Abstract
Objectives
To correlate patterns of 18F-FDG uptake on whole-body PET-CT with MR findings and compare the degree of FDG activity between symptomatic and asymptomatic knees.
Materials and methods
Retrospective database query was performed using codes for knee MRI as well as whole-body PET-CT. Patients with malignant disease involving the knee or hardware were excluded. Patients who had both studies performed within 1 year between 2012 and 2017 were included for analysis. Knee joint osteoarthrosis, meniscal and ligamentous integrity, presence of joint effusion, and synovitis were assessed and recorded. Bone marrow edema lesions (BMELs) were identified, segmented, and analyzed using volumetric analysis. SUVmax was assessed over the suprapatellar joint space, intercondylar notch and Hoffa’s fat pad. Symptomatic and asymptomatic knees were compared in patients with unilateral symptoms.
Results
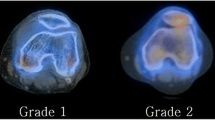
Twenty-two cases (20 patients) with mean age 63.3 years (range, 36–91 years) were included. Two patients had bilateral pain. The most FDG avid regions in both symptomatic and asymptomatic knees were the intercondylar notch (SUVmax = 1.84 vs. 1.51), followed by suprapatellar pouch (SUVmax = 1.74 vs. 1.29) and Hoffa’s fat pad (SUVmax = 1.01 vs. 0.87). SUVmax was significantly associated with cartilage loss (mean modified Outerbridge score) (r = 0.60, p = 0.003) and degree of synovitis (r = 0.48, p = 0023). Overall, mean SUVmax was significantly higher in the presence of a meniscal tear (1.83 ± 0.67 vs. 1.22 ± 0.40, p = 0.030). Nine patients had BMELs (volume: range = 0.6–27.8, mean = 7.79) however there was no significant association between BMEL volume and SUVmax.
Conclusions
Higher FDG activity correlates with intra-articular derangement and the intercondylar notch represents the most metabolically active region of the knee.

Similar content being viewed by others
References
Costelloe CM, Murphy WA Jr, Chasen BA. Musculoskeletal pitfalls in 18F-FDG PET/CT: pictorial review. AJR Am J Roentgenol. 2009;193(3 Suppl):WS1–WS13. Quiz S26-30.
Fischer DR. Musculoskeletal imaging using fluoride PET. Semin Nucl Med. 2013;43(6):427–33.
Burke CJ, Walter WR, Adler RS, Babb JS, Sanger J, Ponzo F. Ultrasound and PET-CT correlation in shoulder pathology: a 5-year retrospective analysis. Clin Nucl Med. 2017;42(10):e424–30.
Elzinga EH, van der Laken CJ, Comans EF, Lammertsma AA, Dijkmans BA, Voskuyl AE. 2-Deoxy-2-[F-18]fluoro-d-glucose joint uptake on positron emission tomography images: rheumatoid arthritis versus osteoarthritis. Mol Imaging Biol. 2007;9(6):357–60.
Hong YH, Kong EJ. (18F)Fluoro-deoxy-d-glucose uptake of knee joints in the aspect of age-related osteoarthritis: a case-control study. BMC Musculoskelet Disord. 2013;14(1):141.
Parsons MA, Moghbel M, Saboury B, Torigian DA, Werner TJ, Rubello D, et al. Increased 18F-FDG uptake suggests synovial inflammatory reaction with osteoarthritis: preliminary in-vivo results in humans. Nucl Med Commun. 2015;36(12):1215–9.
Nakamura H, Masuko K, Yudoh K, Kato T, Nishioka K, Sugihara T, et al. Positron emission tomography with 18F-FDG in osteoarthritic knee. Osteoarthr Cartil. 2007;15(6):673–81.
Saboury B, Parsons MA, Moghbel M, Rubello D, Brothers A, Torigian DA, et al. Quantification of aging effects upon global knee inflammation by 18F-FDG-PET. Nucl Med Commun. 2016;37(3):254–8.
Ju JH, Kang KY, Kim IJ, Yoon JU, Kim HS, Park SH, et al. Visualization and localization of rheumatoid knee synovitis with FDG-PET/CT images. Clin Rheumatol. 2008;27(Suppl 2(S2)):S39–41.
Kubota K, Ito K, Morooka M, Mitsumoto T, Kurihara K, Yamashita H, et al. Whole-body FDG-PET/CT on rheumatoid arthritis of large joints. Ann Nucl Med. 2009;23(9):783–91.
Cimmino MA, Camellino D, Paparo F, Morbelli S, Massollo M, Cutolo M, et al. High frequency of capsular knee involvement in polymyalgia rheumatica/giant cell arteritis patients studied by positron emission tomography. Rheumatology (Oxford). 2013;52(10):1865–72.
Taniguchi Y, Arii K, Kumon Y, Fukumoto M, Ohnishi T, Horino T, et al. Positron emission tomography/computed tomography: a clinical tool for evaluation of enthesitis in patients with spondyloarthritides. Rheumatology (Oxford). 2010;49(2):348–54.
Shao F, Zhang M, Wang Y, Zou Y, Chen Y. Widespread amyloidosis around major joints on 18F-FDG PET/CT. Clin Nucl Med. 2017;42(1):76–8.
de Abreu MR, Falcao M, Sprinz C, Furian R, Marczyk LR. Adhesive capsulitis of the knee. Skelet Radiol. 2013;42(5):741–6.
Okamura K, Yonemoto Y, Okura C, Higuchi T, Tsushima Y, Takagishi K. Evaluation of tocilizumab therapy in patients with rheumatoid arthritis based on FDG-PET/CT. BMC Musculoskelet Disord. 2014;15(1):393.
van der Geest T, Metselaar JM, Gerrits D, van Lent PL, Storm G, Laverman P, et al. [(18)]F FDG PET/CT imaging to monitor the therapeutic effect of liposome-encapsulated prednisolone in experimental rheumatoid arthritis. J Control Release. 2015;209:20–6.
Sorensen J, Michaelsson K, Strand H, Sundelin S, Rahme H. Long-standing increased bone turnover at the fixation points after anterior cruciate ligament reconstruction: a positron emission tomography (PET) study of 8 patients. Acta Orthop. 2006;77(6):921–5.
Itala A, Alihanka S, Kosola J, Kemppainen J, Ranne J, Kajander S. Tendon graft healing in multiligament reconstructed knee detected by FDG-PET/CT: a pilot study. Scand J Surg. 2016;105(2):133–8.
Yue B, Tang T. The use of nuclear imaging for the diagnosis of periprosthetic infection after knee and hip arthroplasties. Nucl Med Commun. 2015;36(4):305–11.
Aksoy SY, Asa S, Ozhan M, Ocak M, Sager MS, Erkan ME, et al. FDG and FDG-labelled leucocyte PET/CT in the imaging of prosthetic joint infection. Eur J Nucl Med Mol Imaging. 2014;41(3):556–64.
Kwee TC, Basu S, Alavi A. Should the nuclear medicine community continue to underestimate the potential of 18F-FDG-PET/CT with present generation scanners for the diagnosis of prosthetic joint infection? Nucl Med Commun. 2015;36(7):756–7.
Stumpe KD, Romero J, Ziegler O, Kamel EM, von Schulthess GK, Strobel K, et al. The value of FDG-PET in patients with painful total knee arthroplasty. Eur J Nucl Med Mol Imaging. 2006;33(10):1218–25.
Roivainen A, Parkkola R, Yli-Kerttula T, Lehikoinen P, Viljanen T, Mottonen T, et al. Use of positron emission tomography with methyl-11C-choline and 2-18F-fluoro-2-deoxy-d-glucose in comparison with magnetic resonance imaging for the assessment of inflammatory proliferation of synovium. Arthritis Rheum. 2003;48(11):3077–84.
Kohl S, Meier S, Ahmad SS, Bonel H, Exadaktylos AK, Krismer A, et al. Accuracy of cartilage-specific 3-Tesla 3D-DESS magnetic resonance imaging in the diagnosis of chondral lesions: comparison with knee arthroscopy. J Orthop Surg Res. 2015;10:191.
Zhang M, Min Z, Rana N, Liu H. Accuracy of magnetic resonance imaging in grading knee chondral defects. Arthroscopy. 2013;29(2):349–56.
Atukorala I, Kwoh CK, Guermazi A, Roemer FW, Boudreau RM, Hannon MJ, et al. Synovitis in knee osteoarthritis: a precursor of disease? Ann Rheum Dis. 2016;75(2):390–5.
Hersh DS, Chun J, Weiner HL, Pulitzer S, Rusinek H, Roth J, et al. Longitudinal quantitative analysis of the tuber-to-brain proportion in patients with tuberous sclerosis. J Neurosurg Pediatr. 2013;12(1):71–6.
Hotca A, Ravichandra S, Mikheev A, Rusinek H, Chang G. Precision of volumetric assessment of proximal femur microarchitecture from high-resolution 3T MRI. Int J Comput Assist Radiol Surg. 2015;10(1):35–43.
Haynes MK, Hume EL, Smith JB. Phenotypic characterization of inflammatory cells from osteoarthritic synovium and synovial fluids. Clin Immunol. 2002;105(3):315–25.
Ishii H, Tanaka H, Katoh K, Nakamura H, Nagashima M, Yoshino S. Characterization of infiltrating T cells and Th1/Th2-type cytokines in the synovium of patients with osteoarthritis. Osteoarthr Cartil. 2002;10(4):277–81.
Haywood L, McWilliams DF, Pearson CI, Gill SE, Ganesan A, Wilson D, et al. Inflammation and angiogenesis in osteoarthritis. Arthritis Rheum. 2003;48(8):2173–7.
Roemer FW, Kassim Javaid M, Guermazi A, Thomas M, Kiran A, Keen R, et al. Anatomical distribution of synovitis in knee osteoarthritis and its association with joint effusion assessed on non-enhanced and contrast-enhanced MRI. Osteoarthr Cartil. 2010;18(10):1269–74.
Kazakia GJ, Kuo D, Schooler J, Siddiqui S, Shanbhag S, Bernstein G, et al. Bone and cartilage demonstrate changes localized to bone marrow edema-like lesions within osteoarthritic knees. Osteoarthr Cartil. 2013;21(1):94–101.
Jungius KP, Schmid MR, Zanetti M, Hodler J, Koch P, Pfirrmann CW. Cartilaginous defects of the femorotibial joint: accuracy of coronal short inversion time inversion-recovery MR sequence. Radiology. 2006;240(2):482–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no relevant conflicts of interest.
Financial disclosures
None.
Additional information
Work was performed at: NYU Langone Orthopedic Hospital, New York, NY
Appendix. Modified Outerbridge grading system [34]
Appendix. Modified Outerbridge grading system [34]
Cartilage loss divided into four grades by MRI, typically using fat-saturated proton-density sequences. This grading system is the modified Outerbridge grading system, which was devised for arthroscopy initially for assessment of chondromalacia patella, but then modified and extended for all chondral surfaces.
-
Grade I
-
focal areas of hyperintensity with normal contour
-
arthroscopically: softening or swelling of cartilage
-
-
Grade II
-
blister-like swelling/fraying of articular cartilage extending to surface
-
arthroscopically: fragmentation and fissuring within soft areas of articular cartilage
-
-
Grade III
-
partial thickness cartilage loss with focal ulceration
-
arthroscopically: partial thickness cartilage loss with fibrillation (crab-meat appearance)
-
-
Grade IV
-
full-thickness cartilage loss with underlying bone reactive changes
-
arthroscopically: cartilage destruction with exposed subchondral bone
-
Rights and permissions
About this article
Cite this article
Burke, C.J., Walter, W.R., Gaddam, S. et al. Correlation of benign incidental findings seen on whole-body PET-CT with knee MRI: patterns of 18F-FDG avidity, intra-articular pathology, and bone marrow edema lesions. Skeletal Radiol 47, 1651–1660 (2018). https://doi.org/10.1007/s00256-018-3001-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-018-3001-x