Abstract
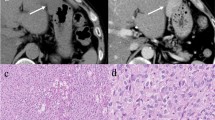
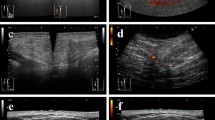
We report the case of a 59-year-old female with progressive bilateral painful swelling of the thighs. MRI revealed multiple intramuscular necrotic masses with similar morphologic patterns. Whole-body CT and 18-FDG PET-CT scans demonstrated additional hypermetabolic muscular masses and a lobulated lesion within the left atrial cavity. As biopsy of a muscular mass was compatible with a poorly differentiated sarcoma with MDM2 oncogene amplification, two diagnoses were discussed: a dedifferentiated liposarcoma with muscle and heart metastases or a primary cardiac sarcoma, mainly a cardiac intimal sarcoma, with muscular metastases, which was finally confirmed by array-comparative genomic hybridization (aCGH) in a sarcoma reference center. This case emphasizes the potential for intimal sarcoma to disseminate in skeletal muscle prior to any other organ and the need for a genomic approach in addition to classical radiopathologic analyses to distinguish primary from secondary locations facing simultaneous tumors of the heart and skeletal muscles with MDM2 amplification.




Similar content being viewed by others
References
Neuville A, Collin F, Bruneval P, Parrens M, Thivolet F, Gomez-Brouchet A, et al. Intimal sarcoma is the most frequent primary cardiac sarcoma: clinicopathologic and molecular retrospective analysis of 100 primary cardiac sarcomas. Am J Surg Pathol. 2014;38(4):461–9.
Sparrow PJ, Kurian JB, Jones TR, Sivananthan MU. MR imaging of cardiac tumors. Radiographics. 2005;25(5):1255–76.
Butany J, Nair V, Naseemuddin A, Yau T, et al. Cardiac tumours: diagnosis and management. Lancet Oncol. 2005;6(4):219–28.
Butany J, Leong SW, Carmichael K, Komeda M. A 30-year analysis of cardiac neoplasms at autopsy. Can J Cardiol. 2005;21(8):675–80.
Mayer F, Aebert H, Hartmann JT, et al. Primary malignant sarcomas of the heart and great vessels in adult patients—a single-center experience. Oncologist. 2007;12(9):1134–42.
Agaimy A, Rösch J, Weyand M, Strecker T. Primary and metastatic cardiac sarcomas: a 12-year experience at a German heart center. Int J Clin Exp Pathol. 2012;5(9):928–38.
Rashid A, Molloy S, Butt S, et al. Metastatic pulmonary intimal sarcoma presenting as cauda equina syndrome: first report of a case. Spine (Phila Pa 1976). 2008;33(15):E516–20.
Shimizu H, Tanibuchi A, Yozu R, et al. Stroke due to undifferentiated aortic intimal sarcoma with disseminated metastatic lesions. Circulation. 2009;120(25):e290–2.
Yamashiro K, Funabe S, Hattori N, et al. Primary aortic sarcoma: a rare but critical cause of stroke. Neurology. 2015;84(7):755–6.
Hoiczyk M, Iliodromitis K, Erbel R, et al. Intimal sarcoma of the pulmonary artery with unusual findings: a case report. Clin Res Cardiol. 2012;101:397–401.
Binh MB, Sastre-Garau X, Coindre JM, et al. MDM2 and CDK4 immunostainings are useful adjuncts in diagnosing well-differentiated and dedifferentiated liposarcoma subtypes: a comparative analysis of 559 soft tissue neoplasms with genetic data. Am J Surg Pathol. 2005;29(10):1340–7.
Coindre J-M, Pédeutour F, Aurias A. Well-differentiated and dedifferentiated liposarcomas. Virchows Arch. 2010;456(2):167–79.
Molina JE, Edwards JE, Ward HB. Primary cardiac tumors: experience at the University of Minnesota. Thorac Cardiovasc Surg. 1990;38(Suppl 2):183–91.
Burke AP, Cowan D, Virmani R. Primary sarcomas of the heart. Cancer. 1992;69(2):387–95.
Simpson L, Kumar SK, Okuno SH, Schaff HV, Porrata LF, Buckner JC, et al. Malignant primary cardiac tumors: review of a single institution experience. Cancer. 2008;112(11):2440–6.
Glockner JF, White LM, Sundaram M, McDonald DJ. Unsuspected metastases presenting as solitary soft tissue lesions: a fourteen-year review. Skelet Radiol. 2000;29(5):270–4.
Surov A, Hainz M, Holzhausen H-J, Arnold D, Katzer M, Schmidt J, et al. Skeletal muscle metastases: primary tumours, prevalence, and radiological features. Eur Radiol. 2010;20(3):649–58.
Djaldetti M, Sredni B, Zigelman R, Verber M, Fishman P. Muscle cells produce a low molecular weight factor with anti-cancer activity. Clin Exp Metastasis. 1996;14(3):189–96.
Kurek JB, Nouri S, Kannourakis G, Murphy M, Austin L. Leukemia inhibitory factor and interleukin-6 are produced by diseased and regenerating skeletal muscle. Muscle Nerve. 1996;19(10):1291–301.
Bar-Yehuda S, Barer F, Volfsson L, Fishman P. Resistance of muscle to tumor metastases: a role for a3 adenosine receptor agonists. Neoplasia N Y N. 2001;3(2):125–31.
Weiss L. Biomechanical destruction of cancer cells in skeletal muscle: a rate-regulator for hematogenous metastasis. Clin Exp Metastasis. 1989;7(5):483–91.
Pretorius ES, Fishman EK. Helical CT of skeletal muscle metastases from primary carcinomas. Am J Roentgenol. 2000;174(2):401–4.
Ote EL, Oriuchi N, Endo K, et al. Pulmonary artery intimal sarcoma: the role of 18F-fluorodeoxyglucose positron emission tomography in monitoring response to treatment. Jpn J Radiol. 2011;29(4):279–82.
Lee DH, Jung TE, Choi JH et al. Pulmonary artery intimal sarcoma: poor 18F–fluorodeoxyglucose uptake in positron emission computed tomography. J Cardiothorac Surg. 2013:7;8:40.
Acknowledgements
The authors would like to thank Agnès Ribeiro, Pr. Jean-Michel Coindre and Dr. Frederic Chibon for their contribution and expertise concerning the pathologic analysis as well as Mrs. Estelle Ternamian for medical writing services.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Additional information
Drs. Crombé and Lintingre jointly directed this work.
Rights and permissions
About this article
Cite this article
Crombé, A., Lintingre, PF., Le Loarer, F. et al. Multiple skeletal muscle metastases revealing a cardiac intimal sarcoma. Skeletal Radiol 47, 125–130 (2018). https://doi.org/10.1007/s00256-017-2768-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-017-2768-5