Abstract
Purpose
Second-generation tau radiotracers for use with positron emission tomography (PET) have been developed for visualization of tau deposits in vivo. For several β-amyloid and first-generation tau-PET radiotracers, it has been shown that early-phase images can be used as a surrogate of neuronal injury. Therefore, we investigated the performance of early acquisitions of the novel tau-PET radiotracer [18F]PI-2620 as a potential substitute for [18F]fluorodeoxyglucose ([18F]FDG).
Methods
Twenty-six subjects were referred with suspected tauopathies or overlapping parkinsonian syndromes (Alzheimer’s disease, progressive supranuclear palsy, corticobasal syndrome, multi-system atrophy, Parkinson’s disease, multi-system atrophy, Parkinson's disease, frontotemporal dementia) and received a dynamic [18F]PI-2620 tau-PET (0–60 min p.i.) and static [18F]FDG-PET (30–50 min p.i.). Regional standardized uptake value ratios of early-phase images (single frame SUVr) and the blood flow estimate (R1) of [18F]PI-2620-PET were correlated with corresponding quantification of [18F]FDG-PET (global mean/cerebellar normalization). Reduced tracer uptake in cortical target regions was also interpreted visually using 3-dimensional stereotactic surface projections by three more and three less experienced readers. Spearman rank correlation coefficients were calculated between early-phase [18F]PI-2620 tau-PET and [18F]FDG-PET images for all cortical regions and frequencies of disagreement between images were compared for both more and less experienced readers.
Results
Highest agreement with [18F]FDG-PET quantification was reached for [18F]PI-2620-PET acquisition from 0.5 to 2.5 min p.i. for global mean (lowest R = 0.69) and cerebellar scaling (lowest R = 0.63). Correlation coefficients (summed 0.5–2.5 min SUVr & R1) displayed strong agreement in all cortical target regions for global mean (RSUVr 0.76, RR1 = 0.77) and cerebellar normalization (RSUVr 0.68, RR1 = 0.68). Visual interpretation revealed high regional correlations between early-phase tau-PET and [18F]FDG-PET. There were no relevant differences between more and less experienced readers.
Conclusion
Early-phase imaging of [18F]PI-2620 can serve as a surrogate biomarker for neuronal injury. Dynamic imaging or a dual time-point protocol for tau-PET imaging could supersede additional [18F]FDG-PET imaging by indexing both the distribution of tau and the extent of neuronal injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tauopathies consist of neurodegenerative diseases including, among others, Alzheimer’s disease (AD), frontotemporal dementia and atypical parkinsonian syndromes such as progressive supranuclear palsy (PSP) and corticobasal syndrome (CBS), with the connective characteristic of misfolded and accumulated tau protein in different parts of the brain [1, 2]. Visualization of tau deposits in vivo has become possible with various tau-targeting ligands for use with positron emission tomography (PET) [3, 4]. While first-generation tau tracers suffered from off-target binding [5, 6] and a resulting large inter- and intra-case variability [7], second-generation tau-PET tracers showed promising first results in vivo with specific binding in affected regions in patients with mild cognitive impairment and AD compared to healthy controls [8,9,10,11].
Tau-PET imaging complements an important biomarker for the characterization of neurodegenerative diseases. For AD, it has been proposed to classify the disease according to the biomarkers for amyloid, tau and neuronal injury by the A/T/N scheme [12]. In this classification scheme, neuronal injury in the pathological definition can be determined in vivo by three different diagnostic approaches. Atrophy in structural magnetic resonance tomography and total tau in cerebrospinal fluid are considered as well as hypometabolism in 18F-fluorodeoxyglucose ([18F]FDG)-PET. This is underpinned by a combined study in prion disease, indicating that metabolic imaging via [18F]FDG correlates with neuropathologic changes including neuronal loss [13].
For first-generation tau tracers and several tracers for amyloid-PET imaging, it has been shown that the perfusion-phase images as obtained by these tracers is comparable to glucose metabolism as assessed by [18F]FDG-PET and can therefore be used as surrogate biomarker of neuronal injury [14,15,16,17,18,19,20,21]. With respect to cost and radiation exposure, such “one-stop-shop” protocols have the opportunity to examine two important biological markers with one procedure.
In contrast to amyloid imaging where the subjects can only be classified as amyloid-positive or amyloid-negative, tau-PET imaging shows characteristic patterns for several different neurodegenerative entities [3]. Therefore, tau-PET imaging could probably also be used to discriminate a range of tauopathies beyond AD. Due to the lack of specificity of first-generation tau tracers, it is of great interest whether and how the perfusion-phase images as obtained by second-generation tracers can be used as a marker of neuronal injury.
Thus, we aimed to investigate the potential of the second-generation tau-PET ligand [18F]PI-2620 [22] as an additional (in addition to detecting tau pathology) biomarker of neuronal injury. We validated early-phase [18F]PI-2620 data against [18F]FDG-PET and focused on optimizing dynamic or coffee break acquisition protocols for tau-PET imaging with dual biomarker information.
Materials and methods
Study design and patient enrolment
Patients with different suspected tauopathies were referred by dementia or movement disorder experts to [18F]PI-2620 tau-PET imaging. We selected all subjects with an additional [18F]FDG-PET acquired < 12 months before/after tau-PET imaging. The cohort consisted of patients with different suspected clinical diagnoses and represented a true clinical cross-section in a tertiary centre with a focus on dementia/ movement disorders. The most likely clinical differential diagnosis was recorded before [18F]PI-2620 tau-PET imaging. All patients provided informed written consent to PET imaging. The study was conducted in accordance with the principles of the Declaration of Helsinki, and approval for scientific data analysis was obtained from the local ethics committee (application number 17–569).
PET imaging
Radiosynthesis
Radiosynthesis of [18F]PI-2620 was achieved by nucleophilic substitution on a BOC-protected nitro precursor using an automated synthesis module (IBA, Synthera). The protecting group was cleaved under the radiolabelling conditions. The product was purified by semipreparative HPLC. Radiochemical purity was 99%. Non-decay corrected yields were about 35% with a molar activity of 8∙106 GBq/mmol at the end of synthesis. [18F]FDG was purchased commercially.
PET acquisition and preparation
All patients were scanned at the Department of Nuclear Medicine, LMU Munich, with a Biograph 64 or a Siemens mCT PET/CT scanner (both Siemens, Erlangen, Germany). A low-dose CT scan preceded the PET acquisition and served for attenuation correction. [18F]PI-2620-PET was performed in a full dynamic 0–60-min setting initiated upon intravenous injection (~ 10 s) of 185 ± 10 MBq of the ligand. [18F]PI-2620-PET data were reconstructed in a series of 23 frames (6 × 0.5 min, 4 × 1.0 min, 4 × 2.0 min, 9 × 5.0 min). [18F]-FDG-PET was acquired after injection of 125 ± 10 MBq [18F]FDG according to the EANM protocol [23]: fasting conditions > 6 h with a blood glucose < 120 mg/dl (6.7 mm) at time of scanning, silent room with dimmed light, headphones and blindfold 20 min prior and after injection. [18F]FDG-PET data was reconstructed in a static 30–50-min frame. PET data were reconstructed iteratively (4 iterations, 21 subsets, 5.0-mm Gauss/5 iterations, 24 subsets, 5.0-mm Gauss) with a matrix size of 336 × 336 × 109/ 400 × 400 × 148, a voxel size of 1.018 × 1.018 × 2.027/1.018 × 1.018 × 1.500 mm3 and a slice thickness of 2.027/1.500 mm. Standard corrections with regard to scatter, decay and random counts were used. Both reconstruction algorithms resulted in images with equal spatial resolution (8 × 8 × 7 mm) as validated by Hofmann phantom measures.
Image processing
All image data were processed and analysed using PMOD (version 3.5, PMOD Technologies Ltd., Zurich, Switzerland). For spatial normalization, tracer-specific templates in the MNI space were created for [18F]PI-2620 (30–60 min) and [18F]-FDG as described previously [14]. Dynamic [18F]PI-2620 images were coregistered to the MNI space by applying the 30–60-min transformation (brain normalization settings: nonlinear warping, 8-mm input smoothing, equal modality, 16 iterations, frequency cut-off 3, regularization 1.0, no thresholding). All images were analysed in MNI space. The regional cerebral blood flow estimate R1 was computed from the dynamic [18F]PI-2620 images by applying the simplified reference tissue model 2 (SRTM2) as described previously [24, 25], using the cerebellum (excluding the dentate nucleus and superior layers) as a reference region. A total number of ten predefined cortical volumes (bilateral frontal, central region, parietal, temporal, occipital) of interests (VOIs) deriving from the Hammers atlas [26] were delineated in the MNI space and standardized uptake value ratios (SUVr) of all VOIs were extracted for the different images (and different time frames) used for the analysis.
In preparation for visual analyses of all images (after selection of the appropriate time frame), dynamic data frames #2-#5 (0.5–2.5 min) were summed. To account for the lower count statistics of the early-phase images, an additional 6.0-mm Gaussian filter was applied for both ([18F]PI-26200.5–2.5min & [18F]PI-2620R1).
PET data evaluation
Correlation of single frames of [18F]PI-2620-PET versus [18F]FDG-PET
The optimal early time window for [18F]PI-2620 early-phase imaging in terms of maximal correlation to [18F]FDG-PET was determined. To this end, [18F]PI-2620-PET SUVrs for the ten cortical VOIs were extracted and correlated with the SUVrs of the corresponding [18F]FDG-PET data after normalization of uptake to global mean (GBM) or by use of a cerebellar reference region (CBL). Pearson’s correlation coefficients (R) were compared between different time frames and the selection of the optimal early-phase time window was based on the extent and significance of single frame agreement.
Regional comparison of optimized early-phase [18F]PI-26200.5–2.5min and [18F]PI-2620R1 versus [18F]FDG
The SUVr values of the optimized summed [18F]PI-2620 early-phase image (0.5–2.5 min), the [18F]PI-2620 R1 image and the [18F]FDG image were correlated for all ten cortical regions to investigate the regional relationship between tau-PET perfusion and glucose metabolism.
Visual analysis of stereotactic surface projections
For visual interpretation of early-phase [18F]PI-26200.5–2.5min, [18F]PI-2620R1 and [18F]FDG-PET images, three-dimensional stereotactic surface projections (3D-SSP) [27] were generated using the software Neurostat (Department of Radiology, University of Washington, Seattle, WA, USA). Voxel-wise Z-scores were calculated in Neurostat by comparing the individual tracer uptake ([18F]PI-26200.5–2.5min, [18F]PI-2620R1 and [18F]FDG) to a historical [18F]FDG-PET database (cognitively healthy individuals, age-matched, N = 18).
Three more experienced nuclear medicine physicians (H.B., T.v.E., M.B.) and three less experienced nuclear medicine interns (L.B., M.U., J.S.) visually assessed the 3D-SSP images using the Z-score maps (GBM scaling) and rated cortical regions used in the clinical routine (bilateral frontal, parietal, temporal, occipital cortex areas) from 0 to 3 (0 = no reduction, 1 = low reduction, 2 = intermediate reduction, 3 = severe reduction 0.5 steps were allowed). Furthermore, whole-brain 3D-SSP images were rated binary (0/1) and according to the severity of present neurodegeneration (0 = no/1 = mild/2 = intermediate/3 = severe neurodegeneration; 0.5 steps were allowed). A significant neuronal injury of the patient (A/T/N: N+) was defined by the majority read of binarized [18F]FDG-PET evaluation by the three more experienced physicians. Readers had access to the pre-PET clinical diagnosis. All 3D-SSP images ([18F]PI-26200.5–2.5min, [18F]PI-2620R1 and [18F]FDG) were randomly and blindly (with regard to the type of image) presented to the readers.
Statistical analysis
Correlations of regional SUVr between early-phase [18F]PI-2620 and [18F]FDG images were evaluated using Pearson’s correlation coefficient (R) and R values were compared using Fisher’s Z-transformation. Quantitative variables were reported as mean ± standard deviation. Comparison of R values for different normalization approaches was also performed by Fisher’s Z-transformation. For visual analysis and the specification of the most likely PET diagnosis, the intra-reader agreement between [18F]PI-26200.5–2.5min, [18F]PI-2620R1 and [18F]FDG was calculated using Spearman’s rank correlation coefficients. The disagreement between visual ratings of [18F]PI-26200.5–2.5min or [18F]PI-2620R1 and [18F]FDG was calculated and evaluated as frequency of all brain regions. A significance level of p < 0.05 was applied in all analyses. All statistical analyses were performed using SPSS (version 25.0, IBM, Armonk, New York, USA).
Results
Demographics
A total of 26 subjects (age = 66 ± 11 years, 17 female) were included in the analysis. The cohort consisted of seven subjects with a most likely diagnosis of AD, 13 subjects with movement disorders and most likely diagnosis of PSP or CBS, one case with most likely frontotemporal dementia, two cases with most likely multi-system atrophy, two cases with most likely Parkinson’s disease, and one case with cognitive impairment of unknown reason. The mean time interval between both PET investigations was 1.2 ± 1.7 months. For details of the study population see Table 1. Visual binary interpretation (majority read of more experienced readers) revealed a significant neuronal injury to [18F]FDG-PET in 65% of all cases.
Optimal time window for [18F]PI-2620-PET early-phase imaging
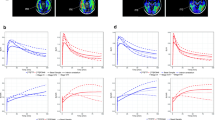
The VOI-based comparison of single frames of [18F]PI-2620-PET (6 × 0.5 min, 4 × 1.0 min, 4 × 2 min, 9 × 5.0 min) and [18F]FDG-PET (30–50 min p.i.) revealed the highest agreement for the frames #2–#5 lasting from 0.5–2.5 min after injection for both global mean scaling (RGBM = 0.728) and a cerebellar reference region (RCBL = 0.665; Fig. 1). The correlation was statistically significant for all cortical regions for frames #2–#7 for global mean scaling (0.5–4 min after injection), but frames #6–#7 showed overall lower correlation coefficients compared to frames #2–#5 (especially with a cerebellar reference region). Because of the fast wash-out and sufficient count statistics for frames #2–#5, the mean values of the 0.5–2.5 min p.i. time window (frames #2-#5) were used for further semiquantitative and visual analyses. This was as they represented the optimal trade-off between correlation with the [18F]FDG-PET data and count statistics. The correlation coefficients between the early [18F]PI-2620 and [18F]FDG-PET data and their degree of significance for all frames are shown in Table 2. The correlations coefficients between both normalization approaches were not significantly different (p = 0.301).
a Averaged (across all study subjects) time-activity curve of [18F]PI-2620 in a cortical composite volume of interest during the dynamic data acquisition. b Correlation coefficients (R) between single frames of [18F]PI-2620-PET and [18F]FDG-PET (30–50 min p.i.). GBM, global mean scaling; CBL, cerebellar reference region. The time-window used for further analyses is highlighted in grey
Semiquantitative VOI-based comparison of early-phase [18F]PI-2620-PET and [18F]FDG-PET
The PET parameters in all cortical brain regions showed highly significant correlations with [18F]FDG-PET for both early-phase [18F]PI-2620-PET approaches (0.5–2.5 min and R1). There was no significant difference in the correlation coefficients of [18F]PI-26200.5–2.5min and [18F]PI-2620R1 with [18F]FDG-PET (R0.5–2.5min = 0.762, RR1 = 0.766, p = 0.487) for global mean normalization. The highest degree of correlation was found in the right parietal cortex (R0.5–2.5min = 0.872, RR1 = 0.884) and the weakest correlation was observed in the left central region (R0.5–2.5min = 0.585, RR1 = 0.586). The semiquantitative comparison of [18F]PI-26200.5–2.5min and [18F]PI-2620R1 with [18F]FDG-PET and cerebellar scaling also showed significant, but overall weaker correlations when compared to global mean normalization for all cortical regions (R0.5–2.5min = 0.683, RR1 = 0.683, p = 0.472). Correlation plots for [18F]PI-26200.5–2.5min and [18F]PI-2620R1 with [18F]FDG-PET (all global mean normalization) are shown in Fig. 2. Corresponding regional values and correlation coefficients determined by comparing regional [18F]PI-26200.5–2.5min and [18F]PI-2620R1 with [18F]FDG-PET SUVr (global mean and cerebellar normalization) are shown in Supplementary Table 1.
Visual 3D-SSP comparison of early-phase [18F]PI-2620-PET and [18F]FDG-PET
After identification of the optimal time window, visual assessment was performed by evaluating 3D-SSP images of early-phase ([18F]PI-26200.5–2.5min and [18F]PI-2620R1) and [18F]FDG-PET (global mean normalization). Representative images of early-phase [18F]PI-26200.5–2.5min, [18F]PI-2620R1 and [18F]FDG-PET 3D-SSP and exemplary summed 0.5–2.5-min section images for three different most likely diagnoses of neurodegenerative disorders are shown in Fig. 3. In all cases, the regional pattern of hypoperfusion in [18F]PI-26200.5–2.5min and [18F]PI-2620R1 3D-SSP resembled the hypometabolism pattern in [18F]FDG-PET. Furthermore, exemplary distribution volume ratio images of those cases with the specific tau-PET binding pattern are shown in Supplementary Fig. 1.
Representative 3D-SSP images (Z-score maps) of [18F]FDG-PET, early-phase [18F]PI-26200.5–2.5min and [18F]PI-2620R1 for three different most likely diagnoses of neurodegenerative disorders: a Alzheimer’s disease, b frontotemporal dementia, c progressive supranuclear palsy (Richardson syndrome). Surface projections from R, right; L, left; LAT, lateral; SUP, superior; INF, inferior; ANT, anterior; POST, posterior; MED, medial
Visual interpretation revealed moderate to high regional correlations for the majority of regions between early-phase tau-PET (both PI-26200.5–2.5min and PI-2620R1) and [18F]FDG-PET (see Table 3).
The frequency of no or only minor disagreement was far higher (0–0.5; 73–83%) when compared to the frequency of moderate (1.0–1.5; 17–25%) or high disagreement (≥ 2.0; 1–2%) regardless of using PI-26200.5–2.5min or PI-2620R1 for early-phase assessment (Fig. 4). Frequencies of disagreement were similar for more and less experienced readers (compare Fig. 4a, b).
Disagreement between visual rating of early-phase a [18F]PI-26200.5–2.5min and b [18F]PI-2620R1 3D-SSP images in contrast to visual rating of [18F]FDG-PET 3D-SSP images for all evaluated brain regions of all cases (n = 624). Less experienced (LER) and more experienced (MER) readers are displayed separately
Discussion
Tau-PET imaging is of great interest as tauopathies make up the majority of neurodegenerative diseases [28]. However, not only information on tau-positivity but also on the presence of neuronal injury is considered in current diagnosis or research criteria of AD [12, 29] and non-AD tauopathies [30]. In accordance with previous amyloid-PET studies [14,15,16,17,18, 21], and considering the known limitations of first-generation tau radiotracers [19, 31], we aimed to evaluate the potential of the early-phase of the second-generation tau-PET radiotracer [18F]PI-2620 [11] as a potential surrogate biomarker of neuronal injury. Our results demonstrate a strong quantitative and visual agreement between reduced perfusion in tau-PET and reduced glucose metabolism in [18F]FDG-PET imaging. Therefore, early-phase tau-PET images may potentially eliminate the need for an additional [18F]FDG-PET. The ability to obtain two different biomarkers reduces costs, time and radiation exposure, and enables improved differential diagnosis through a one-stop-shop procedure, which is especially important for disabled patients.
The semiquantitative comparison of [18F]PI-2620-PET single frames with individual [18F]FDG-PET images revealed the highest agreement at 0.5–2.5 min after injection and decreased steadily until it was no longer significant for all cortical regions later than 4 min after injection. Previous studies evaluating the agreement between early-phase amyloid-PET acquisitions and [18F]FDG-PET suggested time frames up to 10 min after injection, often sparing the first minute after injection [14, 15, 17]. Compared to these studies, we can conclude that the acquisition of early-phase [18F]PI-2620 must include a very early and relatively short time frame to achieve a high agreement between tau-PET and [18F]FDG-PET, likely related to the fast washout of [18F]PI-2620 (Fig. 1a). Although the frames between 0.5 and 4.0 min p.i. indicated a significant correlation with [18F]FDG-PET, we recommend the use of 0.5 to 2.5 min p.i. for early-phase [18F]PI-2620 acquisition as this time window was more robust across different scaling methods. For implementation in study or clinical routine workflows, dynamic or coffee break protocols (included break between early- and late-image acquisitions with the possibility for the patient to rest) could be considered. In this regard, dynamic protocols will have the advantage of being able to perform non-invasive kinetic modelling for the tau-PET read-out, which will be important to account for blood flow changes in longitudinal studies [32]. We also propose to exclude the first half minute of acquisition, which showed more variability, likely related to variance in blood flow (i.e., reduced cardiac ejection fraction), site of injection, and variance of manual injection. Therefore, we additionally calculated the regional cerebral blood flow estimate R1 of [18F]PI-2620 to take such individual differences into account and found similar results by this approach. The VOI-based semiquantitative regional correlation of both the summed early-phase (0.5–2.5 min) and R1 of [18F]PI-2620 showed a highly significant correlation in all assessed cortical regions and no significant advantage for one of the methods. For clinical implementation, the reconstruction of a summed image seems to be more suitable as it can be easily added to conventional reconstruction algorithms of common PET systems.
All semiquantitative analyses were performed with global mean normalization and whole cerebellum scaling. In accordance with our previous investigation on early-phase amyloid-PET [14], the cerebellar normalization approach showed lower correlations for all investigated cortical regions. Since cerebellar perfusion tends to be highly variable, e.g., as a consequence of significant gender differences [33], a global mean normalization is likely superior to cerebellar scaling for interpretation of early-phase tau-PET images at the level of individual patients. Nevertheless, in subjects with an overall high load of neuronal injury or other reasons of a reduced whole brain perfusion (e.g., after ischemic stroke), the cerebellar scaling proved to be a viable alternative approach for normalization.
Based on the semiquantitative results, the visual analysis of summed [18F]PI-2620 perfusion-like images was also performed with 3DSSP images generated by global mean normalization. Both tau-PET early-phase images (summed 0.5–2.5 min and R1) showed an overall high visual agreement for the detection of neuronal injury with the corresponding metabolic imaging. Less and more experienced nuclear medicine physicians had no or only minor disagreement between early-phase [18F]PI-2620-PET and metabolic images in the majority of cases, indicating broad clinical applicability. Different patient preparation prior to injection of [18F]FDG (eye patch and noise cancelling according to the EANM protocol [23]) and [18F]PI-2620 (room noise, no eye patch) needs to be considered as a limitation.
In our population of subjects referred with suspected tauopathies, half of all patients showed a significant neuronal injury pattern. This highlights the potential of this dual-phase protocol for evaluating two different biomarkers with one examination. While the detailed additive value of early-phase tau-PET imaging ultimately needs to be evaluated in larger samples, we already obtained promising results in individual cases. The patient with a final clinical diagnosis of frontotemporal dementia shown in Fig. 3 was initially referred with suspected AD, and the combination of tau-negativity together with a frontal hypoperfusion pattern was in line with the clinical presentation after 1-year follow-up. For the evaluation of perfusion-like images as a surrogate of neuronal injury, the comparison with metabolic imaging has the restriction that even a metabolic correlate might not unequivocally represent the actually existing neuronal injury. Temporary neuronal dysfunctions, for example, in the context of a diaschisis or neuroinflammation, cannot be differentiated from the presence of neuronal damage.
Both reduced metabolism in [18F]FDG-PET and reduced perfusion in perfusion-phase images of different radiotracers are influenced and partially caused by partial volume effects and might not purely reflect the underlying neuronal injury. Nevertheless, a close correlation with atrophy in magnetic resonance imaging [34] or neuropathology findings [13] was demonstrated for metabolic imaging via [18F]FDG. Therefore, most classification schemes, such as ATN [12], list [18F]FDG-PET as a surrogate of neuronal injury together with atrophy in structural magnetic resonance imaging and total tau in cerebrospinal fluid. Our findings clearly demonstrate that dual-phase [18F]PI-2620-PET cannot only provide information on “T” [22] but has also the potential to facilitate assessment of “N”.
Conclusions
The present study demonstrates that early-phase imaging of second-generation [18F]PI-2620 tau-PET can serve as a surrogate biomarker for neuronal injury as it shows excellent semiquantitative and visual agreement with metabolic imaging using [18F]FDG-PET. Dynamic imaging or a dual time-point protocol for tau-PET could replace additional [18F]FDG-PET imaging by indexing two biomarkers in neurodegenerative disease, the distribution of tau and the amount and regional pattern of neuronal injury. The short time required for recording perfusion-like images is a great advantage in terms of patient comfort, examination time, radiation safety and cost-effectiveness.
References
Arendt T, Stieler JT, Holzer M. Tau and tauopathies. Brain Res Bull. 2016;126:238–92. https://doi.org/10.1016/j.brainresbull.2016.08.018.
Rosler TW, Tayaranian Marvian A, Brendel M, Nykanen NP, Hollerhage M, Schwarz SC, et al. Four-repeat tauopathies. Prog Neurobiol. 2019;101644. https://doi.org/10.1016/j.pneurobio.2019.101644.
Villemagne VL, Okamura N. In vivo tau imaging: obstacles and progress. Alzheimers Dement. 2014;10:S254–64. https://doi.org/10.1016/j.jalz.2014.04.013.
Robertson JS, Rowe CC, Villemagne VL. Tau imaging with PET: an overview of challenges, current progress, and future applications. Quarterly J Nuclear Med Molec Imag. 2017;61:405–13. https://doi.org/10.23736/s1824-4785.17.03012-6.
Lemoine L, Leuzy A, Chiotis K, Rodriguez-Vieitez E, Nordberg A. Tau positron emission tomography imaging in tauopathies: the added hurdle of off-target binding. Alzheimers Dement (Amsterdam, Netherlands). 2018;10:232–6. https://doi.org/10.1016/j.dadm.2018.01.007.
Chiotis K, Stenkrona P, Almkvist O, Stepanov V, Ferreira D, Arakawa R, et al. Dual tracer tau PET imaging reveals different molecular targets for (11)C-THK5351 and (11)C-PBB3 in the Alzheimer brain. Eur J Nucl Med Mol Imaging. 2018;45:1605–17. https://doi.org/10.1007/s00259-018-4012-5.
Wren MC, Lashley T, Arstad E, Sander K. Large inter- and intra-case variability of first generation tau PET ligand binding in neurodegenerative dementias. Acta Neuropathol Commun. 2018;6:34. https://doi.org/10.1186/s40478-018-0535-z.
Lohith TG, Bennacef I, Vandenberghe R, Vandenbulcke M, Salinas CA, Declercq R, et al. Brain imaging of Alzheimer dementia patients and elderly controls with (18)F-MK-6240, a PET tracer targeting neurofibrillary tangles. J Nucl Med. 2019;60:107–14. https://doi.org/10.2967/jnumed.118.208215.
Betthauser TJ, Cody KA, Zammit MD, Murali D, Converse AK, Barnhart TE, et al. In vivo characterization and quantification of neurofibrillary tau PET radioligand (18)F-MK-6240 in humans from Alzheimer disease dementia to young controls. J Nucl Med. 2019;60:93–9. https://doi.org/10.2967/jnumed.118.209650.
Wong DF, Comley RA, Kuwabara H, Rosenberg PB, Resnick SM, Ostrowitzki S, et al. Characterization of 3 novel tau radiopharmaceuticals, (11)C-RO-963, (11)C-RO-643, and (18)F-RO-948, in healthy controls and in Alzheimer subjects. J Nucl Med. 2018;59:1869–76. https://doi.org/10.2967/jnumed.118.209916.
Kroth H, Oden F, Molette J, Schieferstein H, Capotosti F, Mueller A, et al. Discovery and preclinical characterization of [(18)F]PI-2620, a next-generation tau PET tracer for the assessment of tau pathology in Alzheimer's disease and other tauopathies. Eur J Nucl Med Mol Imaging. 2019;46:2178–89. https://doi.org/10.1007/s00259-019-04397-2.
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Feldman HH, Frisoni GB, et al. A/T/N: an unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology. 2016;87:539–47. https://doi.org/10.1212/wnl.0000000000002923.
Mente KP, O'Donnell JK, Jones SE, Cohen ML, Thompson NR, Bizzi A, et al. Fluorodeoxyglucose positron emission tomography (FDG-PET) correlation of histopathology and MRI in prion disease. Alzheimer Dis Assoc Disord. 2017;31:1–7. https://doi.org/10.1097/wad.0000000000000188.
Daerr S, Brendel M, Zach C, Mille E, Schilling D, Zacherl MJ, et al. Evaluation of early-phase [(18)F]-florbetaben PET acquisition in clinical routine cases. Neuroimage Clin. 2017;14:77–86. https://doi.org/10.1016/j.nicl.2016.10.005.
Hsiao IT, Huang CC, Hsieh CJ, Hsu WC, Wey SP, Yen TC, et al. Correlation of early-phase 18F-florbetapir (AV-45/Amyvid) PET images to FDG images: preliminary studies. Eur J Nucl Med Mol Imaging. 2012;39:613–20. https://doi.org/10.1007/s00259-011-2051-2.
Florek L, Tiepolt S, Schroeter ML, Berrouschot J, Saur D, Hesse S, et al. Dual time-point [18F]florbetaben PET delivers dual biomarker information in mild cognitive impairment and Alzheimer's disease. J Alzheimers Dis. 2018;66:1105–16. https://doi.org/10.3233/jad-180522.
Tiepolt S, Hesse S, Patt M, Luthardt J, Schroeter ML, Hoffmann KT, et al. Early [(18)F]florbetaben and [(11)C]PiB PET images are a surrogate biomarker of neuronal injury in Alzheimer's disease. Eur J Nucl Med Mol Imaging. 2016;43:1700–9. https://doi.org/10.1007/s00259-016-3353-1.
Meyer PT, Hellwig S, Amtage F, Rottenburger C, Sahm U, Reuland P, et al. Dual-biomarker imaging of regional cerebral amyloid load and neuronal activity in dementia with PET and 11C-labeled Pittsburgh compound B. J Nucl Med. 2011;52:393–400. https://doi.org/10.2967/jnumed.110.083683.
Brendel M, Wagner L, Levin J, Zach C, Lindner S, Bartenstein P, et al. Perfusion-phase [(18)F]THK5351 tau-PET imaging as a surrogate marker for neurodegeneration. J Alzheimers Dis Rep. 2017;1:109–13. https://doi.org/10.3233/adr-170023.
Rodriguez-Vieitez E, Leuzy A, Chiotis K, Saint-Aubert L, Wall A, Nordberg A. Comparability of [(18)F]THK5317 and [(11)C]PIB blood flow proxy images with [(18)F]FDG positron emission tomography in Alzheimer’s disease. J Cereb Blood Flow Metab. 2017;37:740–9. https://doi.org/10.1177/0271678x16645593.
Rostomian AH, Madison C, Rabinovici GD, Jagust WJ. Early 11C-PIB frames and 18F-FDG PET measures are comparable: a study validated in a cohort of AD and FTLD patients. J Nucl Med. 2011;52:173–9. https://doi.org/10.2967/jnumed.110.082057.
Mueller A, Bullich S, Barret O, Madonia J, Berndt M, Papin C, et al. Tau PET imaging with (18)F-PI-2620 in patients with Alzheimer’s disease and healthy controls: a first-in-human study. J Nucl Med. 2019. https://doi.org/10.2967/jnumed.119.236224.
Varrone A, Asenbaum S, Vander Borght T, Booij J, Nobili F, Nagren K, et al. EANM procedure guidelines for PET brain imaging using [18F]FDG, version 2. Eur J Nucl Med Mol Imaging. 2009;36:2103–10. https://doi.org/10.1007/s00259-009-1264-0.
Lammertsma AA, Hume SP. Simplified reference tissue model for PET receptor studies. NeuroImage. 1996;4:153–8. https://doi.org/10.1006/nimg.1996.0066.
Wu Y, Carson RE. Noise reduction in the simplified reference tissue model for neuroreceptor functional imaging. J Cereb Blood Flow Metab. 2002;22:1440–52. https://doi.org/10.1097/01.WCB.0000033967.83623.34.
Hammers A, Allom R, Koepp MJ, Free SL, Myers R, Lemieux L, et al. Three-dimensional maximum probability atlas of the human brain, with particular reference to the temporal lobe. Hum Brain Mapp. 2003;19:224–47. https://doi.org/10.1002/hbm.10123.
Minoshima S, Frey KA, Koeppe RA, Foster NL, Kuhl DE. A diagnostic approach in Alzheimer's disease using three-dimensional stereotactic surface projections of fluorine-18-FDG PET. J Nucl Med. 1995;36:1238–48.
Erkkinen MG, Kim MO, Geschwind MD. Clinical neurology and epidemiology of the major neurodegenerative diseases. Cold Spring Harb Perspect Biol. 2018;10. https://doi.org/10.1101/cshperspect.a033118.
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, et al. NIA-AA research framework: toward a biological definition of Alzheimer's disease. Alzheimers Dement. 2018;14:535–62. https://doi.org/10.1016/j.jalz.2018.02.018.
Hoglinger GU, Respondek G, Stamelou M, Kurz C, Josephs KA, Lang AE, et al. Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord. 2017;32:853–64. https://doi.org/10.1002/mds.26987.
Ng KP, Pascoal TA, Mathotaarachchi S, Therriault J, Kang MS, Shin M, et al. Monoamine oxidase B inhibitor, selegiline, reduces 18F-THK5351 uptake in the human brain. Alzheimers Res Ther. 2017;9:25. https://doi.org/10.1186/s13195-017-0253-y.
van Berckel BN, Ossenkoppele R, Tolboom N, Yaqub M, Foster-Dingley JC, Windhorst AD, et al. Longitudinal amyloid imaging using 11C-PiB: methodologic considerations. J Nucl Med. 2013;54:1570–6. https://doi.org/10.2967/jnumed.112.113654.
Amen DG, Trujillo M, Keator D, Taylor DV, Willeumier K, Meysami S, et al. Gender-based cerebral perfusion differences in 46,034 functional neuroimaging scans. J Alzheimers Dis. 2017;60:605–14. https://doi.org/10.3233/jad-170432.
Alvarez-Linera Prado J, Jimenez-Huete A. Neuroimaging in dementia. Clin Radiol Correlat Radiol. 2019;61:66–81. https://doi.org/10.1016/j.rx.2018.08.004.
Acknowledgements
The authors thank the staff of the departments of nuclear medicine and neurology at the University Hospital LMU Munich. We thank the patients and their families.
Funding
Open Access funding provided by Projekt DEAL. C.P. received research funding from Lüneburg heritage. M.T.B. received research funding from the Felgenhauer-Stiftung, Forschungspool Klinische Studien (University of Cologne), H2020, Medtronic and Boston Scientific. G.U.H was funded by the Deutsche Forschungsgemeinschaft (DFG) under Germany’s Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy—ID 390857198), the German Federal Ministry of Education and Research (BMBF, 01EK1605A HitTau), the NOMIS foundation (FTLD project). and received research support from GE Healthcare and Neuropore.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M.B. received speaker honoraria from GE healthcare and LMI and is an advisor of LMI. M.T.B. received speaker’s honoraria from Medtronic, Boston Scientific, Abbott (formerly St. Jude), GE Medical, UCB and Bial. G.U.H. has ongoing research collaborations with Orion and Prothena; serves as a consultant for AbbVie, AlzProtect, Asceneuron, Biogen, Biohaven, Lundbeck, Novartis, Roche, Sanofi, UCB; received honoraria for scientific presentations from AbbVie, Biogen, Roche, Teva, UCB, and Zambon; and holds a patent on PERK Activation for the Treatment of Neurodegenerative Diseases (PCT/EP2015/068734). All other authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (LMU Munich—application number 17–569) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all patients. All patients provided informed written consent to PET imaging. The study was conducted in accordance with the principles of the Declaration of Helsinki, and approval for scientific data analysis was obtained from the local ethics committee (application number 17–569).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neurology
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Beyer, L., Nitschmann, A., Barthel, H. et al. Early-phase [18F]PI-2620 tau-PET imaging as a surrogate marker of neuronal injury. Eur J Nucl Med Mol Imaging 47, 2911–2922 (2020). https://doi.org/10.1007/s00259-020-04788-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-020-04788-w