Abstract
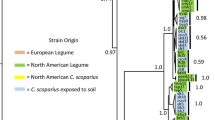
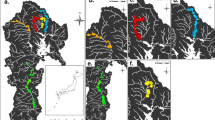
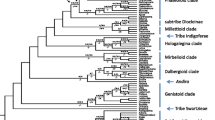
The patterns and drivers of bacterial strain dominance remain poorly understood in natural populations. Here, we cultured 1292 Bradyrhizobium isolates from symbiotic root nodules and the soil root interface of the host plant Acmispon strigosus across a >840-km transect in California. To investigate epidemiology and the potential role of accessory loci as epidemic drivers, isolates were genotyped at two chromosomal loci and were assayed for presence or absence of accessory “symbiosis island” loci that encode capacity to form nodules on hosts. We found that Bradyrhizobium populations were very diverse but dominated by few haplotypes—with a single “epidemic” haplotype constituting nearly 30 % of collected isolates and spreading nearly statewide. In many Bradyrhizobium lineages, we inferred presence and absence of the symbiosis island suggesting recurrent evolutionary gain and or loss of symbiotic capacity. We did not find statistical phylogenetic evidence that the symbiosis island acquisition promotes strain dominance and both symbiotic and non-symbiotic strains exhibited population dominance and spatial spread. Our dataset reveals that a strikingly few Bradyrhizobium genotypes can rapidly spread to dominate a landscape and suggests that these epidemics are not driven by the acquisition of accessory loci as occurs in key human pathogens.


Similar content being viewed by others
References
Bright M, Bulgheresi S (2010) A complex journey: transmission of microbial symbionts. Nat Rev Microbiol 8:218–230. doi:10.1038/nrmicro2262
Sachs JL, Skophammer RG, Bansal N, Stajich JE (2013) Evolutionary origins and diversification of proteobacterial mutualists. Proc R Soc B Biol Sci 281:20132146–20132146. doi:10.1098/rspb.2013.2146
Groisman EA, Ochman H (1996) Pathogenicity islands: bacterial evolution in quantum leaps. Cell 87:791–794. doi:10.1016/S0092-8674(00)81985-6
Jain R, Rivera MC, Moore JE, Lake JA (2003) Horizontal gene transfer accelerates genome innovation and evolution. Mol Biol Evol 20:1598–1602. doi:10.1093/molbev/msg154
Gal-Mor O, Finlay BB (2006) Pathogenicity islands: a molecular toolbox for bacterial virulence. Cell Microbiol 8:1707–1719. doi:10.1111/j.1462-5822.2006.00794.x
Bach S (2000) The Yersinia high-pathogenicity island is present in different members of the family Enterobacteriaceae. FEMS Microbiol Lett 183:289–294. doi:10.1016/S0378-1097(00)00005-7
Diep BA, Gill SR, Chang RF et al (2006) Complete genome sequence of USA300, an epidemic clone of community-acquired meticillin-resistant Staphylococcus aureus. Lancet 367:731–739. doi:10.1016/S0140-6736(06)68231-7
Qiu X, Gurkar AU, Lory S (2006) Interstrain transfer of the large pathogenicity island (PAPI-1) of Pseudomonas aeruginosa. Proc Natl Acad Sci 103:19830–19835. doi:10.1073/pnas.0606810104
Sprent JI (2001) Nodulation in legumes. Royal Botanic Gardens, Kew
Sugawara M, Epstein B, Badgley BD et al (2013) Comparative genomics of the core and accessory genomes of 48 Sinorhizobium strains comprising five genospecies. Genome Biol 14:R17. doi:10.1186/gb-2013-14-2-r17
Cleveland CC, Townsend AR, Schimel DS et al (1999) Global patterns of terrestrial biological nitrogen (N2) fixation in natural ecosystems. Glob Biogeochem Cycles 13:623–645. doi:10.1029/1999GB900014
Graham PH, Vance CP (2003) Legumes: importance and constraints to greater use. Plant Physiol 131:872–877. doi:10.1104/pp. 017004
Uchiumi T, Ohwada T, Itakura M et al (2004) Expression islands clustered on the symbiosis island of the Mesorhizobium loti genome. J Bacteriol 186:2439–2448. doi:10.1128/JB.186.8.2439-2448.2004
Pessi G, Ahrens CH, Rehrauer H et al (2007) Genome-wide transcript analysis of Bradyrhizobium japonicum bacteroids in soybean root nodules. Mol Plant Microbe Interact 20:1353–1363. doi:10.1094/MPMI-20-11-1353
Kaneko T (2000) Complete genome structure of the nitrogen-fixing symbiotic bacterium Mesorhizobium loti. DNA Res 7:331–338. doi:10.1093/dnares/7.6.331
Galibert F (2001) The composite genome of the legume symbiont Sinorhizobium meliloti. Science 293:668–672. doi:10.1126/science.1060966
Kaneko T, Nakamura Y, Sato S et al (2002) Complete genomic sequence of nitrogen-fixing symbiotic bacterium Bradyrhizobium japonicum USDA110. DNA Res 9:189–197. doi:10.1093/dnares/9.6.189
Young JPW, Crossman LC, Johnston AWB et al (2006) The genome of Rhizobium leguminosarum has recognizable core and accessory components. Genome Biol 7:R34. doi:10.1186/gb-2006-7-4-r34
Lee K-B, De Backer P, Aono T et al (2008) The genome of the versatile nitrogen fixer Azorhizobium caulinodans ORS571. BMC Genomics 9:271. doi:10.1186/1471-2164-9-271
Young JPW (1996) Diversity and phylogeny of rhizobia. New Phytol 133:87–94
Perret X, Staehelin C, Broughton WJ (2000) Molecular basis of symbiotic promiscuity. Microbiol Mol Biol Rev 64:180–201. doi:10.1128/MMBR.64.1.180-201.2000
Moulin L, Béna G, Boivin-Masson C, Stępkowski T (2004) Phylogenetic analyses of symbiotic nodulation genes support vertical and lateral gene co-transfer within the Bradyrhizobium genus. Mol Phylogenet Evol 30:720–732. doi:10.1016/S1055-7903(03)00255-0
Segovia L, Piñero D, Palacios R, Martínez-Romero E (1991) Genetic structure of a soil population of Rhizobium leguminosarum. Appl Environ Microbiol 57:426–433
Sachs JL, Ehinger MO, Simms EL (2010) Origins of cheating and loss of symbiosis in wild Bradyrhizobium. J Evol Biol 23:1075–1089. doi:10.1111/j.1420-9101.2010.01980.x
Sullivan JT, Eardly BD, van Berkum P, Ronson CW (1996) Four unnamed species of nonsymbiotic rhizobia isolated from the rhizosphere of Lotus corniculatus. Appl Environ Microbiol 62:2818–2825
Saito A, Mitsui H, Hattori R et al (1998) Slow-growing and oligotrophic soil bacteria phylogenetically close to Bradyrhizobium japonicumin. FEMS Microbiol Ecol 25:277–286. doi:10.1111/j.1574-6941.1998.tb00480.x
Pongsilp N, Teaumroong N, Nuntagij A et al (2002) Genetic structure of indigenous non-nodulating and nodulating populations of Bradyrhizobium in soils from Thailand. Symbiosis 33:39–58
Okubo T, Tsukui T, Maita H et al (2012) Complete genome sequence of Bradyrhizobium sp. S23321: insights into symbiosis evolution in soil oligotrophs. Microbes Environ JSME 27:306–315
Bhatt AS, Freeman SS, Herrera AF et al (2013) Sequence-based discovery of Bradyrhizobium enterica in cord colitis syndrome. N Engl J Med 369:517–528. doi:10.1056/NEJMoa1211115
Chaintreuil C, Giraud E, Prin Y et al (2000) Photosynthetic bradyrhizobia are natural endophytes of the African wild rice Oryza breviligulata. Appl Environ Microbiol 66:5437–5447. doi:10.1128/AEM.66.12.5437-5447.2000
Costello EK, Carlisle EM, Bik EM, et al. (2013) Microbiome assembly across multiple body sites in low-birthweight infants. mBio 4:e00782–13–e00782–13. doi: 10.1128/mBio.00782-13
Hunt KM, Foster JA, Forney LJ et al (2011) Characterization of the diversity and temporal stability of bacterial communities in human milk. PLoS ONE 6, e21313. doi:10.1371/journal.pone.0021313
Sachs JL, Kembel SW, Lau AH, Simms EL (2009) In situ phylogenetic structure and diversity of wild Bradyrhizobium communities. Appl Environ Microbiol 75:4727–4735. doi:10.1128/AEM.00667-09
Vinuesa P, Silva C, Werner D, Martínez-Romero E (2005) Population genetics and phylogenetic inference in bacterial molecular systematics: the roles of migration and recombination in Bradyrhizobium species cohesion and delineation. Mol Phylogenet Evol 34:29–54. doi:10.1016/j.ympev.2004.08.020
Vinuesa P, Rojas-Jimenez K, Contreras-Moreira B et al (2008) Multilocus sequence analysis for assessment of the biogeography and evolutionary genetics of four Bradyrhizobium species that nodulate soybeans on the Asiatic continent. Appl Environ Microbiol 74:6987–6996. doi:10.1128/AEM.00875-08
Azevedo A, Martins-Lopes F, Silla RP, Hungria M (2015) A database for the taxonomic and phylogenetic identification of the genus Bradyrhizobium using multilocus sequence analysis. BMC Genomics 16:S10
VanInsberghe D, Maas KR, Cardenas E et al (2015) Non-symbiotic Bradyrhizobium ecotypes dominate North American forest soils. ISME J. doi:10.1038/ismej.2015.54
Rivas R, Martens M, de Lajudie P, Willems A (2009) Multilocus sequence analysis of the genus Bradyrhizobium. Syst Appl Microbiol 32:101–110. doi:10.1016/j.syapm.2008.12.005
Parker MA (2015) The spread of Bradyrhizobium lineages across host legume clades: from Abarema to Zygia. Microb Ecol. doi:10.1007/s00248-014-0503-5
Göttfert M, Röthlisberger S, Kündig C et al (2001) Potential symbiosis-specific genes uncovered by sequencing a 410-kilobase DNA region of the Bradyrhizobium japonicum chromosome. J Bacteriol 183:1405–1412. doi:10.1128/JB.183.4.1405-1412.2001
Giraud E, Moulin L, Vallenet D et al (2007) Legumes symbioses: absence of Nod genes in photosynthetic bradyrhizobia. Science 316:1307–1312. doi:10.1126/science.1139548
Maddison WP, Maddison DR (2005) MacClade: analysis of phylogeny and character evolution
Sachs JL, Russell JE, Hollowell AC (2011) Evolutionary instability of symbiotic function in Bradyrhizobium japonicum. PLoS ONE 6, e26370. doi:10.1371/journal.pone.0026370
Ehinger M, Mohr TJ, Starcevich JB et al (2014) Specialization-generalization trade-off in a Bradyrhizobium symbiosis with wild legume hosts. BMC Ecol 14:8. doi:10.1186/1472-6785-14-8
Parker M (2000) Divergent Bradyrhizobium symbionts on Tachigali versicolor from Barro Colorado Island, Panama. Syst Appl Microbiol 23:585–90
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. doi:10.1038/nmeth.2109
Guindon S, Dufayard JF, Lefort V et al (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59:307–321. doi:10.1093/sysbio/syq010
Anisimova M, Gascuel O (2006) Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Syst Biol 55:539–552. doi:10.1080/10635150600755453
Shimodaira H, Hasegawa M (1999) Multiple comparisons of log-likelihoods with applications to phylogenetic inference. Mol Biol Evol 16:1114
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. doi:10.1093/bioinformatics/btp187
Mcinnes A (2004) Structure and diversity among rhizobial strains, populations and communities—a review. Soil Biol Biochem 36:1295–1308. doi:10.1016/j.soilbio.2004.04.011
Lewontin RC (1964) The interaction of selection and linkage. I. General considerations; heterotic models. Genetics 49:49–67
Tajima F (1983) Evolutionary relationship of DNA sequences in finite populations. Genetics 105:437–460
Hudson RR, Kaplan NL (1985) Statistical properties of the number of recombination events in the history of a sample of DNA sequences. Genetics 111:147–164
Hudson RR (1987) Estimating the recombination parameter of a finite population model without selection. Genet Res 50:245–250
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Harmon LJ, Weir JT, Brock CD et al (2007) GEIGER: investigating evolutionary radiations. Bioinformatics 24:129–131. doi:10.1093/bioinformatics/btm538
Orme D (2012) The caper package: comparative analysis of phylogenetics and evolution in R
Maddison, W. P., Maddison, D. R. (2011) Mesquite
SAS Institute Inc (1989) JMP. SAS Institute Inc., Cary, NC
Rosenberg MS, Anderson CD (2011) PASSaGE: pattern analysis, spatial statistics and geographic exegesis. Version 2: PASSaGE. Methods Ecol Evol 2:229–232. doi:10.1111/j.2041-210X.2010.00081.x
Hamady M, Lozupone C, Knight R (2010) Fast UniFrac: facilitating high-throughput phylogenetic analyses of microbial communities including analysis of pyrosequencing and PhyloChip data. ISME J 4:17–27. doi:10.1038/ismej.2009.97
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. doi:10.1111/j.1755-0998.2010.02847.x
Silva C, Eguiarte LE, Souza V (1999) Reticulated and epidemic population genetic structure of Rhizobium etli biovar phaseoli in a traditionally managed locality in Mexico. Mol Ecol 8:277–287. doi:10.1046/j.1365-294X.1999.00564.x
Baldwin BG, Goldman DH (2012) The Jepson manual: vascular plants of California, 2nd edn. University of California Press, Berkeley, Calif
Regus JU, Gano KA, Hollowell AC, Sachs JL (2014) Efficiency of partner choice and sanctions in Lotus is not altered by nitrogen fertilization. Proc R Soc B Biol Sci 281:20132587–20132587. doi:10.1098/rspb.2013.2587
Thompson CC, Amaral GR, Campeao M et al (2015) Microbial taxonomy in the post-genomic era: rebuilding from scratch? Arch Microbiol 197:359–370. doi:10.1007/s00203-014-1071-2
Dupuy N, Willems A, Pot B et al (1994) Phenotypic and genotypic characterization of bradyrhizobia nodulating the leguminous tree Acacia albida. Int J Syst Bacteriol 44:461–473. doi:10.1099/00207713-44-3-461
Geurts R (1996) Signal transduction in Rhizobium-induced nodule formation. Plant Physiol 112:447–453. doi:10.1104/pp. 112.2.447
Hollowell AC, Gano KA, Lopez G et al (2015) Native California soils are selective reservoirs for multidrug-resistant bacteria. Environ Microbiol Rep. doi:10.1111/1758-2229.12269
Dobrindt U, Hochhut B, Hentschel U, Hacker J (2004) Genomic islands in pathogenic and environmental microorganisms. Nat Rev Microbiol 2:414–424. doi:10.1038/nrmicro884
Berg G, Smalla K (2009) Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere: plant species, soil type and rhizosphere communities. FEMS Microbiol Ecol 68:1–13. doi:10.1111/j.1574-6941.2009.00654.x
Acknowledgments
The following grants supported this study: to ACH a Herbert Kraft Scholarship and a UC Riverside Graduate Research Mentorship Fellowship and to JLS NSF DEB 0816663 and NSF DEB 1150278.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hollowell, A.C., Regus, J.U., Gano, K.A. et al. Epidemic Spread of Symbiotic and Non-Symbiotic Bradyrhizobium Genotypes Across California. Microb Ecol 71, 700–710 (2016). https://doi.org/10.1007/s00248-015-0685-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-015-0685-5