMER-E Ltd, Plymouth
Baeckens S, Martín J, García-Roa R, Pafilis P, Huyghe K, Van Damme R (2018) Environmental conditions shape the chemical signal design of lizards. Funct Ecol 32:566–580. https://doi.org/10.1111/1365-2435.12984
Barbosa D, Font E, Desfilis E, Carretero MA (2006) Chemically mediated species recognition in closely related Podarcis wall lizards. J Chem Ecol 32:1587–1598. https://doi.org/10.1111/1365-2435.12984
Barton NH, De Cara MAR (2009) The evolution of strong reproductive isolation. Evolution 63:1171–1190. https://doi.org/10.1111/j.1558-5646.2009.00622.x
Bates D, Maechler M, Bolker B (2012) Linear mixed-effects models using S4 classes. http://cran.r-project.org/web/packages/lme4/index.html. Accessed 15 Jun 2020
Boughman JW (2001) Divergent sexual selection enhances reproductive isolation in sticklebacks. Nature 411:944–948. https://doi.org/10.1038/35082064
Boughman JW (2002) How sensory drive can promote speciation. Trends Ecol Evol 17:571–577. https://doi.org/10.1016/S0169-5347(02)02595-8
Boughman JW, Rundle HD, Schluter D (2005) Parallel evolution of sexual isolation in sticklebacks. Evolution 59:361–373. https://doi.org/10.1111/j.0014-3820.2005.tb00995.x
Carazo P, Font E, Desfilis E (2007) Chemosensory assessment of rival competitive ability and scent mark function in a lizard (Podarcis hispanica). Anim Behav 74:895–902. https://doi.org/10.1016/j.anbehav.2007.02.01
Carazo P, Font E, Desfilis E (2008) Beyond ‘nasty neighbours’ and ‘dear enemies’? Individual recognition by scent marks in a lizard (Podarcis hispanica). Anim Behav 76:1953–1963. https://doi.org/10.1016/j.anbehav.2008.08.018
Carpenter CC (1962) Patterns of behavior in two Oklahoma lizards. Am Midl Nat 67:132–151. https://doi.org/10.2307/2422824
Castiglia R, Annesi F, Bezerra AM, García A, Flores-Villela O (2010) Cytotaxonomy and DNA taxonomy of lizards (Squamata, Sauria) from a tropical dry forest in the Chamela-Cuixmala Biosphere Reserve on the coast of Jalisco, Mexico. Zootaxa 2508:1–29. https://doi.org/10.11646/zootaxa.2508.1.1
Clarke KR, Gorley RN (2006) PRIMER v6: User manual/tutorial. PRIMER-E Ltd, Plymouth
Cooper WE (1998) Evaluation of swab and related tests as a bioassay for assessing responses by squamate reptiles to chemical stimuli. J Chem Ecol 24:841–866. https://doi.org/10.1023/A:1022373517653
Cooper WE, Burghardt GM (1990) A comparative analysis of scoring methods for chemical discrimination of prey by squamate reptiles. J Chem Ecol 16:45–65. https://doi.org/10.1007/BF01021267
Cooper WE, Vitt LJ (1986) Interspecific odour discrimination among syntopic congeners in scincid lizards (genus Eumeces). Behaviour 97:1–9. https://doi.org/10.1163/156853986X00289
Cooper WE, Paulissen MA, Habegger JJ (2000) Discrimination of prey, but not plant, chemicals by actively foraging, insectivorous lizards, the lacertid Takydromus sexlineatus and the teiid Cnemidophorus gularis. J Chem Ecol 26:1623–1634. https://doi.org/10.1023/A:1005534828701
Coyne JA, Orr HA (2004) Speciation. Sinauer Associates, Sunderland
Crawley MJ (2013) The R book, 2nd edn. Wiley, Chichester
Dietemann V, Peeters C, Liebig J, Thivet V, Hölldobler B (2003) Cuticular hydrocarbons mediate discrimination of reproductives and nonreproductives in the ant Myrmecia gulosa. Proc Natl Acad Sci U S A 100:10341–10346. https://doi.org/10.1073/pnas.1834281100
Duellman WE, Wellman J (1960) A systematic study of the lizards of the deppei group (genus Cnemidophorus) in Mexico and Guatemala. Miscellaneous Publications, No. 111, Museum of Zoology, University of Michigan, Ann Arbor
Faraway JJ (2005) Extending the linear model with R. Generalized linear, mixed effects and nonparametric regression models. Chapman and Hall/CRC, Boca Raton
Gabirot M, Castilla AM, López P, Martín J (2010a) Differences in chemical signals may explain species recognition between an island lizard, Podarcis atrata, and related mainland lizards, P. hispanica. Biochem Syst Ecol 38:521–528. https://doi.org/10.1016/j.bse.2010.05.008
Gabirot M, Castilla AM, López P, Martín J (2010b) Chemosensory species recognition may reduce the frequency of hybridization between native and introduced lizards. Can J Zool 88:73–80. https://doi.org/10.1139/Z09-120
Gabirot M, López P, Martín J (2012) Differences in chemical sexual signals may promote reproductive isolation and cryptic speciation between Iberian wall lizard populations. Int J Evol Biol 2012:698520–698513. https://doi.org/10.1155/2012/698520
Gabirot M, López P, Martín J (2013) Female mate choice based on pheromone content may inhibit reproductive isolation between distinct populations of Iberian wall lizards. Curr Zool 59:210–220. https://doi.org/10.1093/czoolo/59.2.210
García-Roa R, Sáiz J, Gómara B, López P, Martín J (2017) Dietary constraints can preclude the expression of an honest chemical sexual signal. Sci Rep 7:6073. https://doi.org/10.1038/s41598-017-06323-8
Gardner W, Mulvey EP, Shaw EC (1995) Regression analyses of counts and rates: Poisson, overdispersed Poisson, and negative binomial models. Psychol Bull 118:392–404. https://doi.org/10.1037/0033-2909.118.3.392
Güizado-Rodríguez MA, Casas-Andreu G (2011) Facultative specialization in the diet of the twelve-lined whiptail, Aspidoscelis lineatissima. J Herpetol 45:287–290. https://doi.org/10.1670/10-078.1
Heathcote RJP, While GM, MacGregor HEA, Sciberras J, Leroy C, D’Ettorre P, Uller T (2016) Male behaviour drives assortative reproduction during the initial stage of secondary contact. J Evol Biol 29:1003–1015. https://doi.org/10.1111/jeb.12840
Heilbron DC (1994) Zero-altered and other regression models for count data with added zeros. Biometrical J 36:531–547. https://doi.org/10.1002/bimj.4710360505
Henneken J, Goodger JQ, Jones TM, Elgar MA (2017) Diet-mediated pheromones and signature mixtures can enforce signal reliability. Front Ecol Evol 4:145. https://doi.org/10.3389/fevo.2016.00145
Hernández-Salinas U, Ramírez-Bautista A, Pavón NP, Pacheco LFR (2014) Morphometric variation in island and mainland populations of two lizard species from the Pacific Coast of Mexico. Rev Chil Hist Nat 87:21. https://doi.org/10.1186/S40693-014-0021-3
Ibáñez A, Menke M, Quezada G, Jiménez-Uzcátegui G, Schulz S, Steinfartz S (2017) Diversity of compounds in femoral secretions of Galápagos iguanas (genera: Amblyrhynchus and Conolophus), and their potential role in sexual communication in lek-mating marine iguanas (Amblyrhynchus cristatus). PeerJ 5:e3689. https://doi.org/10.7717/peerj.3689
Jackman S (2007) The pscl package. http://cran.rproject.org/src/contrib/Descriptions/pscl.html. Accessed 15 Jun 2020
Kabir MS, Radhika V, Thaker M (2019) Mismatch in receiver responses to multimodal signals in a diurnal gecko. Anim Behav 147:115–123. https://doi.org/10.1016/j.anbehav.2018.11.011
López P, Martín J (2002) Chemical rival recognition decreases aggression levels in male Iberian wall lizards, Podarcis hispanica. Behav Ecol Sociobiol 51:461–465. https://doi.org/10.1007/s00265-001-0447-x
López P, Martín J (2011) Male Iberian rock lizards may reduce the costs of fighting by scent-matching of the resource holders. Behav Ecol Sociobiol 65:1891–1898. https://doi.org/10.1007/s00265-011-1198-y
López P, Martín J, Cuadrado M (2002) Pheromone mediated intrasexual aggression in male lizards, Podarcis hispanicus. Aggressive Behav 28:154–163. https://doi.org/10.1002/ab.90017
López P, Amo L, Martín J (2006) Reliable signaling by chemical cues of male traits and health state in male lizards, Lacerta monticola. J Chem Ecol 32:473–488. https://doi.org/10.1007/s10886-005-9012-9
López P, Gabirot M, Martín J (2009) Immune activation affects chemical sexual ornaments of male Iberian wall lizards. Naturwissenschaften 96:65–69. https://doi.org/10.1007/s00114-008-0451-3
MacGregor HE, Lewandowsky RA, d’Ettorre P, Leroy C, Davies NW, While GM, Uller T (2017) Chemical communication, sexual selection, and introgression in wall lizards. Evolution 71:2327–2343. https://doi.org/10.1111/evo.13317
Martín J, López P (2000) Chemoreception, symmetry and mate choice in lizards. Proc R Soc Lond B 267:1265–1269. https://doi.org/10.1098/rspb.2000.1137
Martín J, López P (2006) Links between male quality, male chemical signals, and female mate choice in Iberian rock lizards. Funct Ecol 20:1087–1096. https://doi.org/10.1111/j.1365-2435.2006.01183.x
Martín J, López P (2007) Scent may signal fighting ability in male Iberian rock lizards. Biol Lett 3:125–127. https://doi.org/10.1098/rsbl.2006.0589
Martín J, López P (2011) Pheromones and reproduction in reptiles. In: Norris DO, López KH (eds) Hormones and reproduction of vertebrates, vol. 3. Reptiles. Academic Press, San Diego, pp 141–167
Martín J, López P (2013) Effects of global warming on sensory ecology of rock lizards: increased temperatures alter the efficacy of sexual chemical signals. Funct Ecol 27:1332–1340. https://doi.org/10.1111/1365-2435.12128
Martín J, López P (2014) Pheromones and chemical communication in lizards. In: Rheubert JL, Siegel DS, Trauth SE (eds) The reproductive biology and phylogeny of lizards and tuatara. CRC Press, Boca Raton, pp 43–77
Martín J, López P (2015) Condition-dependent chemosignals in reproductive behavior of lizards. Horm Behav 68:14–24. https://doi.org/10.1016/j.yhbeh.2014.06.009
Martín J, Civantos E, Amo L, López P (2007) Chemical ornaments of male lizards Psammodromus algirus may reveal their parasite load and health state to females. Behav Ecol Sociobiol 62:173–179. https://doi.org/10.1007/s00265-007-0451-x
Martín J, Chamut S, Manes ME, López P (2011) Chemical constituents of the femoral gland secretions of male tegu lizards (Tupinambis merianae) (fam.Teiidae). Z Naturforsch C 66:434–440. https://doi.org/10.1515/znc-2011-7-817
Martín J, Ortega J, López P (2013) Lipophilic compounds in femoral secretions of male collared lizards, Crotaphytus bicinctores (Iguania, Crotaphytidae). Biochem Syst Ecol 47:5–10. https://doi.org/10.1016/j.bse.2012.09.025
Martín J, Ortega J, López P (2015) Interpopulational variations in sexual chemical signals of Iberian wall lizards may allow maximizing signal efficiency under different climatic conditions. PLoS ONE 10:e0131492. https://doi.org/10.1371/journal.pone.0131492
Martín J, López P, Iraeta P, Díaz JA, Salvador A (2016) Differences in males’ chemical signals between genetic lineages of the lizard Psammodromus algirus promote male intrasexual recognition and aggression but not female mate preferences. Behav Ecol Sociobiol 70:1657–1668. https://doi.org/10.1007/s00265-016-2171-6
Mason RT, Parker MR (2010) Social behavior and pheromonal communication in reptiles. J Comp Physiol A 196:729–749. https://doi.org/10.1007/s00359-010-0551-3
Mayr E (1963) Animal species and evolution. Harvard University Press, Cambridge
McArdle BH, Anderson MJ (2001) Fitting multivariate models to community data: a comment on distance-based redundancy analysis. Ecology 82:290–297. https://doi.org/10.1890/0012-9658(2001)082[0290:FMMTCD]2.0.CO;2
Müller-Schwarze D (2006) Chemical Ecology of Vertebrates. Cambridge University Press, Cambridge
Navarro-García JC, García A, Méndez de la Cruz FR (2008) Estacionalidad, eficiencia termorreguladora de Aspidoscelis lineatissima (sauria: teiidae) y la calidad térmica del bosque tropical caducifolio en Chamela, Jalisco, México. Rev Mex Biodivers 79:413–419
Nosil P, Crespi BJ, Gries R, Gries G (2007) Natural selection and divergence in mate preference during speciation. Genetica 129:309–327. https://doi.org/10.1007/s10709-006-0013-6
Olsson M, Madsen T, Nordby J, Wapstra E, Ujvari B, Wittsell H (2003) Major histocompatibility complex and mate choice in sand lizards. Proc R Soc Lond B 270:S254–S256. https://doi.org/10.1098/rsbl.2003.0079
Panhuis TM, Butlin R, Zuk M, Tregenza T (2001) Sexual selection and speciation. Trends Ecol Evol 16:364–371. https://doi.org/10.1016/S0169-5347(01)02160-7
Pinheiro J, Bates D (2006) Mixed-effects models in S and S-PLUS. Berlin, Springer Science and Business Media
Punzo F (2007) Chemosensory cues associated with snake predators affect locomotor activity and tongue flick rate in the whiptail lizard, Aspidoscelis dixoni Scudday 1973 (Squamata Teiidae). Ethol Ecol Evol 19:225–235. https://doi.org/10.1080/08927014.2007.9522564
Punzo F (2008) Chemosensory recognition of the marbled whiptail lizard, Aspidoscelis marmorata (Squamata: Teiidae) to odors of sympatric lizards (Crotaphytus collaris, Coleonyx brevis, Eumeces obsoletus and Uta stansburiana) that represent different predation risks. J Environ Biol 29:57–61
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org. Accessed 26 Feb 2020
Ramírez-Bautista A, Balderas-Valdivia C, Vitt LJ (2000) Reproductive ecology of the whiptail lizard Cnemidophorus lineatissimus (squamata: teiidae) in a tropical dry forest. Copeia 2000:712–722. https://doi.org/10.1643/00458511(2000)000[0712:REOTWL]2.0.CO;2
Ribeiro LB, Gogliath M, Sales RFDD, Freire EMX (2011) Mating behavior and female accompaniment in the whiptail lizard Cnemidophorus ocellifer (Squamata, Teiidae) in the Caatinga region of northeastern Brazil. Biota Neotrop 11:363–368. https://doi.org/10.1590/S1676-06032011000400031
Ritchie MG (2007) Sexual selection and speciation. Ann Rev Ecol Evol S 38:79–102. https://doi.org/10.1146/annurev.ecolsys.38.091206.095733
Runemark A, Gabirot M, Svensson EI (2011) Population divergence in chemical signals and the potential for premating isolation between islet- and mainland populations of the Skyros wall lizard (Podarcis gaigeae). J Evol Biol 24:795–809. https://doi.org/10.1111/j.1420-9101.2010.02214.x
Safran RJ, Scordato ES, Symes LB, Rodríguez RL, Mendelson TC (2013) Contributions of natural and sexual selection to the evolution of premating reproductive isolation: a research agenda. Trends Ecol Evol 28:643–650. https://doi.org/10.1016/j.tree.2013.08.004
Schaefer HM, Ruxton GD (2015) Signal diversity, sexual selection, and speciation. Annu Rev Ecol Evol S 46:573–592. https://doi.org/10.1146/annurev-ecolsys-112414-054158
Schluter D (2009) Evidence for ecological speciation and its alternative. Science 323:737–741. https://doi.org/10.1126/science.1160006
Shine R, Reed RN, Shetty S, Lemaster M, Mason RT (2002) Reproductive isolating mechanisms between two sympatric sibling species of sea snakes. Evolution 56:1655–1662. https://doi.org/10.1111/j.0014-3820.2002.tb01477.x
Smadja C, Butlin RK (2009) On the scent of speciation: the chemosensory system and its role in premating isolation. Heredity 102:77–97. https://doi.org/10.1038/hdy.2008.55
Sobel JM, Chen GF, Watt LR, Schemske DW (2010) The biology of speciation. Evolution 64:295–315. https://doi.org/10.1111/j.1558-5646.2009.00877.x
Symonds MRE, Elgar MA (2008) The evolution of pheromone diversity. Trends Ecol Evol 23:220–228. https://doi.org/10.1016/j.tree.2007.11.009
Tobias JA, Aben J, Brumfield RT, Derryberry EP, Halfwerk W, Slabbekoorn H, Seddon N (2010) Song divergence by sensory drive in Amazonian birds. Evolution 64:2820–2839. https://doi.org/10.1111/j.1558-5646.2010.01067.x
Tu W (2014) Zero-inflated data. In: ElShaarawi AH, Piegorsch WW (eds) Encyclopedia of environmetric, vol 4. John Wiley and Sons, Chichester, pp 2387–2391. https://doi.org/10.1002/9781118445112.stat07451
Walker JM (1970) Morphological variation and clutch size in a population of Cnemidophorus lineatissimus Cope in Michoacán, México. Herpetologica 26:359–365. https://doi.org/10.2307/3891267
Wang IJ, Bradburd GS (2014) Isolation by environment. Mol Ecol 23:5649–5662. https://doi.org/10.1111/mec.12938
Weldon PJ, Flachsbarth B, Schulz S (2008) Natural products from the integument of nonavian reptiles. Nat Prod Rep 25:738–756. https://doi.org/10.1039/b509854h
Wilkins MR, Seddon N, Safran RJ (2013) Evolutionary divergence in acoustic signals: causes and consequences. Trends Ecol Evol 28:156–166. https://doi.org/10.1016/j.tree.2012.10.002
Wyatt TD (2014) Pheromones and animal behaviour: chemical signals and signatures. Cambridge University Press, Cambridge
Zozaya SM, Higgie M, Moritz C, Hoskin CJ (2019) Are pheromones key to unlocking cryptic lizard diversity? Am Nat 194:168–182. https://doi.org/10.1086/704059
Acknowledgments
We thank Secretaría de Medio Ambiente y Recursos Naturales for issuing the collecting permit number SGPA/DGVS/03701/17. We are grateful to Isaac Arteaga, Jeronimo Hernandez, Roberto Colín, Ayult ecotourism center, and the indigenous community of Maruata for assistance and services provided during our study. We are also grateful to the anonymous reviewers for their suggestions and considerations that markedly improved this document.
Funding
This study was funded by the project “Sucesión en bosques tropicales: recuperando biodiversidad, servicios y funciones del ecosistema” (CONACyT 2015-255544). A postgraduate scholarship was granted by the Programa Institucional de Doctorado en Ciencias Biológicas of the Universidad Michoacana de San Nicolás de Hidalgo and the Consejo Nacional de Ciencia y Tecnología (CONACYT) to ERG (CVU #589601) Ph.D. thesis of the principal author, under the direction of ISO.
Author information
Authors and Affiliations
Contributions
Conceptualization: ERG, ISO; Methodology: ERG, JM, JCG; Formal analysis and investigation: ERG, JCG, JM; Writing—original draft preparation: ERG; Writing—review and editing: ISO, JAD, EMR, JM, JCG; Funding acquisition: ISO; Resources: ISO, JCG; Supervision: ISO, JAD, EMR.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was carried out in strict accordance with the guidelines for use of live amphibians and reptiles in field research compiled by the American Society of Ichthyologists and Herpetologists (ASIH). In the research protocol, adult lizards were visually detected and captured without any harm. During the experimental trials, all experimental organisms were maintained under optimal conditions with access to water and food ad libitum. At the end of the experiments, these individuals were returned in good health to the original capture sites. All procedures with animals were carried out in accordance with the standards of bioethics and biosafety of the Universidad Michoacana de San Nicolás de Hidalgo.
Additional information
Communicated by T. Madsen
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raya-García, E., Suazo-Ortuño, I., Campos-García, J. et al. Chemical signal divergence among populations influences behavioral discrimination in the whiptail lizard Aspidoscelis lineattissimus (squamata: teiidae). Behav Ecol Sociobiol 74, 144 (2020). https://doi.org/10.1007/s00265-020-02931-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-020-02931-z