Abstract
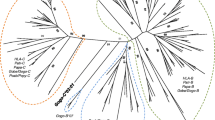
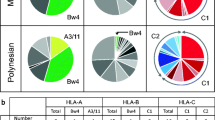
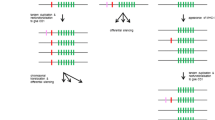
The immunoglobulin-like receptor (KIR) gene family in New World primates (Platyrrhini) has been characterized only in the owl monkey (Aotus sp.). To gain a better understanding of the KIR system in Platyrrhini, we analyzed a KIR haplotype in Ateles geoffroyi, and sequenced KIR complementary DNAs (cDNAs) from other three Atelidae species, Ateles hybridus, Ateles belzebuth, and Lagothrix lagotricha. Atelidae expressed a variable set of activating and inhibitory KIRs that diversified independently from their Catarrhini counterparts. They had a unique mechanism to generate activating receptors from inhibitory ones, involving a single nucleotide deletion in exon 7 and a change in the donor splice site of intron 7. The A. geoffroyi haplotype contained at least six gene models including a pseudogene, two coding inhibitory receptors, and three coding activating receptors. The centromeric region was in a tail-to-tail orientation with respect to the telomeric region. The owl monkey KIR haplotype shared this organization, and in phylogenetic trees, the centromeric genes clustered together with those of A. geoffroyi, whereas their telomeric genes clustered independently. KIR cDNAs from the other Atelidae species conformed to this pattern. Signatures of positive selection were found in residues predicted to interact with the major histocompatibility complex. Such signatures, however, primarily explained variability between paralogous genes but not between alleles in a locus. Atelidae, therefore, has expanded the KIR family in a bimodal fashion, where an inverted centromeric region has remained relatively conserved and the telomeric region has diversified by a rapid process of gene duplication and divergence, likely favored by positive selection for ligand binding.




Similar content being viewed by others
References
Abi-Rached L, Parham P (2005) Natural selection drives recurrent formation of activating killer cell immunoglobulin-like receptor and Ly49 from inhibitory homologues. J Exp Med 201(8):1319–1332
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22(2):195–201
Averdam A, Petersen B, Rosner C, Neff J, Roos C, Eberle M, Aujard F, Munch C, Schempp W, Carrington M, Shiina T, Inoko H, Knaust F, Coggill P, Sehra H, Beck S, Abi-Rached L, Reinhardt R, Walter L (2009) A novel system of polymorphic and diverse NK cell receptors in primates. PLoS Genet 5(10):e1000688
Barrow AD, Trowsdale J (2008) The extended human leukocyte receptor complex: diverse ways of modulating immune responses. Immunol Rev 224:98–123
Benkert P, Biasini M, Schwede T (2011) Toward the estimation of the absolute quality of individual protein structure models. Bioinformatics 27(3):343–350
Blokhuis JH, van der Wiel MK, Doxiadis GG, Bontrop RE (2011) The extreme plasticity of killer cell Ig-like receptor (KIR) haplotypes differentiates rhesus macaques from humans. Eur J Immunol 41(9):2719–2728
Cadavid LF, Lun CM (2009) Lineage-specific diversification of killer cell Ig-like receptors in the owl monkey, a New World primate. Immunogenetics 61(1):27–41
Campbell KS, Purdy AK (2011) Structure/function of human killer cell immunoglobulin-like receptors: lessons from polymorphisms, evolution, crystal structures and mutations. Immunology 132(3):315–325
Cheent K, Khakoo SI (2009) Natural killer cells: integrating diversity with function. Immunology 126(4):449–457
Dobromylskyj M, Ellis S (2007) Complexity in cattle KIR genes: transcription and genome analysis. Immunogenetics 59(6):463–472
Fabre PH, Rodrigues A, Douzery EJ (2009) Patterns of macroevolution among primates inferred from a supermatrix of mitochondrial and nuclear DNA. Mol Phylogenet Evol 53(3):808–825
Guethlein LA, Abi-Rached L, Hammond JA, Parham P (2007a) The expanded cattle KIR genes are orthologous to the conserved single-copy KIR3DX1 gene of primates. Immunogenetics 59(6):517–522
Guethlein LA, Older Aguilar AM, Abi-Rached L, Parham P (2007b) Evolution of killer cell Ig-like receptor (KIR) genes: definition of an orangutan KIR haplotype reveals expansion of lineage III KIR associated with the emergence of MHC-C. J Immunol 179(1):491–504
Hao L, Nei M (2005) Rapid expansion of killer cell immunoglobulin-like receptor genes in primates and their coevolution with MHC Class I genes. Gene 347(2):149–159
Hershkovitz P (1977) Living New World monkeys (Platyrrhini) with an introduction to primates, vol 1. University of Chicago Press, Chicago
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17(8):754–755
Humphrey W, Dalke A, Schulten K (1996) VMD: visual molecular dynamics. J Mol Graph 14(1):33–38, 27–38
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Lanier LL (1998) Activating and inhibitory NK cell receptors. Adv Exp Med Biol 452:13–18
Lanier LL (2003) Natural killer cell receptor signaling. Curr Opin Immunol 15(3):308–314
McQueen KL, Parham P (2002) Variable receptors controlling activation and inhibition of NK cells. Curr Opin Immunol 14(5):615–621
Moretta L, Mingari MC, Bottino C, Pende D, Biassoni R, Moretta A (2003) Cellular and molecular basis of natural killer and natural killer-like activity. Immunol Lett 88(2):89–93
Murrell B, Wertheim JO, Moola S, Weighill T, Scheffler K, Kosakovsky Pond SL (2012) Detecting individual sites subject to episodic diversifying selection. PLoS Genet 8(7):e1002764
Opazo JC, Wildman DE, Prychitko T, Johnson RM, Goodman M (2006) Phylogenetic relationships and divergence times among New World monkeys (Platyrrhini, primates). Mol Phylogenet Evol 40(1):274–280
Palacios C, Cuervo L, Cadavid LF (2011) Evolutionary patterns of killer cell Ig-like receptors in Old World monkeys. Gene 474(1–2):39–51
Parham P, Abi-Rached L, Matevosyan L, Moesta AK, Norman PJ, Older Aguilar AM, Guethlein LA (2010) Primate-specific regulation of natural killer cells. J Med Primatol 39(4):194–212
Parham P, Moffett A (2013) Variable NK cell receptors and their MHC class I ligands in immunity, reproduction and human evolution. Nat Rev Immunol 13(2):133–144
Pertea M, Lin X, Salzberg SL (2001) GeneSplicer: a new computational method for splice site prediction. Nucleic Acids Res 29(5):1185–1190
Porter CA, Czelusniak J, Schneider H, Schneider MP, Sampaio I, Goodman M (1997) Sequences of the primate epsilon-globin gene: implications for systematics of the marmosets and other New world primates. Gene 205(1–2):59–71
Posada D, Crandall KA (1998) MODELTEST: testing the model of DNA substitution. Bioinformatics 14(9):817–818
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19(12):1572–1574
Ryan JC, Seaman WE (1997) Divergent functions of lectin-like receptors on NK cells. Immunol Rev 155:79–89
Sambrook JG, Bashirova A, Andersen H, Piatak M, Vernikos GS, Coggill P, Lifson JD, Carrington M, Beck S (2006) Identification of the ancestral killer immunoglobulin-like receptor gene in primates. BMC Genomics 7:209
Sambrook JG, Bashirova A, Palmer S, Sims S, Trowsdale J, Abi-Rached L, Parham P, Carrington M, Beck S (2005) Single haplotype analysis demonstrates rapid evolution of the killer immunoglobulin-like receptor (KIR) loci in primates. Genome Res 15(1):25–35
Sawai H, Kawamoto Y, Takahata N, Satta Y (2004) Evolutionary relationships of major histocompatibility complex class I genes in simian primates. Genetics 166(4):1897–1907
Scheffler K, Martin DP, Seoighe C (2006) Robust inference of positive selection from recombining coding sequences. Bioinformatics 22(20):2493–2499
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680
Vivian JP, Duncan RC, Berry R, O'Connor GM, Reid HH, Beddoe T, Gras S, Saunders PM, Olshina MA, Widjaja JM, Harpur CM, Lin J, Maloveste SM, Price DA, Lafont BA, McVicar DW, Clements CS, Brooks AG, Rossjohn J (2011) Killer cell immunoglobulin-like receptor 3DL1-mediated recognition of human leukocyte antigen B. Nature 479(7373):401–405
Acknowledgments
This work was supported by a grant from the Universidad Nacional de Colombia’s Research Division to LFC. We thank Marcela Fuquen and Alejandro Cerón for technical support. Colombia’s Ministry of the Environment and Sustainable Development authorized the access to genetic resources for scientific research without commercial interests (contract no. 1 of 2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Nucleotide sequence data reported are available in the GenBank database under accession numbers KF011940-KF011974.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Deduced amino acid sequence alignment of Platyrrhini KIRs. Atge, A. geoffroyi; Athy, A. hybridus; Atbe, A. belzebuth; Lala, L. lagotricha; Om, owl monkey. (DOC 103 kb)
Supplementary Fig. 2
Homology modeling of Platyrrhini predicted KIR3DL proteins. a QMEAN4 values and root mean square deviation (RMSD) for representative structure models from the four Platyrrhini clades showed in Fig. 3b. Structure models were constructed using as template the crystallized structure of KIR3DL1*001 (PDB 3VH8). The allele KIR3DL1*031 was also modeled to be used as a baseline comparison. b Superposition analysis between KIR3DL1*001 (gray) and the structural model with the highest RMSD value, Atge-KIR3DL2 (blue), showing a nearly exact match. (JPEG 655 kb)
Supplementary Fig. 3
Atelidae KIR3DL sequences are divided into three classes according the length and presence of functional motifs in the cytoplasmic domain. (JPEG 7771 kb)
Rights and permissions
About this article
Cite this article
Cadavid, L.F., Palacios, C. & Lugo, J.S. Bimodal evolution of the killer cell Ig-like receptor (KIR) family in New World primates. Immunogenetics 65, 725–736 (2013). https://doi.org/10.1007/s00251-013-0719-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-013-0719-4