Abstract
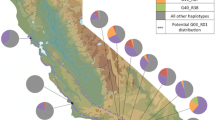
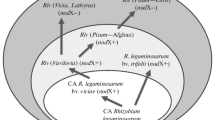
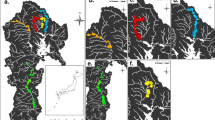
New data from sites in Spain, Sicily and North America establish that the spread of the European legume Cytisus scoparius (Scotch broom) across North America involved the concurrent invasion of a European mobile genetic element (symbiosis island [SI]) into North American Bradyrhizobium nodule bacteria. At four SI loci, bacteria from nine C. scoparius populations across the continent were all identical to haplotypes present in European Bradyrhizobium strains. At seven non-symbiotic (housekeeping) loci, these American C. scoparius bacteria grouped into eight diverse lineages related to, or in some cases identical to, symbionts from several native North American legumes. Inoculation experiments showed that American Bradyrhizobium strains with the European SI variant conferred superior growth to C. scoparius plants compared to strains with SI variants found indigenously in North America. Cytisus scoparius invasion in North America thus enabled proliferation of a European SI variant that spread to multiple indigenous American Bradyrhizobium lineages via horizontal gene transfer. Spread of this SI variant, in turn, likely facilitated C. scoparius invasion of North American habitats.


Similar content being viewed by others
Availability of data and material
All DNA sequences obtained for the project have been made available in GenBank (Supplementary Table S2).
References
Andrews M, De Meyer S, James EK, Stępkowski T, Hodge S, Simon MF, Young JPW (2018) Horizontal transfer of symbiosis genes within and between rhizobial genera: occurrence and importance. Genes 9:321. https://doi.org/10.3390/genes9070321
Avontuur JR, Palmer M, Beukes CW, Chan WY, Coetzee MPA, Blom J, Stepkowski T, Kyrpides NC, Woyke T, Shapiro N, Whitman WB, Venter SN, Steenkamp ET (2019) Genome-informed Bradyrhizobium taxonomy: where to from here? Syst Appl Microbiol 42:427–439. https://doi.org/10.1016/j.syapm.2019.03.006
Boudehouche W, Parker MA, Boulila F (2020) Relationships of Bradyrhizobium strains nodulating three Algerian Genista species. Syst Appl Microbiol 43:126074. https://doi.org/10.1016/j.syapm.2020.126074
Braga RR, Gómez-Aparicio L, Heger T et al (2018) Structuring evidence for invasional meltdown: broad support but with biases and gaps. Biol Invasions 20:923–936. https://doi.org/10.1007/s10530-017-1582-2
Cardinale M, Lanza A, Bonnì ML, Marsala S, Puglia AM, Quatrini P (2008) Diversity of rhizobia nodulating wild shrubs of Sicily and some neighbouring islands. Arch Microbiol 190:461–470. https://doi.org/10.1007/s00203-008-0394-2
Epstein B, Tiffin P (2021) Comparative genomics reveals high rates of horizontal transfer and strong purifying selection on rhizobial symbiosis genes. Proc Roy Soc Lond B 288:20201804. https://doi.org/10.1098/rspb.2020.1804
Horn K, Parker IM, Malek W, Rodríguez-Echeverría S, Parker MA (2014) Disparate origins of Bradyrhizobium symbionts for invasive populations of Cytisus scoparius (Leguminosae) in North America. FEMS Microbiol Ecol 89:89–98. https://doi.org/10.1111/1574-6941.12335
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23:254–267. https://doi.org/10.1093/molbev/msj030
Kalita M, Małek W (2017) Molecular phylogeny of Bradyrhizobium bacteria isolated from root nodules of tribe Genisteae plants growing in southeast Poland. Syst Appl Microbiol 40:482–491. https://doi.org/10.1016/j.syapm.2017.09.001
Kaneko T, Nakamura Y, Sato S et al (2002) Complete genomic sequence of nitrogen-fixing symbiotic bacterium Bradyrhizobium japonicum USDA110. DNA Res 9:189–197
Koppell JH, Parker MA (2012) Phylogenetic clustering of Bradyrhizobium symbionts on legumes indigenous to North America. Microbiol 158:2050–2059. https://doi.org/10.1099/mic.0.059238-0
Lafay B, Burdon JJ (2006) Molecular diversity of rhizobia nodulating the invasive legume Cytisus scoparius in Australia. J Appl Microbiol 100:1228–1238. https://doi.org/10.1111/j.1365-2672.2006.02902.x
Le Roux JJ, Hui C, Keet JH, Ellis AG (2017) Co-introduction vs ecological fitting as pathways to the establishment of effective mutualisms during biological invasions. New Phytol 215:1354–1360. https://doi.org/10.1111/nph.14593
Ormeño-Orrillo E, Martínez-Romero E (2019) A genomotaxonomy view of the Bradyrhizobium genus. Front Microbiol 10:1–13. https://doi.org/10.3389/fmicb.2019.01334
Parker MA (2001) Mutualism as a constraint on invasion success for legumes and rhizobia. Divers Distrib 7:125–136. https://doi.org/10.1046/j.1472-4642.2001.00103.x
Parker MA (2012) Legumes select symbiosis island sequence variants in Bradyrhizobium. Mol Ecol 21:1769–1778. https://doi.org/10.1111/j.1365-294X.2012.05497.x
Peterson DJ, Prasad R (1998) The biology of Canadian weeds. 109. Cytisus scoparius (L.) Link. Can J Plant Sci 78:497–504. https://doi.org/10.4141/P97-079
Rodríguez-Echeverría S (2010) Rhizobial hitchhikers from Down Under: Invasional meltdown in a plant-bacteria mutualism? J Biogeogr 37:1611–1622. https://doi.org/10.1111/j.1365-2699.2010.02284.x
Rodríguez-Echeverría S, Fajardo S, Ruiz-Díez B, Fernáne-Pascual M (2012) Differential effectiveness of novel and old legume-rhizobia mutualisms: implications for invasion by exotic legumes. Oecologia 170:253–261. https://doi.org/10.1007/s00442-012-2299-7
Rodríguez-Echeverría S, Moreno S, Bedmar EJ (2014) Genetic diversity of root nodulating bacteria associated with Retama sphaerocarpa in sites with different soil and environmental conditions. Syst Appl Microbiol 37:305–310. https://doi.org/10.1016/j.syapm.2013.11.003
Ronquist F, Teslenko M, Van Der Mark P et al (2012) MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Simberloff D, Von Holle B (1999) Positive interactions of nonindigenous species: invasional meltdown? Biol Invasions 1:21–32. https://doi.org/10.1007/s10530-017-1582-2
Stępkowski T, Banasiewicz J, Granada CE, Andrews M, Passaglia LMP (2018) Phylogeny and phylogeography of rhizobial symbionts nodulating legumes of the Tribe Genisteae. Genes 9:163. https://doi.org/10.3390/genes9030163
Sullivan JT, Ronson CW (1998) Evolution of rhizobia by acquisition of a 500-kb symbiosis island that integrates into a phe-tRNA gene. Proc Natl Acad Sci USA 95:5145–5149. https://doi.org/10.1073/pnas.95.9.5145
Traveset A, Richardson DM (2014) Mutualistic interactions and biological invasions. Annu Rev Ecol Evol Syst 45:89–113. https://doi.org/10.1146/annurev-ecolsys-120213-091857
Wilkinson HH, Spoerke JM, Parker MA (1996) Divergence in symbiotic compatibility in a legume-Bradyrhizobium mutualism. Evolution 50:1470–1477. https://doi.org/10.1111/j.1558-5646.1996.tb03920.x
Acknowledgements
We thank T. Powell and K. Prior for helpful suggestions on the manuscript, A. Biro for laboratory assistance, I.M. Parker and W. Malek for providing bacterial strains, and A.R. Parker for help with seed collection. Financial support was provided by NSF Grants DEB-0212369 and MCB-0640246.
Funding
Financial support was provided by NSF Grants DEB-0212369 and MCB-0640246.
Author information
Authors and Affiliations
Contributions
All authors collected strains of nodule bacteria for the research. R.F.K. and M.A.P. performed the DNA sequencing and data analysis. R.F.K. wrote the manuscript with support from S.R.-E., P.Q., and M.A.P.
Corresponding author
Ethics declarations
Conflicts of interest
All of the authors declare that they have no conflicts of interest or competing financial, professional, or personal interests that might have influenced the work described in this manuscript.
Consent for publication
All authors give the Publisher permission to publish the work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kruger, R.F., Rodríguez-Echeverría, S., Quatrini, P. et al. Invasional meltdown via horizontal gene transfer of a European symbiosis island variant in North American nodule symbionts of Cytisus scoparius. Biol Invasions 24, 2113–2120 (2022). https://doi.org/10.1007/s10530-022-02776-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-022-02776-9