Abstract
Purpose
To build and validate a magnetic resonance imaging-based radiomics model to preoperatively evaluate tumor budding (TB) in locally advanced rectal cancer (LARC).
Methods
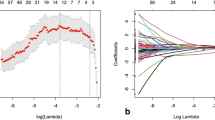
Pathologically confirmed LARC cases submitted to preoperative rectal MRI in two distinct hospitals were enrolled in this retrospective study and assigned to cohort 1 (training set, n = 77; test set, n = 51) and cohort 2 (validation set, n = 96). Radiomics features were obtained from multiple sequences, comprising high-resolution T2, contrast-enhanced T1, and diffusion-weighted imaging (T2WI, CE-T1WI, and DWI, respectively). The least absolute shrinkage and selection operator (LASSO) was utilized to select the optimal features from T2WI, CE-T1WI, DWI, and the combination of multi-sequences, respectively. A support vector machine (SVM) classifier was utilized to construct various radiomics models for discriminating the TB grades. Receiver operating characteristic curve analysis and decision curve analysis (DCA) were carried out to determine the diagnostic value.
Results
Five optimal features associated with TB grade were determined from combined multi-sequence data. Accordingly, a radiomics model based on combined multi-sequences had an area under the curve of 0.796, with an accuracy of 81.2% in the validation set, showing a better performance in comparison with other models in both cohorts (p < 0.05). DCA exhibited a clinical benefit for this radiomics model.
Conclusion
The novel MRI-based radiomics model combining multiple sequences is an effective and non-invasive approach for evaluating TB grade preoperatively in patients with LARC.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
[1] Nagtegaal ID, Odze RD, Klimstra D et al. (2020) The 2019 WHO classification of tumours of the digestive system. Histopathology 76:182-188. https://doi.org/10.1111/his.13975
[2] Oronsky B, Reid T, Larson C, Knox SJ (2020) Locally advanced rectal cancer: The past, present, and future. Semin Oncol 47:85-92. https://doi.org/10.1053/j.seminoncol.2020.02.001
[3] Benson AB, Venook AP, Al-Hawary MM et al. (2018) Rectal Cancer, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 16:874-901. https://doi.org/10.6004/jnccn.2018.0061
[4] Dawson H, Galuppini F, Träger P et al. (2019) Validation of the International Tumor Budding Consensus Conference 2016 recommendations on tumor budding in stage I-IV colorectal cancer. Hum Pathol 85:145-151. https://doi.org/10.1016/j.humpath.2018.10.023
[5] Rogers AC, Winter DC, Heeney A et al. (2016) Systematic review and meta-analysis of the impact of tumour budding in colorectal cancer. Br J Cancer 115:831-840. https://doi.org/10.1038/bjc.2016.274
[6] Ueno H, Hase K, Hashiguchi Y et al. (2014) Novel risk factors for lymph node metastasis in early invasive colorectal cancer: a multi-institution pathology review. J Gastroenterol 49:1314-1323. https://doi.org/10.1007/s00535-013-0881-3
[7] Petrelli F, Pezzica E, Cabiddu M et al. (2015) Tumour Budding and Survival in Stage II Colorectal Cancer: a Systematic Review and Pooled Analysis. J Gastrointest Cancer 46:212-218. https://doi.org/10.1007/s12029-015-9716-1
[8] Nakamura T, Mitomi H, Kanazawa H, Ohkura Y, Watanabe M (2008) Tumor budding as an index to identify high-risk patients with stage II colon cancer. Dis Colon Rectum 51:568-572. https://doi.org/10.1007/s10350-008-9192-9
[9] van Wyk HC, Roseweir A, Alexander P et al. (2019) The Relationship between tumor budding, tumor microenvironment, and survival in patients with primary operable colorectal cancer. Ann Surg Oncol 26:4397-4404. https://doi.org/10.1245/s10434-019-07931-6
[10] Lino-Silva LS, Salcedo-Hernández RA, Gamboa-Domínguez A (2018) Tumour budding in rectal cancer. A comprehensive review. Contemp Oncol (Pozn) 22:61-74. https://doi.org/10.5114/wo.2018.77043
[11] Lambin P, Rios-Velazquez E, Leijenaar R et al. (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441-446. https://doi.org/10.1016/j.ejca.2011.11.036
[12] Kumar V, Gu Y, Basu S et al. (2012) Radiomics: the process and the challenges. Magn Reson Imaging 30:1234-1248. https://doi.org/10.1016/j.mri.2012.06.010
[13] Aerts HJ, Velazquez ER, Leijenaar RT et al. (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:4006. https://doi.org/10.1038/ncomms5006
[14] Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: Images Are More than Pictures, They Are Data. Radiology 278:563-577. https://doi.org/10.1148/radiol.2015151169
[15] Chen F, Ma X, Li S et al. (2020) MRI-Based Radiomics of Rectal Cancer: Assessment of the Local Recurrence at the Site of Anastomosis. Acad Radiol https://doi.org/10.1016/j.acra.2020.09.024
[16] Ma X, Shen F, Jia Y, Xia Y, Li Q, Lu J (2019) MRI-based radiomics of rectal cancer: preoperative assessment of the pathological features. BMC Med Imaging 19:86. https://doi.org/10.1186/s12880-019-0392-7
[17] Liu M, Ma X, Shen F, Xia Y, Jia Y, Lu J (2020) MRI-based radiomics nomogram to predict synchronous liver metastasis in primary rectal cancer patients. Cancer Med 9:5155-5163. https://doi.org/10.1002/cam4.3185
[18] Zhou X, Yi Y, Liu Z et al. (2020) Radiomics-Based Preoperative Prediction of Lymph Node Status Following Neoadjuvant Therapy in Locally Advanced Rectal Cancer. Front Oncol 10:604. https://doi.org/10.3389/fonc.2020.00604
[19] Yu X, Song W, Guo D et al. (2020) Preoperative Prediction of Extramural Venous Invasion in Rectal Cancer: Comparison of the Diagnostic Efficacy of Radiomics Models and Quantitative Dynamic Contrast-Enhanced Magnetic Resonance Imaging. Front Oncol 10:459. https://doi.org/10.3389/fonc.2020.00459
[20] Zhang Y, He K, Guo Y et al. (2020) A Novel Multimodal Radiomics Model for Preoperative Prediction of Lymphovascular Invasion in Rectal Cancer. Front Oncol 10:457. https://doi.org/10.3389/fonc.2020.00457
[21] Park H, Kim KA, Jung JH, Rhie J, Choi SY (2020) MRI features and texture analysis for the early prediction of therapeutic response to neoadjuvant chemoradiotherapy and tumor recurrence of locally advanced rectal cancer. Eur Radiol 30:4201-4211. https://doi.org/10.1007/s00330-020-06835-4
[22] Liu Z, Zhang XY, Shi YJ et al. (2017) Radiomics Analysis for Evaluation of Pathological Complete Response to Neoadjuvant Chemoradiotherapy in Locally Advanced Rectal Cancer. Clin Cancer Res 23:7253-7262. https://doi.org/10.1158/1078-0432.ccr-17-1038
[23] Nie K, Shi L, Chen Q et al. (2016) Rectal Cancer: Assessment of Neoadjuvant Chemoradiation Outcome based on Radiomics of Multiparametric MRI. Clin Cancer Res 22:5256-5264. https://doi.org/10.1158/1078-0432.ccr-15-2997
[24] Cui Y, Yang X, Shi Z et al. (2019) Radiomics analysis of multiparametric MRI for prediction of pathological complete response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Eur Radiol 29:1211-1220. https://doi.org/10.1007/s00330-018-5683-9
[25] Zhang XY, Wang L, Zhu HT et al. (2020) Predicting rectal cancer response to neoadjuvant chemoradiotherapy using deep learning of diffusion kurtosis MRI. Radiology 296:56-64. https://doi.org/10.1148/radiol.2020190936
[26] Li Y, Liu W, Pei Q et al. (2019) Predicting pathological complete response by comparing MRI-based radiomics pre- and postneoadjuvant radiotherapy for locally advanced rectal cancer. Cancer Med 8:7244-7252. https://doi.org/10.1002/cam4.2636
[27] Amin MB, Greene FL, Edge SB et al. (2017) The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more "personalized" approach to cancer staging. CA Cancer J Clin 67:93-99. https://doi.org/10.3322/caac.21388
[28] Lugli A, Kirsch R, Ajioka Y et al. (2017) Recommendations for reporting tumor budding in colorectal cancer based on the International Tumor Budding Consensus Conference (ITBCC) 2016. Mod Pathol 30:1299-1311. https://doi.org/10.1038/modpathol.2017.46
[29] Choi HJ, Park KJ, Shin JS, Roh MS, Kwon HC, Lee HS (2007) Tumor budding as a prognostic marker in stage-III rectal carcinoma. Int J Colorectal Dis 22:863-868. https://doi.org/10.1007/s00384-006-0249-8
[30] Guzińska-Ustymowicz K (2005) The role of tumour budding at the front of invasion and recurrence of rectal carcinoma. Anticancer Res 25:1269-1272.
[31] Syk E, Lenander C, Nilsson PJ, Rubio CA, Glimelius B (2011) Tumour budding correlates with local recurrence of rectal cancer. Colorectal Dis 13:255-262. https://doi.org/10.1111/j.1463-1318.2009.02119.x
[32] Okuyama T, Nakamura T, Yamaguchi M (2003) Budding is useful to select high-risk patients in stage II well-differentiated or moderately differentiated colon adenocarcinoma. Dis Colon Rectum 46:1400-1406. https://doi.org/10.1007/s10350-004-6757-0
[33] Price WN, 2nd, Cohen IG (2019) Privacy in the age of medical big data. Nat Med 25:37-43. https://doi.org/10.1038/s41591-018-0272-7
[34] De Smedt L, Palmans S, Sagaert X (2016) Tumour budding in colorectal cancer: what do we know and what can we do? Virchows Arch 468:397-408. https://doi.org/10.1007/s00428-015-1886-5
[35] Koelzer VH, Zlobec I, Lugli A (2016) Tumor budding in colorectal cancer--ready for diagnostic practice? Hum Pathol 47:4-19. https://doi.org/10.1016/j.humpath.2015.08.007
[36] Koelzer VH, Zlobec I, Berger MD et al. (2015) Tumor budding in colorectal cancer revisited: results of a multicenter interobserver study. Virchows Arch 466:485-493. https://doi.org/10.1007/s00428-015-1740-9
Acknowledgements
We thank our funders for coming up with the main ideas and designing the studies.
Funding
The present study was supported by the Project of the Action Plan of Major Diseases Prevention and Treatment (2017ZX01001-S12) and the Special Project of Integrated Traditional Chinese and Western Medicine in General Hospitals of Shanghai (ZHYY-ZXYJHZX-201901). They provide the main ideas and design the studies.
Author information
Authors and Affiliations
Contributions
ZL performed segmentation and is a major contributor in writing the manuscript. FC performed segmentation and analyzed and interpreted the patient data regarding radiomics features. SZ analyzed and interpreted the patient data regarding radiomics features. XM acquired the data. YX performed statistical analysis. FS conceived the project. YL conceived the project. CS conceived the project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The present study was approved by the Biomedicine Ethics Review Committees of Ruijin Hospital and Changhai Hospital.
Consent to participate
Informed consent was waived.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
261_2021_3311_MOESM1_ESM.docx
Supplementary file1—Supplemental Table 1. Detailed parameters applied for T2WI, T1WI, and DWI and used for radiomics model building. (DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Li, Z., Chen, F., Zhang, S. et al. The feasibility of MRI-based radiomics model in presurgical evaluation of tumor budding in locally advanced rectal cancer. Abdom Radiol 47, 56–65 (2022). https://doi.org/10.1007/s00261-021-03311-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03311-5