Abstract
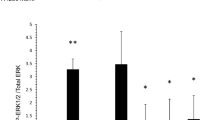
Activated mast cells are often found in the tumor microenvironment. They have both pro- and anti-tumorigenic roles, depending on the tumor type. Several lines of evidence suggest that the tumor microenvironment contains multiple soluble factors that can drive mast cell recruitment and activation. However, it is not yet clear how mast cells are activated by tumor cells. In this study, we explored whether tumor-derived microvesicles (TMV) from non-small cell lung cancer (NSCLC) cells interact with human mast cells, activate them to release cytokines, and affect their migratory ability. PKH67-labelled TMV isolated from NSCLC cell lines were found to be internalized by mast cells. This internalization was first noticed after 4 h and peaked within 24 h of co-incubation. Furthermore, internalization of TMV derived from NSCLC cell lines or from surgical lung tissue specimens resulted in ERK phosphorylation, enhanced mast cell migratory ability and increased release of cytokines and chemokines, such as TNF-α and MCP-1. Our data are thus, consistent with the conclusion that TMV have the potential to influence mast cell activity and thereby, affect tumorigenesis.




Similar content being viewed by others
Abbreviations
- ATCC:
-
American type culture collection
- CCL2:
-
Chemokine (C–C motif) ligand 2
- ERK:
-
Extracellular signal-regulated kinase
- EVs:
-
Extracellular vesicles
- MC:
-
Mast cells
- MCP-1:
-
Monocyte chemoattractant protein 1
- MV:
-
Microvesicles
- mvT*:
-
Microvesicles derived from activated T cells
- NSCLC:
-
Non-small cell lung cancer
- TMV:
-
Tumor-derived microvesicles
References
Kalesnikoff J, Galli SJ (2008) New developments in mast cell biology. Nat Immunol 9(11):1215–1223. https://doi.org/10.1038/ni.f.216
Shefler I, Salamon P, Reshef T, Mor A, Mekori YA (2010) T cell-induced mast cell activation: a role for microparticles released from activated T cells. J Immunol (Baltimore, MD: 1950) 185(7):4206–4212. https://doi.org/10.4049/jimmunol.1000409
Varricchi G, Galdiero MR, Loffredo S, Marone G, Iannone R, Marone G, Granata F (2017) Are mast cells MASTers in cancer? Front Immunol 8:424. https://doi.org/10.3389/fimmu.2017.00424
Dyduch G, Kaczmarczyk K, Okon K (2012) Mast cells and cancer: enemies or allies? Pol J Pathol 63(1):1–7
Attarha S, Roy A, Westermark B, Tchougounova E (2017) Mast cells modulate proliferation, migration and stemness of glioma cells through downregulation of GSK3beta expression and inhibition of STAT3 activation. Cell Signal 37:81–92. https://doi.org/10.1016/j.cellsig.2017.06.004
Sullivan R, Maresh G, Zhang X, Salomon C, Hooper J, Margolin D, Li L (2017) The emerging roles of extracellular vesicles as communication vehicles within the tumor microenvironment and beyond. Front Endocrinol 8:194. https://doi.org/10.3389/fendo.2017.00194
Iero M, Valenti R, Huber V, Filipazzi P, Parmiani G, Fais S, Rivoltini L (2008) Tumour-released exosomes and their implications in cancer immunity. Cell Death Differ 15(1):80–88. https://doi.org/10.1038/sj.cdd.4402237
Zhang P, Yeo JC, Lim CT (2019) Advances in technologies for purification and enrichment of extracellular vesicles. SLAS Technol 24(5):477–488. https://doi.org/10.1177/2472630319846877
Tricarico C, Clancy J, D'Souza-Schorey C (2017) Biology and biogenesis of shed microvesicles. Small GTPases 8(4):220–232. https://doi.org/10.1080/21541248.2016.1215283
Bian X, Xiao YT, Wu T, Yao M, Du L, Ren S, Wang J (2019) Microvesicles and chemokines in tumor microenvironment: mediators of intercellular communications in tumor progression. Mol Cancer 18(1):50. https://doi.org/10.1186/s12943-019-0973-7
Kogure A, Kosaka N, Ochiya T (2019) Cross-talk between cancer cells and their neighbors via miRNA in extracellular vesicles: an emerging player in cancer metastasis. J Biomed Sci 26(1):7. https://doi.org/10.1186/s12929-019-0500-6
Maacha S, Bhat AA, Jimenez L, Raza A, Haris M, Uddin S, Grivel JC (2019) Extracellular vesicles-mediated intercellular communication: roles in the tumor microenvironment and anti-cancer drug resistance. Mol Cancer 18(1):55. https://doi.org/10.1186/s12943-019-0965-7
O'Driscoll L (2015) Expanding on exosomes and ectosomes in cancer. N Engl J Med 372(24):2359–2362. https://doi.org/10.1056/NEJMcibr1503100
Wysoczynski M, Ratajczak MZ (2009) Lung cancer secreted microvesicles: underappreciated modulators of microenvironment in expanding tumors. Int J Cancer 125(7):1595–1603. https://doi.org/10.1002/ijc.24479
Shikotra A, Ohri CM, Green RH, Waller DA, Bradding P (2016) Mast cell phenotype, TNF alpha expression and degranulation status in non-small cell lung cancer. Sci Rep 6:38352. https://doi.org/10.1038/srep38352
Shefler I, Pasmanik-Chor M, Kidron D, Mekori YA, Hershko AY (2014) T cell-derived microvesicles induce mast cell production of IL-24: relevance to inflammatory skin diseases. J Allergy Clin Immunol 133(1):217–224.e211–213. https://doi.org/10.1016/j.jaci.2013.04.035
Baram D, Vaday GG, Salamon P, Drucker I, Hershkoviz R, Mekori YA (2001) Human mast cells release metalloproteinase-9 on contact with activated T cells: juxtacrine regulation by TNF-alpha. J Immunol (Baltimore, MD: 1950) 167(7):4008–4016
Mor A, Shefler I, Salamon P, Kloog Y, Mekori YA (2010) Characterization of ERK activation in human mast cells stimulated by contact with T cells. Inflammation 33(2):119–125. https://doi.org/10.1007/s10753-009-9165-8
Wang X, Lin Y (2008) Tumor necrosis factor and cancer, buddies or foes? Acta Pharmacol Sin 29(11):1275–1288. https://doi.org/10.1111/j.1745-7254.2008.00889.x
Yoshimura T (2018) The chemokine MCP-1 (CCL2) in the host interaction with cancer: a foe or ally? Cell Mol Immunol 15(4):335–345. https://doi.org/10.1038/cmi.2017.135
Misiak-Tloczek A, Brzezinska-Blaszczyk E (2009) IL-6, but not IL-4, stimulates chemokinesis and TNF stimulates chemotaxis of tissue mast cells: involvement of both mitogen-activated protein kinases and phosphatidylinositol 3-kinase signalling pathways. APMIS Acta Pathol Microbiol Immunol Scand 117(8):558–567. https://doi.org/10.1111/j.1600-0463.2009.02518.x
Derakhshani A, Vahidian F, Alihasanzadeh M, Mokhtarzadeh A, Lotfi Nezhad P, Baradaran B (2019) Mast cells: a double-edged sword in cancer. Immunol Lett 209:28–35. https://doi.org/10.1016/j.imlet.2019.03.011
Khazaie K, Blatner NR, Khan MW, Gounari F, Gounaris E, Dennis K, Bonertz A, Tsai FN, Strouch MJ, Cheon E, Phillips JD, Beckhove P, Bentrem DJ (2011) The significant role of mast cells in cancer. Cancer Metastasis Rev 30(1):45–60. https://doi.org/10.1007/s10555-011-9286-z
Stoyanov E, Uddin M, Mankuta D, Dubinett SM, Levi-Schaffer F (2012) Mast cells and histamine enhance the proliferation of non-small cell lung cancer cells. Lung Cancer (Amsterdam, Netherlands) 75(1):38–44. https://doi.org/10.1016/j.lungcan.2011.05.029
Baj-Krzyworzeka M, Szatanek R, Weglarczyk K, Baran J, Urbanowicz B, Branski P, Ratajczak MZ, Zembala M (2006) Tumour-derived microvesicles carry several surface determinants and mRNA of tumour cells and transfer some of these determinants to monocytes. Cancer Immunol Immunother CII 55(7):808–818. https://doi.org/10.1007/s00262-005-0075-9
Gorzalczany Y, Akiva E, Klein O, Merimsky O, Sagi-Eisenberg R (2017) Mast cells are directly activated by contact with cancer cells by a mechanism involving autocrine formation of adenosine and autocrine/paracrine signaling of the adenosine A3 receptor. Cancer Lett 397:23–32. https://doi.org/10.1016/j.canlet.2017.03.026
Zhang H, Yu Y, Zhou L, Ma J, Tang K, Xu P, Ji T, Liang X, Lv J, Dong W, Zhang T, Chen D, Xie J, Liu Y, Huang B (2018) Circulating tumor microparticles promote lung metastasis by reprogramming inflammatory and mechanical niches via a macrophage-dependent pathway. Cancer Immunol Res 6(9):1046–1056. https://doi.org/10.1158/2326-6066.cir-17-0574
Sung BH, Weaver AM (2017) Exosome secretion promotes chemotaxis of cancer cells. Cell Adhes Migrat 11(2):187–195. https://doi.org/10.1080/19336918.2016.1273307
Sung BH, Weaver AM (2018) Directed migration: cells navigate by extracellular vesicles. J Cell Biol 217(8):2613–2614. https://doi.org/10.1083/jcb.201806018
Oskeritzian CA, Alvarez SE, Hait NC, Price MM, Milstien S, Spiegel S (2008) Distinct roles of sphingosine kinases 1 and 2 in human mast-cell functions. Blood 111(8):4193–4200. https://doi.org/10.1182/blood-2007-09-115451
Kirshenbaum AS, Akin C, Wu Y, Rottem M, Goff JP, Beaven MA, Rao VK, Metcalfe DD (2003) Characterization of novel stem cell factor responsive human mast cell lines LAD 1 and 2 established from a patient with mast cell sarcoma/leukemia; activation following aggregation of FcepsilonRI or FcgammaRI. Leuk Res 27(8):677–682
Acknowledgements
Faye Schreiber, MSc edited the manuscript. She is an employee of Meir Medical Center.
Funding
This work was supported in part by the Israel Cancer Association (#20190025).
Author information
Authors and Affiliations
Contributions
IS and YAM designed the study. IS and PS performed the experiments. The first draft of the manuscript was written by IS. All authors reviewed and commented on the different versions of the manuscript and approved its final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval and ethical standards
The study was approved by the Meir Medical Center Institutional Review Board (Helsinki Committee; #0059-14-MMC, July 2015) and complies with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Written informed consent was obtained from all individual participants included in the study. Patients who underwent surgery for newly diagnosed lung cancer agreed, prior to their surgery, to the use of their residual specimens for medical research and publication.
Animal source
This article does not contain any studies with animals performed by any of the authors.
Cell line authentication
For lines A549, H1299 and H1975, cell line authentication was performed at the Genomics Center of Biomedical Core Facility, Technion, Haifa, Israel. The test was performed using the Promega GenePrint 24 System to determine short tandem repeat (STR) profile of 23 loci plus Amelogenin for sex determination (X or XY). In addition, the male-specific DYS391 locus is included to identify null Y allele results for Amelogenin. The results were analyzed using the 3500xl Genetic Analyzer (Life Echnologies NY, USA) and GeneMapper IDX software. For line H1299, the sample profile matches 8 of the 9 available loci of the STR profile from ATCC (American Type Culture Collection). In light of these results, it seems that the tested cell line is indeed NCI-H1299. For line H1975, the sample profile matches 8 of the 9 available loci of the STR profile from ATCC. Locus D13S317 presents a loss of heterozygosity (LOH). In light of these results, it seems that the tested cell line is indeed NCI-H1975. For line A549, the sample profile matches 9 of the 9 available loci of the STR profile from ATCC for line A549 (CCL-185). In light of these results, it seems that the tested cell line is indeed A549. The cell lines H1299, H1975 and A549 were kindly provided from the Lung Cancer Research Laboratory, Meir Medical Center, Kfar Saba, Israel. The LAD2 cell line was established from bone marrow aspirates from a patient with mast cell sarcoma/leukemia. Thus, no STR profile was available for authentication analysis. All experiments were conducted on cell lines at the same passages. The human LAD2 MC were kindly provided by Dr. A.S. Kirshenbaum [31].
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salamon, P., Mekori, Y.A. & Shefler, I. Lung cancer-derived extracellular vesicles: a possible mediator of mast cell activation in the tumor microenvironment. Cancer Immunol Immunother 69, 373–381 (2020). https://doi.org/10.1007/s00262-019-02459-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02459-w