Abstract
Objective
Fluorodeoxyglucose (FDG) uptake in joint lesions in patients with rheumatoid arthritis (RA) reportedly represents the degree of synovial inflammation. Most previous studies have focused on small joints, and the application of whole-body positron emission tomography (PET) combined with computed tomography (CT) (PET/CT) for the evaluation of inflammatory activity in large joints has not been well studied.
Methods
Eighteen patients with RA underwent FDG-PET/CT. FDG uptake in the knee, hip, carpal, wrist, elbow, shoulder, and atlanto-axial joint (total of 13 joints) and in the axillary lymph nodes was evaluated by calculating the maximum standardized uptake value (SUVmax) and the visual uptake scores as follows: 0, no uptake; 1, slight uptake; 2, moderate uptake (same as in liver); 3, higher than in liver; 4, highest uptake. The number of painful/swollen joints, the white blood cell (WBC) count, and the C-reactive protein (CRP) level were also evaluated.
Results
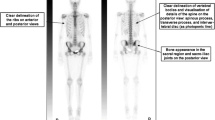
Whole-body FDG-PET/CT delineated large-joint lesions in patients with RA, and the metabolic activity of inflammation was accurately overlaid on the joint anatomy. The total FDG score for all 13 joints was significantly correlated with the CRP level (r = 0.653, p < 0.01, n = 18). The total SUVmax and the CRP level were weakly, but not significantly, correlated (r = 0.377, p > 0.05). The WBC count was not correlated with any other parameter. The mean number of joints per patient with an FDG uptake score of 2 or more was significantly larger than the mean number of painful/swollen joints (6.2 ± 3.3 vs. 3.1 ± 2.7, n = 18, p < 0.01) and both parameters were strongly correlated (r = 0.588, p < 0.01, n = 18). Also, FDG uptake score and SUV of painful/swollen joints were significantly higher than these of not painful/swollen joints. FDG uptake was significantly different from patients of remission and patients of active arthritis. Uptake in the atlanto-axial joint was observed in five (mostly asymptomatic) patients (5/18, 28%), and the uptake score was significantly correlated with the total FDG score (r = 0.669, p < 0.01, n = 18). The axillary lymph nodes score was correlated with the arm joints score.
Conclusion
FDG-PET/CT represents the inflammatory activity in large joints in patients with RA accurately and sensitively and may be helpful for early evaluations of the extent of RA throughout the whole body including high risk lesion of atlanto-axial joint. Furthermore, the visual FDG uptake score may be useful for evaluating arthritis in large joints.





Similar content being viewed by others
References
Majithia V, Geraci SA. Rheumatoid arthritis: diagnosis and management. Am J Med. 2007;120:936–9.
Boutry N, More M, Flipo RM, Demondion X, Cotten A. Early rheumatoid arthritis: a review of MRI and sonographic findings. Am J Roentgenol. 2007;189:1502–9.
Tahara T, Ichiya Y, Kuwabara Y, Otsuka M, Miyake Y, Gunasekera R, et al. High [18F]fluorodeoxyglucose uptake in abdominal abscess: a PET study. J Coumput Assist Tomogr. 1989;13:829–31.
Kubota R, Yamada S, Kubota K, Ishiwata K, Tamahashi N, Ido T. Intratumoral distribution of fluorine-18-fluorodeosxyglucose in vivo: high accumulation in macrophages and granulation tissues studied by microautoradiography. J Nucl Med. 1992;33:1972–80.
Yamada S, Kubota K, Kubota R, Ido T, Tamahashi N. High accumulation of fluorine-18-fluorodeoxyglucose in turpentine-induced inflammatory tissue. J Nucl Med. 1995;36:1301–6.
El-Haddad G, Alavi A, Zhuang H. Value of 18-Fluoro-2-deoxyglucose PET in the management of patients with fever of unknown origin. In: Alavi A, Zhuang H, editors. Infection and inflammation. PET Clinics, vol 1, no. 2. Philadelphia: Saunders; 2006. pp. 163–78.
Palmer WE, Rosenthal DJ, Schoenberg OI, Fischman AJ, Simon LS, Rubin RH, et al. Quantification of inflammation in the wrist with gadolinium-enhanced MR imaging and PET with 2-[F-18]-fluoro-2-deoxy-d-glucose. Radiology. 1995;196:647–55.
Beckers C, Ribbens C, André B, Marcelis S, Kaya O, Mathy L, et al. Assessment of disease activity in rheumatoid arthritis with 18F-FDG PET. J Nucl Med. 2004;45:956–64.
Beckers C, Jeukens X, Ribbens C, André B, Marcelis S, Leclercq P, et al. 18F-FDG PET imaging of rheumatoid knee synovitis correlates with dynamic magnetic resonance and sonographic assessments as well as with the serum level of metalloproteinase-3. Eur J Nucl Med Mol Imaging. 2006;33:275–80.
Goerres GW, Forster A, Uebelhart D, Seifert B, Treyer V, Michel B, et al. F-18 FDG whole-body PET for the assessment of disease activity in patients with rheumatoid arthritis. Clin Nucl Med. 2006;31:386–90.
Arnett FC, Edworthy SM, Block DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315–24.
Guidelines for the management of rheumatoid arthritis. American College of Rheumatology Ad Hoc Committee on clinical guidelines. Arthritis Rheum. 1996;39:713–22.
Seldin DW, Habib I, Soudry G. Axillary lymph node visualization of F-18 FDG PET body scan in patients with rheumatoid arthritis. Clin Nucl Med. 2007;32:524–6.
Calgüneri M, Oztürk MA, Ozbalkan Z, Akdogan A, Ureten K, Kiraz S, et al. Frequency of lymphadenopathy in rheumatoid arthritis and systemic lupus erythematosus. J Int Med Res. 2003;31:345–9.
Neva MH, Häkkinen A, Mäkinen H, Hannonen P, Kauppi M, Sokka T. High prevalence of asymptomatic cervical spine subluxation in patients with rheumatoid arthritis waiting for orthopaedic surgery. Ann Rheum Dis. 2006;65:884–8.
Kaneta T, Hakamatsuka T, Yamada T, Takase K, Sato A, Higano S, et al. Atlantoaxial osteoarthritis in rheumatoid arthritis: FDG PET/CT findings. Clin Nucl Med. 2006;31:209.
Linn-Rasker SP, van der Helm-van Mil AH, Breedveld FC, Huizinga TWJ. Arthritis of the large joints-in particular, the knee-at first presentation is predictive fro a high level of radiological destruction of the small joints in rheumatoid arthritis. Ann Rheum Dis. 2007;66:646–50.
Backhaus M, Kamradt T, Sandrok D, et al. Arthritis of the finger joints: a comprehensive approach comparing conventional radiograph, scintigraphy, ultrasound, and contrast-enhanced magnetic resonance imaging. Arthritis Rheum. 1999;42:1232–45.
Ribbens C, Martiny Porras M, Franchimont N, Kaiser M-J, Jaspar J-M, et al. Increase matrix metalloproteinase-3 serum levels in rheumatic diseases: relationship with synovitis and steroid treatment. Ann Rheum Dis. 2002;61:161–6.
Hodgson RJ, Connor PO, Moots R. MRI of rheumatoid arthritis-image quantification for the assessment of disease activity, progression and response to therapy. Rheumatology. 2008;47:13–21.
Taouli B, Zaim S, Peterfy CG, Lynch JA, Stork A, Guermazi A, et al. Rheumatoid arthritis of the hand and wrist: comparison of three imaging techniques. Am J Roentgenol. 2004;182:937–43.
Acknowledgments
We would like to thank Kazuhiko Nakajima, M.Sc., Yasuhiro Kashimura, BS, and Akio Ishibashi, BS, for their excellent technical assistance. Part of this study was supported by a Grant-in-aid for cancer research (No. 17-12) from the Ministry of Health, Labor and Welfare and by a Grant for International Health Research (21A126) from the Ministry of Health, Labor and Welfare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kubota, K., Ito, K., Morooka, M. et al. Whole-body FDG-PET/CT on rheumatoid arthritis of large joints. Ann Nucl Med 23, 783–791 (2009). https://doi.org/10.1007/s12149-009-0305-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-009-0305-x