Abstract
The increasing proliferation of cyanobacterial blooms prolongs the impact of cyanobacteria on aquatic fauna, potentially altering trophic relationships. We hypothesized that any effect of dissolved microcystins (toxins produced by cyanobacteria) on plankton assemblages would be more evident in artificial reservoirs and ponds than in natural ones. The concentrations of dissolved microcystins in the waters we studied ranged widely from 0.07 to 0.81 μg/L. We showed that the artificial ponds were subjected to more frequent and longer-lasting harmful algal blooms. The plankton occurring in them were exposed to significantly higher concentrations of dissolved microcystins than those in natural oxbow lakes. Using a general linear model (GLM) regression, our study identified a significant relationship between dissolved microcystins and both the density and biomass of particular zooplankton groups (ciliates, rotifers, cladocerans, copepods). The density, biomass, and richness of the animal plankton were significantly lower in the artificial ponds than in the natural oxbow lakes. The impact of microcystins and the length of time that they remained in the water caused structural homogenization of the plankton.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Growing en masse in water, cyanobacteria create a phenomenon known as cyanobacterial blooms. Local and global warming and increasing anthropogenic eutrophication and pollution of water have led to the proliferation of harmful algal blooms (HABs) that show accelerated and prolonged activity [1]. Because “cyanoHABs” are toxic, cause hypoxia, decrease biodiversity, and disrupt food webs [2, 3], they present a serious threat to water ecosystems.
The most threatened ecosystems are those in small, shallow reservoirs, ponds, and oxbow lakes, which are biodiversity hotspots, serve as water migration corridors, diversify the landscape, and provide habitats for many rare and valuable species [4,5,6]. Because they are naturally eutrophic, these types of waterbodies naturally host cyanobacterial blooms, but the increasing proliferation of such blooms adds a new factor: it prolongs the impact of cyanobacteria on aquatic fauna, potentially altering trophic relationships, damaging these exceptionally important ecosystems, and compromising their ecosystem services.
Cyanobacteria change trophic interactions through several mechanisms. First, they are a poor food source due to their large size, low digestibility [7] and lack of long-chain polyunsaturated fatty acids (PUFAs) [8]. Second, they produce toxins. The most common of the several types of cyanotoxins are microcystins. Microcystins are produced by and retained in cyanobacterial cells during the growth and stationary phases of blooms [9]. When the blooms decay and their cells deteriorate, metabolites are released, raising the concentration of toxins in the water. The presence of microcystins is reported in 50 to 90% of samples taken during bloom events [10]. Toxins released in the water can remain there for up to 3 weeks [11], causing harm even after the cyanobacteria are gone. More than 100 microcystin analogues are known [12]. The analogues differ in toxicity; microcystin-LR (MC-LR) has been found to be the most toxic one, followed by microcystin-YR (MC-YR) and microcystin-RR (MC-RR) [13]. It is well known that microcystins harm humans and other mammals by altering cell metabolism and triggering a cascade of events that leads to cell necrosis or apoptosis [14]. Such effects do not require direct contact with cyanobacteria cells and occur even if the toxins cannot readily diffuse across the plasma membrane. There is evidence that hydrophobic toxins (e.g., MC-YR) can affect membranes that have packing defects [15]. Some hydrophobic microcystins can, by pinocytosis, penetrate the cell along with other material associated with the plasma membrane [16].
Dissolved cyanobacterial toxins released during bloom decay have negative effects on feeding and on the growth of fish larvae [17]. Cyanotoxins may be transferred to higher trophic levels through primary consumers such as protozooplankton [18] and metazooplankton [19]. Relatively little is known about the response of plankton to toxins, especially to dissolved toxins. It is difficult to draw conclusions about the processes and relationships that operate during CyanoHAB events, and effects measured in the laboratory may not always mirror the natural processes that occur in the field [20].
Finally, cyanotoxins may harm humans following chronic exposure to low concentrations of microcystins via consumption of contaminated water and food (e.g., agricultural products, fish, prawns, mollusks), dermal exposure, and inhalation [14].
Some species feed on cyanobacteria and are exposed to the toxins present in cyanobacterial cells. Many more species are exposed to cyanotoxins dissolved in the water. It is ever more important to understand how the presence of dissolved microcystins affects the structure and trophic network of plankton communities. Some field and laboratory studies have shown that toxins dissolved in the water affect the protozooplankton and metazooplankton living there [21,22,23].
Protozooplankton and metazooplankton organisms are basic and critical parts of the food web in aquatic ecosystems, able to transfer carbon to higher levels [24]. We studied the effect of dissolved microcystins on the shape of protozooplankton and metazooplankton assemblages in small waterbodies. With increasing anthropopression, we will see further proliferation of CyanoHABs. We need to know exactly how plankton assemblages will be affected by those blooms. For this study, we postulated that the effect of dissolved microcystins on plankton assemblages would be more pronounced in artificial waterbodies than in natural ones.
Material and Methods
Study Area and Materials
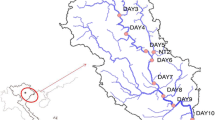
This study used samples from four waterbodies in which cyanobacterial blooms occur: two natural oxbow lakes (Piekary, P; Tyniec, T) formed by the Vistula River and two artificial ponds (Podkamycze 1, P1; Podkamycze 2, P2) (Fig. 1). All the studied waterbodies are relatively small, covering 1.56–17.28 ha and ranging in maximum depth from 2.5 and 4.0 m. They all are classified as eutrophic [25] and are near each other, so their weather conditions are very similar.
Sampling Procedure
Samples were collected from the central point of each waterbody between May and October 2014. Sampling was done each month before cyanobacterial blooms formed and then each week during bloom events. In total, 64 sample sets were collected for biological analyses (cyanobacteria, ciliates, metazooplankton) and to determine the concentration of microcystins in the water. Because the studied oxbow lakes are shallow and polymictic, they were not stratified into epilimnion, metalimnion and hypolimnion.
Although, the Ruttner sampler is not a perfect device for quantifying zooplankton abundance [26, 27], but it is broadly use in ecological studies [28]; therefore, we decided to use it. Samples were taken at 1 m depth using a 5-L Ruttner sampler and were concentrated from 10 L with plankton nets (mesh sizes 10 μm for cyanobacteria and ciliates; 50 μm for metazooplankton). Immediately after collection, the samples were fixed for quantitative analyses (with Lugol’s solution for algae and ciliates; with 4% formaldehyde for metazooplankton). Additional fresh (not fixed) samples were concentrated as described above, and the live material was taken for species composition analysis. See [24] for the keys used for taxonomic identification of cyanobacteria. The living ciliates were identified in 1 mL chambers with glass covers, according to [28] and [29], and their density was averaged from three counts. Total biomass of ciliates was calculated according to [30,31,32,33].
Metazooplankton samples were analyzed in 0.5 mL chambers, and their density was calculated as means of five counts. The keys we used for identification of animal species are listed in [23]. Dry weight was calculated by a regression equation defining the body length and weight of each species (see [23] for references). Because the phytoplankton and ciliates were calculated as fresh biomass, zooplankton dry mass was recalculated according to the index proposed by [34]. All microscopy of phytoplankton, ciliates, and metazooplankton employed a Nikon H550L light microscope at × 40–× 1000.
Toxin Analysis
Microcystin concentrations (analogues: MC-LR, MC-RR, MC-YR) were determined by high-performance liquid chromatography (HPLC) using an Agilent 1100 apparatus with a diode matrix (DAD) in the Central Laboratory of the Municipal Water and Sewage Company in Krakow, Poland [35].
Statistical Analysis
The Mann-Whitney U test was used to ascertain the statistical significance of differences between the artificial ponds and natural oxbow lakes. The factors analyzed included the microcystin concentrations and the population parameters for the protozooplankton (Ciliata), metazooplankton, and particular groups of metazooplankton (Cladocera, Copepoda, Rotifera). Canonical correspondence analysis (CCA; constrained ordination) was applied to analyze the effect of type of waterbody on species composition; the same weight was given to each species in the analysis, regardless of the count of a given species in the samples.
We applied a set of generalized linear models (GLMs) to determine whether the density and biomass of the protozooplankton and metazooplankton depended on the dissolved microcystins, using Poisson error distributions for the density and biomass data from the different plankton groups. GLM residuals were graphically examined to test the model assumptions (residual distribution, independence, homoscedasticity). Finally, we used partial residual plots to visualize significant relationships between the density or biomass of the protozooplankton and metazooplankton and the dissolved microcystins. According to [36], both of the methods we used are good options for spatial modeling of species distributions.
All of our analyzed data were log-transformed. The statistical analyses employed Statistica 12 (descriptive statistics, Mann-Whitney U test), Past 3.10 (box plots), and Canoco 5.04 (CCA, GLM).
Results
Cyanobacterial Blooms and Microcystins
Cyanobacterial blooms were observed in all four waterbodies. The blooms persisted for up to 3 months in the two oxbow lakes (P, T) and for up to 6 months in the two artificial ponds (P1, P2). Cyanobacterial toxins (microcystins) occurred in the water of all studied waterbodies but varied in concentration and duration (Table 1; Fig. 2).
The dissolved microcystin concentrations were highest in the artificial ponds (P1, P2) and varied the most in P2 (Fig. 2a); the concentrations were lower and more uniform in the natural oxbow lakes (P, T) (Fig. 2b–d). The microcystin forms differed in their patterns of occurrence: in the artificial ponds, the highest concentration of MC-LR was found in P1 and the highest concentration MC-RR and MC-YR in P2 (Fig. 2b–d).
The differences in dissolved microcystin concentrations between the natural oxbow lakes and the artificial ponds were statistically significant (for MCtot Mann-Whitney U test, z = − 3.00 and p < 0.000; for MC-LR Mann-Whitney U test, z = − 2.43 and p = 0.015).
Zooplankton Structure
The zooplankton organisms were divided into protozooplankton (Ciliata) and metazooplankton (Cladocera, Copepoda, Rotifera). We recorded 15 Ciliata taxa and 54 metazooplankton taxa (see supplementary data). The average number of Ciliata taxa was lower than the average number of metazoan taxa, but Spearman rank correlations showed a positive relationship between the number of Ciliata taxa and the number of metazooplankton taxa (r = 0.46, p < 0.05).
CCA partially differentiated the protozooplankton of the natural waterbodies (P, T) from that of the artificial ponds (P1, P2) along the first axis based on the species composition of the samples, but those results were not statistically significant (Fig. 3).
CCA plot diagram. Composition of Ciliata assemblages, samples, and waterbodies. The samples are grouped as follows: blue envelope—Piekary oxbow lake (natural reservoir); black envelope—Tyniec oxbow lake (natural reservoir); brown and green envelopes—Podkamycze 1 and 2 (artificial ponds). Total variation = 3.24; explanatory variables account for 4.0%. Eigenvalues for axis 1 = 0.067 and for axis 2 = 0.042. Permutation test results: on first axis pseudo-F = 1.2, P = 0.81; on all axes pseudo-F = 0.8, P = 0.836. Explained fitted variation (cumulative) for axis 1 = 51.94 and axis 2 = 84.33.
CCA of the metazooplankton showed differences in species composition between the natural (P, T) and artificial (P1, P2) waterbodies along the first axis based on the species composition of the samples. Those differences were statistically significant (Fig. 4).
CCA plot diagram. Composition of metazooplankton assemblages, samples, and waterbodies. Samples are grouped as follows: blue envelope—Piekary oxbow lake (natural reservoir); black envelope—Tyniec oxbow lake (natural reservoir); brown and green envelopes—Podkamycze 1 and 2 (artificial ponds). Total variation = 2.74; explanatory variables account for 13.9%. Eigenvalues for axis 1 = 0.207 and for axis 2 = 0.097. Permutation test results: on first axis pseudo-F = 4.9, P = 0.002; on all axes pseudo-F = 3.2, P = 0.002. Explained fitted variation (cumulative) for axis 1 = 54.29 and axis 2 = 79.81
Zooplankton vs. Dissolved Microcystins
GLM showed statistically significant negative relationships between the biomass and the density of several zooplankton groups and the concentrations of MCtot and MC-LR (Tables 2 and 3), but not for MC-RR or MC-YR.
Population Parameters of Proto- and Metazooplankton Assemblages
The richness, total density, and total biomass of Ciliata species in the natural oxbow lakes (P, T), having lower microcystin concentrations, were significantly higher than in the artificial ponds (P1, P2), having higher microcystin concentrations (Fig. 5a–c).
Box plots for a number of species, b total density, and c total biomass of Ciliata in particular waterbodies. Mann-Whitney U test showed statistically significant differences between the natural (P, T) and artificial waterbodies (P1, P2) for all parameters (number of species z = 4.215, p < 0.000; density z = 4.833, p < 0.000; biomass z = 4.472, p < 0.000). Dark horizontal lines represent medians; boxes enclose 25th and 75th percentiles; whiskers represent 5th and 95th percentiles
The richness and density of metazooplankton species were significantly higher in waterbodies that had shorter-duration cyanobacterial blooms and lower microcystin concentrations (Fig. 6a–c), but total metazooplankton biomass did not show such a correlation. The natural and artificial waterbodies differed significantly for biomass of Rotifera (Fig. 6d–f) and Copepoda (Fig. 6g–i), but surprisingly not for biomass of Cladocera (Fig. 6j–l).
Box plots. a Total number of metazooplankton species (Mann-Whitney U test z = 5.001, p < 0.000). b Total density of metazooplankton (Mann-Whitney U test z = 5.235, p < 0.000). c Total biomass of metazooplankton (not statistically significant). d Total number of Rotifera species (Mann-Whitney U z = 2.039, p = 0.041). e Total density of Rotifera (Mann-Whitney U test z = 5.151, p < 0.000). f Total biomass of Rotifera (Mann-Whitney U test z = 4.937, p < 0.000). g Total number of Copepoda species (not statistically significant). h Total density of Copepoda (Mann-Whitney U test z = 3.314, p < 0.000). i Total biomass of Copepoda (Mann-Whitney U test z = 2.364, p = 0.018). j Total number of Cladocera species (Mann-Whitney U test z = 5.077, p < 0.000). k Total density of Cladocera (Mann-Whitney U test z = 3.842, p < 0.000). l Total biomass of Cladocera (not statistically significant). Dark horizontal lines represent medians; boxes enclose 25th and 75th percentiles; whiskers represent 5th and 95th percentiles
Discussion
Microcystins are a group of toxins often present in water, as they are produced by species that commonly occur there (e.g., species of the genera Planktothrix, Microcystis, Aphanizomenon, Nostoc, Anabaena) [37]. In the studied waterbodies, we found three microcystin analogues: MC-YR, MC-RR, and MC-LR. The first two occurred at small concentrations, and for them, we found no significant differences between the waterbodies nor any relationships with plankton parameters. Only dissolved MC-LR was associated with the parameters of the plankton, both protozooplankton (Ciliata) and metazooplankton. Differences in hydrophobicity can make microcystins differ in the way that they are taken up by animals. They may be ingested with food [38] or may bind to membranes and penetrate cells by pinocytosis [16]. The microcystins affected the plankton animals in different ways in the studied waterbodies. We showed that they were more harmful to these organisms in the artificial ponds than in the natural oxbow lakes. There were significant differences in dissolved MC-LR concentration between the natural and artificial waterbodies. MC-LR is known to be the most potent toxin [39]; we infer that the significantly higher and longer-persisting concentrations of that analogue in the artificial ponds shaped the structure of the ciliate and metazooplankton assemblages.
Species-specific adaptations in zooplankton have led to variation of the observed responses to cyanobacteria blooms [40] and cyanobacterial toxins. In the literature, information about the response of ciliates [21, 22, 41, 42], rotifers [43, 44], copepods [20, 45], and cladocerans [46, 47] to cyanotoxins is contradictory and unclear. Our GLM analyses showed significant negative correlations between the dissolved microcystins and both the density and the biomass of Ciliata. Other research indicates that cyanobacterial blooms generally affect communities of ciliates by lowering their diversity: only a few ciliate species were found to develop during the culminating stage of cyanobacterial blooms [48].
The richness, total biomass, and density of Ciliata species in particular samples were significantly lower in the two artificial ponds (P1, P2), where microcystins occurred at significantly higher concentrations and remained in the water longer than in the oxbow lakes (P, T). The composition of Ciliata assemblages in particular samples was more uniform in the ponds and assumed a more typical structure in the oxbow lakes (CCA). That uniformity or homogeneity of Ciliata assemblages in the artificial ponds reflects their longer exposure to dissolved cyanotoxins. The more typical structure of the assemblages found in the oxbow lakes reflects the operation of an ecosystem in which toxins are present at lower concentrations and for a shorter period.
The response of the metazoan assemblages was similar to that of the ciliate assemblages. GLM regression showed negative relationships between dissolved microcystins and both the density and the biomass of the metazooplankton. We found significantly fewer species and lower total density of metazooplankton in the ponds (P1, P2) than in the oxbow lakes (P, T), but surprisingly we did not find significant differences in total biomass.
Since metazooplankton organisms form a heterogeneous group consisting of various subgroups, we also analyzed data from particular groups. We found a significant relationship between microcystins and the density of Rotifera and a decrease in the number of species, total density, and total biomass of rotifers in the ponds, which had higher dissolved microcystin concentrations.
Copepod biomass was also negatively correlated with dissolved microcystin concentration. However, copepods are able to discriminate between toxic and nontoxic cyanobacteria [44], but they can assimilate toxins directly from the water or via ciliates [49, 50], and they may adsorb toxins and then transfer them to higher trophic levels [51]. Analyses of copepod biomass and density showed statistically significant differences between the ponds (P1, P2) and the oxbow lakes (P, T), in line with laboratory studies [45] which showed that an elevated concentration of microcystins reduced the survival of Eurytemora affinis.
The relationship between toxins and Cladocera is even more complicated. It has been demonstrated that Daphnia species may adapt to the presence of toxins [47]. Small cladocerans such as Bosmina may not be sensitive to the effects of microcystins. Bosmina and Daphnia are species that ingest toxic cyanobacteria, leading to microcystin accumulation [52, 53] and transferring them to higher trophic levels [54]. In our study, Cladocera showed significant negative correlations with microcystins, mainly MC-LR. There were significant differences in the total density but not the biomass of Cladocera between the artificial and natural waterbodies: the oxbow lakes showed higher density of Cladocera species but their biomass was higher in the ponds. This suggests that the large cladocerans (Daphnia) in our waterbodies were adapted to higher concentrations of those toxins.
Conclusion
We demonstrated that in waterbodies with higher and longer-persisting microcystin concentrations, various parameters (density, biomass, richness) of the zooplankton population decreased, and the structure of the species assemblages tended toward uniformity. The studied artificial ponds were more exposed to harmful cyanobacterial blooms, and for a longer period, than the natural oxbow lakes. The general problem can be expressed in this way: increasing artificiality of the aquatic environment (transformation, destruction, creation of new waterbodies) + eutrophication + global warming = increased proliferation of toxic cyanobacterial blooms + homogenization of plankton species structure.
References
Paerl HW, Gardner WS, Havens KE, Joyner AR, McCarthy MJ, Newell SE, Qin B, Scott JT (2016) Mitigating cyanobacterial harmful algal blooms in aquatic ecosystems impacted by climate change and anthropogenic nutrients. Harmful Algae 54:213–222
Carmichael WW (2001) Health effects of toxin producing cyanobacteria: the cyano HABs. Hum. Ecol. Risk. Assess. 7:1393–1407
Moustaka-Gouni M, Vardaka E, Michaloudi E, Kosmas KA, Tryfon E, Mihalatou H, Gkelis S, Lanaras T (2006) Plankton food web structure in a eutrophic polymictic lake with a history of toxic cyanobacterial blooms. Limnol. Oceanogr. 51:715–727
Dembowska EA, Napiórkowski P (2015) A case study of the planktonic communities in two hydrologically different oxbow lakes, Vistula River, Central Poland. J. Limnol. 74:2008–2015
Wilk-Woźniak E, Ligęza S, Shubert E (2014) Effect of water quality on phytoplankton structure in oxbow lakes under anthropogenic and non-anthropogenic impacts. Clean Soil Air Water 42:421–427
Goździejewska A, Glińska-Lewczuk K, Obolewski K, Grzybowski M, Kujawa R, Lew S, Grabowska M (2016) Effects of lateral connectivity on zooplankton community structure in floodplain lakes. Hydrobiologia 774:7–21
Wilson AE, Sarnelle O, Tillmanns AR (2006) Effects of cyanobacterial toxicity and morphology on the population growth of freshwater zooplankton: metaanalyses of laboratory experiments. Limnol. Oceanogr. 51:1915–1924
Martin-Creuzburg D, Wacker A, Von Elert E (2005) Life history consequences of sterol availability in the aquatic keystone species Daphnia. Oecologia 144:362–372
Rapala J, Sivonen K, Lyra C, Niemelä SI (1997) Variation of microcystins, cyanobacterial hepatotoxins, in Anabaena spp. as a function of growth stimuli. Appl. Environ. Microbiol. 63:2206–2212
Sivonen K, Bartram J (1999) Toxic cyanobacteria in water: a guide to their public health consequences, monitoring and management. E. and F.N. Spon, New York
Miller MA, Kudela RM, Mekebri A, Crane D, Oates SC, Tinker MT, et al. (2010) Evidence for a novel marine harmful algal bloom: cyanotoxin (microcystin) transfer from land to sea otters. PLoS One 5(9):e12576
Buratti FM, Manganelli M, Vichi S, Stefanelli M, Scardala S, Testai E, Funari E (2017) Cyanotoxins: producing organisms, occurrence, toxicity, mechanism of action and human health toxicological risk evaluation. Arch. Toxicol. 91:1049–1130
Jakubowska N, Szeląg-Wasielewska E (2015) Toxic picoplanktonic cyanobacteria—review. Mar Drugs 13:1497–1518
Campos A, Vasconcelos V (2010) Molecular mechanisms of microcystin toxicity in animal cells. Int. J. Mol. Sci. 11:268–287
Lankoff A, Kolataj A (2001) Influence of microcystin-YR and nodularin on the activity of some proteolytic enzymes in mouse liver. Toxicon 39:419–423
Vesterkvist PS, Meriluoto JA (2003) Interaction between microcystins of different hydrophobicities and lipid monolayers. Toxicon 41:349–355
Karjalainen M, Reinikainen M, Spoof L, Meriluoto JAO, Sivonen K, Markku V (2005) Trophic transfer of cyanobacterial toxins from zooplankton to planktivores: consequences for pike larvae and mysid shrimps. Environ. Toxicol. 20:354–362
Sherr EB, Sherr BF (2002) Significance of predation by protists in aquatic microbial food webs. Antonie Van Leeuwenhoek. Int J Gen Mol Microbiol 81:293–308
Irigoien X, Harris RP, Head RN, Cummings D, Harbour B, Myer-Harms B (2000) Feeding selectivity and egg production of Calanus helgolandicus in the English Channel. Limnol. Oceanogr. 45:44–54
Ger KA, Teh SJ, Baxa DV, Lesmeister S, Goldman CR (2010) The effects of dietary Microcystis aeruginosa and microcystin on the copepods of the upper San Francisco estuary. Freshw. Biol. 55:1548–1559
Ransom RE, Nerad TA, Meier PG (1978) Acute toxicity of some bluegreen algae to the protozoan Paramecium caudatum. J. Phycol. 14:114–116
Zurawell RW, Chen H, Burke JM, Prepas EE (2005) Hepatotoxic cyanobacteria: a review of the biological importance of microcystins in freshwater environments. J Toxicol Environ Health B Crit Rev 8:1–37
Krztoń W, Pudaś K, Pociecha A, Strzesak M, Kosiba J, Walusiak E, Szarek-Gwiazda E, Wilk-Woźniak E (2017) Microcystins affect zooplankton biodiversity in oxbow lakes. Environ. Toxicol. Chem. 36:165–174
Kosiba J, Wilk-Woźniak E, Krztoń W, Strzesak M, Pociecha A, Walusiak E, Pudaś K, Szarek-Gwiazda E (2017) The trophic network in shallow oxbow lakes exposed to cyanobacterial blooms. Microb. Ecol. 73(1):17–28
Carlson RE, Simpson J (1996) A coordinator’s guide to volunteer lake monitoring methods. North American Lake Management Society, Madison
Patalas K (1954) Zespoly skorupiakow pelagicznych 28 jezior pomorskich [Communities of pelagic crustacean of 28 Pomeranian Lakes]. Ekol Pol 2(1):61–88
Kvam OV, Kleiven OT (1995) Diel horizontal migration and swarm formation in Daphnia in response to Chaoborus. Hydrobiologia 307(1–3):177–184
Foissner W, Berger H (1996) A user-friendly guide to the ciliates (Protozoa, Ciliophora) commonly used by hydrobiologists as bioindicators in rivers, lakes, and waste waters, with notes on their ecology. Freshw. Biol. 35:375–482
Foissner W, Berger H, Schaumburg J (1999) Identification and ecology of limnetic plankton ciliates. Informationsberichte des Bayer. Landesamtes für Wasserwirtschaft, München
Jerome CA, Montagnes DJS, Taylor FJR (1993) The effect of the quantitative protargol stain and Lugols and Buinos fixatives on cell size: a more accurate estimate of ciliate species biomass. J Eukryot Microbiol 40:254–259
Menden-Deuer S, Lessard EJ (2000) Carbon to volume relationships for dinoflagellates, diatoms, and other protist plankton. Limnol. Oceanogr. 45:569–579
Wiąckowski K, Doniec A, Fyda J (1994) An empirical study of the effect of fixation on ciliate cell volume. Mar Microb Food Webs 8:59–69
Putt M, Stoecker DK (1989) An experimentally determined carbon: volume ratio for marine “oligotrichous” ciliates from estuarine and coastal waters. Limnol. Oceanogr. 34:1097–1103
Bottrell HH, Duncan A, Gliwicz ZM, Grygierek E, Herzig A, Hillbricht-Ilkowska A, Kurasawa H, Larsson P, Wegleńska T (1976) A review of some problems in zooplankton production studies. Norw J Zool 24:419–456
Meriluoto J, Codd GA (2005) Cyanobacterial monitoring and cyanotoxin analysis. Acta Acad Abo 65:1–145
Guisan A, Weiss SB, Weiss AD (1999) GLM versus CCA spatial modeling of plant species distribution. Plant Ecol. 143:107–122
Van Apeldoorn ME, Van Egmond HP, Speijers GJ, Bakker GJ (2007) Toxins of cyanobacteria. Mol. Nutr. Food Res. 51:7–60
Wiegand C, Pflugmacher S (2005) Ecotoxicological effects of selected cyanobacterial metabolites: a short review. Toxicol. Appl. Pharmacol. 203:201–218
Gupta N, Pant SC, Vijayaraghavan R, Lakshmana Rao PV (2003) Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in mice. Toxicology 188:285–296
Gustafsson S, Hansson L-A (2004) Development of tolerance against toxic cyanobacteria in Daphnia. Aquatic Ecol 38:37–44
Fabbro L, Baker M, Duivenvoorden L, Pegg G, Shiel R (2001) The effects of the ciliate Paramecium cf. caudatum Ehrenberg on toxin producing Cylindrospermopsis isolated from the Fitzroy River. Australia Environ Toxicol 16:489–497
Ward CJ, Codd GA (1999) Comparative toxicity of four microcystins of different hydrophobicities to the protozoan, Tetrahymena pyriformis. J. Appl. Microbiol. 86:874–882
Gilbert JJ (1996) Effect of food availability on the response of planktonic rotifers to a toxic strain of the cyanobacterium Anabaena flos-aquae. Limnol. Oceanogr. 41:1565–1572
Fulton RS, Paerl H (1987) Effects of colonial morphology on zooplankton utilization of algal resources during blue-green algal (Microcystis aeruginosa) blooms. Limnol. Oceanogr. 2:634–644
Reinikainen M, Lindvall F, Meriluoto JAO, Pepka S, Sivonen K, Spoof L, Wahlsten M (2002) Effects of dissolved cyanobacterial toxins on the survival and egg hatching of estuarine calanoid copepods. Mar. Biol. 140:577–583
Hansson LA, Gustafsson S, Rengefors K, Bomark L (2007) Cyanobacterial chemical warfare affects zooplankton community composition. Freshw. Biol. 52:1290–1301
Wojtal-Frankiewicz A, Bernasińska J, Frankiewicz P, Gwoździński K, Jurczak T (2014) Response of Daphnia’s antioxidant systems to spatial heterogeneity in cyanobacteria concentrations in a lowland reservoir. PLoS One 9:e112597
Tirjaková E, Krajčovičová K, Illyová M, Vdačny P (2016) Interaction of ciliate communities with cyanobacterial water bloom in a shallow, hypertrophic reservoir. Acta Protozool. 3:173–188
Karjalainen M, Reinikainen M, Lindvall F, Spoof L, Meriluoto JAO (2003) Uptake and accumulation of dissolved, radiolabeled nodularin in Baltic Sea zooplankton. Environ. Toxicol. 18:52–60
Agasild H, Zingel P, Karus K, Kangro K, Salujoe J, Noges T (2013) Does metazooplankton regulate the ciliate community in a shallow eutrophic lake? Freshw. Biol. 58(1):183–191
Sopanen S, Uronen P, Kuuppo P, Svensen C, Rühl A, Tamminen T, et al. (2009) Transfer of nodularin to the copepod Eurytemora affinis through the microbial food web. Aquat. Microb. Ecol. 55(2):115–130
Watanabe MM, Kaya K, Takamura N (1992) Fate of the toxic cyclic heptapeptides, the microcystins, from blooms of Microcystis(cyanobacteria) in a hypertrophic lake. J. Phycol. 28:761–767
Thostrup L, Christoffersen K (1999) Accumulation of microcystin in Daphnia magna feeding on toxic Microcystis. Arch. Hydrobiol. 145:447–467
Tönno I, Agasild H, Kõiv T, Freiberg R, Noges P, Noges T (2016) Algal diet of small bodied crustacean zooplankton in a cyanobacteria-dominated eutrophic lake. PLoS One 11:e0154526
Acknowledgements
We thank Dr. Edward Walusiak for assistance with sampling, Krzysztof Pudaś for analyses of microcystins, and the anonymous reviewers for helpful comments and suggestions. Michael Jacobs line-edited the manuscript for submission.
Funding Information
This study was supported by the Institute of Nature Conservation, Polish Academy of Sciences, through its statutory fund and a grant for young scientists and Ph.D. candidates.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
ESM 1
(DOC 105 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kosiba, J., Krztoń, W. & Wilk-Woźniak, E. Effect of Microcystins on Proto- and Metazooplankton Is More Evident in Artificial Than in Natural Waterbodies. Microb Ecol 75, 293–302 (2018). https://doi.org/10.1007/s00248-017-1058-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-017-1058-z