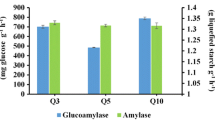
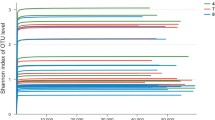
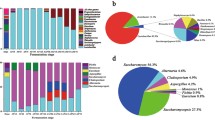
Abstract
Solid-state acetic acid fermentation (AAF), a natural or semi-controlled fermentation process driven by reproducible microbial communities, is an important technique to produce traditional Chinese cereal vinegars. Highly complex microbial communities and metabolites are involved in traditional Chinese solid-state AAF, but the association between microbiota and metabolites during this process are still poorly understood. In this study, we performed amplicon 16S rRNA gene sequencing on the Illumina MiSeq platform, PCR-denaturing gradient gel electrophoresis, and metabolite analysis to trace the bacterial dynamics and metabolite changes under AAF process. A succession of bacterial assemblages was observed during the AAF process. Lactobacillales dominated all the stages. However, Acetobacter species in Rhodospirillales were considerably accelerated during AAF until the end of fermentation. Quantitative PCR results indicated that the biomass of total bacteria showed a “system microbe self-domestication” process in the first 3 days, and then peaked at the seventh day before gradually decreasing until the end of AAF. Moreover, a total of 88 metabolites, including 8 organic acids, 16 free amino acids, and 66 aroma compounds were detected during AAF. Principal component analysis and cluster analyses revealed the high correlation between the dynamics of bacterial community and metabolites.





Similar content being viewed by others
References
Armstrong DW, Martin SM, Yamazaki H (1984) Production of ethyl acetate from dilute ethanol solutions by Candida utilis. Biotechnol Bioeng 26(9):1038–1041
Bartowsky EJ, Pretorius IS (2009) Microbial formation and modification of flavor and off-flavor compounds in wine. In: König H, Unden G, Fröhlich J (eds) Biology of microorganisms on grapes, in must and in wine, 11th edn. Springer, Berlin, pp 209–231
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Pena AG, Goodrich JK, Gordon JI (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336. doi:10.1038/nmeth.f.303
Chang CW, Laird DA, Mausbach MJ, Hurburgh CR (2001) Near-infrared reflectance spectroscopy–principal components regression analyses of soil properties. Soil Sci Soc Am J 65(2):480–490. doi:10.2136/sssaj2001.652480x
Chen JC, Chen QH, Guo Q, Ruan S, Ruan H, He GQ, Gu Q (2010) Simultaneous determination of acetoin and tetramethylpyrazine in traditional vinegars by HPLC method. Food Chem 122(4):1247–1252. doi:10.1016/j.foodchem.2010.03.072
Cheng L, Luo J, Li P, Yu H, Huang J, Luo L (2014) Microbial diversity and flavor formation in onion fermentation. Food Funct 5(9):2338–2347. doi:10.1039/C4FO00196F
De Faveri D, Torre P, Molinari F, Perego P, Converti A (2003) Carbon material balances and bioenergetics of 2, 3-butanediol bio-oxidation by Acetobacter hansenii. Enzym Microbiol Technol 33(5):708–719. doi:10.1016/S0141-0229(03)00218-7
Degnan PH, Ochman H (2012) Illumina-based analysis of microbial community diversity. ISME J 6(1):183–194. doi:10.1038/ismej.2011.74
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10(10):996–998. doi:10.1038/nmeth.2604
Fadda S, Oliver G, Vignolo G (2002) Protein degradation by Lactobacillus plantarum and Lactobacillus casei in a sausage model system. J Food Sci 67(3):1179–1183. doi:10.1111/j.1365-2621.2002.tb09473.x
Guillamón JM, Mas A (2009) Acetic acid bacteria. In: König H, Unden G, Fröhlich J (eds) Biology of microorganisms on grapes, in must and in wine, 2nd edn. Springer, Berlin, pp 31–46
Gullo M, Giudici P (2008) Acetic acid bacteria in traditional balsamic vinegar: phenotypic traits relevant for starter cultures selection. Int J Food Microbiol 125(1):46–53. doi:10.1016/j.ijfoodmicro.2007.11.076
Haruta S, Ueno S, Egawa I, Hashiguchi K, Fujii A, Nagano M, Ishii M, Igarashi Y (2006) Succession of bacterial and fungal communities during a traditional pot fermentation of rice vinegar assessed by PCR-mediated denaturing gradient gel electrophoresis. Int J Food Microbiol 109(1):79–87. doi:10.1016/j.ijfoodmicro.2006.01.015
Ilabaca C, Navarrete P, Mardones P, Romero J, Mas A (2008) Application of culture culture-independent molecular biology based methods to evaluate acetic acid bacteria diversity during vinegar processing. Int J Food Microbiol 126(1):245–249. doi:10.1016/j.ijfoodmicro.2008.05.001
Kashima Y, Iijima M, Okamoto A, Koizumi Y, Udaka S, Yanagida F (1998) Purification and characterization of intracellular esterases related to ethylacetate formation in Acetobacter pasteurianus. J Ferment Bioeng 85(6):584–588. doi:10.1016/S0922-338X(98)80009-3
Kemp PF, Aller JY (2004) Bacterial diversity in aquatic and other environments: what 16S rDNA libraries can tell us. FEMS Microbiol Ecol 47(2):161–177. doi:10.1016/S0168-6496(03)00257-5
Kirchner O, Tauch A (2003) Tools for genetic engineering in the amino acid-producing bacterium Corynebacterium glutamicum. J Biotechnol 104(1):287–299. doi:10.1016/S0168-1656(03)00148-2
Lamoolphak W, Goto M, Sasaki M, Suphantharika M, Muangnapoh C, Prommuag C, Shotipruk A (2006) Hydrothermal decomposition of yeast cells for production of proteins and amino acids. J Hazard Mater 137(3):1643–1648. doi:10.1016/j.jhazmat.2006.05.029
Li P, Li S, Cheng L, Luo L (2014) Analyzing the relation between the microbial diversity of DaQu and the turbidity spoilage of traditional Chinese vinegar. Appl Microbiol Biotechnol 98(13):6073–6084. doi:10.1007/s00253-014-5697-4
Li P, Liang H, Lin WT, Feng F, Luo L (2015a) Microbiota dynamics associated with environmental conditions and potential roles of cellulolytic communities in traditional Chinese cereal starter solid-state fermentation. Appl Environ Microbiol 81(15):5144–5156. doi:10.1128/AEM.01325-15
Li S, Li P, Feng F, Luo LX (2015b) Microbial diversity and their roles in the vinegar fermentation process. Appl Microbiol Biotechnol 99(12):4997–5024. doi:10.1007/s00253-015-6659-1
Lin L, Zhuang M, Lei F, Bao Y, Zhao M (2013) GC/MS analysis of volatiles obtained by headspace solid-phase microextraction and simultaneous-distillation extraction from Rabdosia serra (MAXIM.) HARA leaf and stem. Food Chem 136(2):555–562. doi:10.1016/j.foodchem.2012.08.048
Lindahl BD, Nilsson RH, Tedersoo L, Abarenkov K, Carlsen T, Kjøller R, Kõljalg U, Pennanen T, Rosendahl S, Stenlid J (2013) Fungal community analysis by high‐throughput sequencing of amplified markers—a user’s guide. New Phytol 199(1):288–299. doi:10.1111/nph.12243
Lockwood LB, Nelson GE (1946) The oxidation of pentoses by Pseudomonas. J Bacteriol 52(5):581
Lozupone C, Lladser ME, Knights D, Stombaugh J, Knight R (2011) UniFrac: an effective distance metric for microbial community comparison. ISME J 5(2):169. doi:10.1038/ismej.2010.133
Lu ZM, Xu W, Yu NH, Zhou T, Li GQ, Shi JS, Xu ZH (2011) Recovery of aroma compounds from Zhenjiang aromatic vinegar by supercritical fluid extraction. Int J Food Sci Technol 46(7):1508–1514. doi:10.1111/j.1365-2621.2011.02649.x
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21):2957–2963. doi:10.1093/bioinformatics/btr507
Matsunaga A, Koyama N, Nosoh Y (1974) Purification and properties of esterase from Bacillus stearothermophilus. Arch Biochem Biophys 160(2):504–513. doi:10.1016/0003-9861(74)90427-5
McDonald L, Fleming H, Hassan H (1990) Acid tolerance of Leuconostoc mesenteroides and Lactobacillus plantarum. Appl Environ Microbiol 56(7):2120–2124
Medeiros ABP, Pandey A, Vandenberghe LPS, Pastore GM, Soccol CR (2006) Production and recovery of aroma compounds produced by solid-state fermentation using different adsorbents. Food Technol Biotechnol 44(1):47–51
Meghji K, Ward OP, Araujo A (1991) Production, purification, and properties of extracellular carboxyl esterases from Bacillus subtilis NRRL 365. Appl Environ Microbiol 56(12):3735–3740
Nanda K, Taniguchi M, Ujike S, Ishihara N, Mori H, Ono H, Murooka Y (2001) Characterization of acetic acid bacteria in traditional acetic acid fermentation of rice vinegar (komesu) and unpolished rice vinegar (kurosu) produced in Japan. Appl Environ Microbiol 67(2):986–990. doi:10.1128/AEM.67.2.986-990.2001
Nie Z, Zheng Y, Wang M, Han Y, Wang Y, Luo J, Niu D (2013) Exploring microbial succession and diversity during solid-state fermentation of Tianjin duliu mature vinegar. Bioresour Technol 148:325–333. doi:10.1016/j.biortech.2013.08.152
Nie Z, Zheng Y, Du H, Xie S, Wang M (2015) Dynamics and diversity of microbial community succession in traditional fermentation of Shanxi aged vinegar. Food Microbiol 47:62–68. doi:10.1016/j.fm.2014.11.006
Papagianni M (2007) Advances in citric acid fermentation by Aspergillus niger: biochemical aspects, membrane transport and modeling. Biotechnol Adv 25(3):244–263. doi:10.1016/j.biotechadv.2007.01.002
Peiffer JA, Spor A, Koren O, Jin Z, Tringe SG, Dangl JL, Buckler ES, Ley RE (2013) Diversity and heritability of the maize rhizosphere microbiome under field conditions. Proc Natl Acad Sci U S A 110(16):6548–6553. doi:10.1073/pnas.1302837110
Peleg Y, Stieglitz B, Goldberg I (1988) Malic acid accumulation by Aspergillus flavus. Appl Microbiol Biotechnol 28(1):69–75. doi:10.1007/BF00250501
Pfeiler EA, Klaenhammer TR (2007) The genomics of lactic acid bacteria. Trends Microbiol 15(12):546–553. doi:10.1016/j.tim.2007.09.010
Pines O, Even-Ram S, Elnathan N, Battat E, Aharonov O, Gibson D, Goldberg I (1996) The cytosolic pathway of L-malic acid synthesis in Saccharomyces cerevisiae: the role of fumarase. Appl Microbiol Biotechnol 46(4):393–399. doi:10.1007/BF00166235
Plata C, Millan C, Mauricio J, Ortega J (2003) Formation of ethyl acetate and isoamyl acetate by various species of wine yeasts. Food Microbiol 20(2):217–224
Ragsdale S (1990) Mechanism of formation of the carboxyl of acetate by acetogenic bacteria. Wisconsin Univ, Milwaukee, WI (USA)
Rojas V, Gil JV, Piñaga F, Manzanares P (2003) Acetate ester formation in wine by mixed cultures in laboratory fermentations. J Food Microbiol 86(1):181–188. doi:10.1016/S0168-1605(03)00255-1
Shimoji Y, Tamura Y, Nakamura Y, Nanda K, Nishidai S, Nishikawa Y, Ishihara N, Uenakai K, Ohigashi H (2002) Isolation and identification of DPPH radical scavenging compounds in Kurosu (Japanese unpolished rice vinegar). J Agric Food Chem 50(22):6501–6503. doi:10.1021/jf020458f
Sierra G (1967) Dissociation of esterase from proteinase activity of Bacillus subtilis Spores. Can J of Microbiol 13(6). doi:10.1139/m67-089
Tesfaye W, Morales M, Garcıa Parrilla M, Troncoso A (2002) Wine vinegar: technology, authenticity and quality evaluation. Trends Food Sci Technol 13(1):12–21. doi:10.1016/S0924-2244(02)00023-7
Unden G, Zaunmüller T (2009) Metabolism of sugars and organic acids by lactic acid bacteria from wine and must. In: König H, Unden G, Fröhlich J (eds) Biology of microorganisms on grapes, in must and in wine, 7th edn. Springer, Berlin, pp 135–147
Vegas C, Mateo E, González Á, Jara C, Guillamón JM, Poblet M, Torija MJ, Mas A (2010) Population dynamics of acetic acid bacteria during traditional wine vinegar production. Int J Food Microbiol 138(1):130–136. doi:10.1016/j.ijfoodmicro.2010.01.006
Vegas C, González Á, Mateo E, Mas A, Poblet M, Torija MJ (2013) Evaluation of representativity of the acetic acid bacteria species identified by culture-dependent method during a traditional wine vinegar production. Food Res Int 51(1):404–411. doi:10.1016/j.foodres.2012.12.055
Wu JJ, Ma YK, Zhang FF, Chen FS (2012a) Biodiversity of yeasts, lactic acid bacteria and acetic acid bacteria in the fermentation of “Shanxi aged vinegar”, a traditional Chinese vinegar. Food Microbiol 30(1):289–297. doi:10.1016/j.fm.2011.08.010
Wu JJ, Ma YK, Zhang FF, Chen FS (2012b) Culture-dependent and culture-independent analysis of lactic acid bacteria from Shanxi aged vinegar. Ann Microbiol 62(4):1825–1830. doi:10.1007/s13213-011-0396-1
Xiao Z, Xu P (2007) Acetoin metabolism in bacteria. Crit Rev Microbiol 33(2):127–140. doi:10.1080/10408410701364604
Xu P, Xiao Z, Wei Z, Du Y (2011) Bacillus pumilus strain for high yield of tetramethylpyrazine. US
Xu W, Huang Z, Zhang X, Li Q, Lu Z, Shi J, Xu Z, Ma Y (2011b) Monitoring the microbial community during solid-state acetic acid fermentation of Zhenjiang aromatic vinegar. Food Microbiol 28(6):1175–1181. doi:10.1016/j.fm.2011.03.011
Zhang Q, Zhang S, Xie C, Zeng D, Fan C, Li D, Bai Z (2006) Characterization of Chinese vinegars by electronic nose. Sensors Actuators B Chem 119(2):538–546. doi:10.1016/j.snb.2006.01.007
Acknowledgments
This project was supported by the National Natural Foundation of China (No. 31271924).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
We confirm that this manuscript has not been published elsewhere and is not under consideration by another journal. This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
This project was supported by the National Natural Foundation of China (No. 31271924).
Conflict of interest
The authors declare no conflict of interest.
Additional information
Sha Li and Pan Li contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1201 kb)
Rights and permissions
About this article
Cite this article
Li, S., Li, P., Liu, X. et al. Bacterial dynamics and metabolite changes in solid-state acetic acid fermentation of Shanxi aged vinegar. Appl Microbiol Biotechnol 100, 4395–4411 (2016). https://doi.org/10.1007/s00253-016-7284-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7284-3