Abstract
In order to understand the drivers of the distribution and abundance of pelagic seabirds it is necessary to know what they eat, yet there remains little detailed, geo-referenced information on the diets of pelagic seabird. In particular, due to sampling difficulties, information is lacking for non-breeding stages, smaller species, such as storm-petrels, and remote oceanic areas, which may include important diversity hotspots. In this study, we aimed to characterize the trophic ecology of Leach’s storm-petrels foraging in the vicinity of the North Atlantic Current and Evlanov Seamount Marine Protected Area using a combination of survey methods. On a cruise undertaken in June 2017 to characterize the distribution and ecology of seabirds in the region, nineteen Leach’s storm-petrels were caught, sampled and released unharmed. Regurgitations and faecal samples were collected, as well as blood and feather tissues for stable isotope analyses. Of the 12 males, 6 females and 1 unsexed bird, 84% were in active flight feather moult, suggesting they were non-breeders. Prey species, assayed via molecular metabarcoding, were dominated by mesopelagic fish, principally lanternfishes (Myctophidae) and hatchetfishes (Sternoptychidae), but also included cephalopods and crustaceans. Additionally, almost half of the birds had microplastics in their regurgitates. Stable isotopic ratios did not differ significantly between sexes. Our results, combined with those of previous studies, suggest that within the oceanic North Atlantic, Leach’s storm-petrels feed at a similar trophic level to much larger seabirds, targeting mesopelagic fishes that are a key component of the North Atlantic pelagic food web.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seabirds are evidently important members of pelagic ecosystems (Barret et al. 2006, Cury et al. 2011). However, in many cases, their functional roles, such as top-down control and nutrient redistribution (Hunt Jr and McKinnell 2006; Browning et al. 2023), and the limiting effects of the environment on their distributions, remain unclear (Grémillet and Boulinier 2009; Young et al. 2015). This is partly due to a lack of basic information on what they consume (Lewison et al. 2012). In particular, because of sampling difficulties, there are very few data on the diets of non-breeding life-history stages, areas far from land, and smaller species (Barrett et al. 2007; Karnovsky et al. 2012; Rodríguez et al. 2019). Also, the marine environment is undergoing unprecedented changes, including global circulation patterns and physico-chemical properties of the water, pollution, and overexploitation (Greene and Pershing 2007; Herbert-Read et al. 2022). Consequently, it is important to be able to resolve trophic relationships and detect shifts in the marine community dynamics and structure of food webs. To do so requires collection of baseline information on the distribution and ecological significance of predators and prey. In this regard, major oceanic fronts and other hotspots of marine productivity should be surveyed in detail due to their role in supporting complex trophic webs (Mangolte et al. 2023), and thus potential to affect numerous taxa including many high-level predators (Ramírez et al. 2017).
Storm-petrels (family Hydrobatidae and Oceanitidae) are among the most abundant seabirds, but they are particularly challenging to study due to their small size, cavity-nesting, tendency to return to their colonies at night, and wide foraging areas (Warham 1990; Rodriguez et al. 2019). Most direct dietary data come from breeding adults sampled at their colonies (Carreiro et al. 2020; Pollet et al. 2021; Alho et al. 2022). There remains a lack of knowledge of the prey targeted by non-breeding individuals, including immatures, because it is very difficult to sample these stages or even to discriminate them from breeding adults at sea. Non-breeding stages occupy a large proportion of life histories of petrels, so this lack of information hampers assessment of their population responses to changes in the marine environment.
Approximately 6.7–8.3 million pairs of Leach’s storm-petrels (Hydrobates leucorhous) breed in colonies in the North Pacific and North Atlantic, with > 90% of the latter population breeding in colonies bordering the NW Atlantic (Brooke 2004; Pollet et al. 2021). Despite this abundance, they are currently categorised as vulnerable by the IUCN due to a decline of > 30% over the past four decades (BirdLife International 2023). The main causes for these declines, which have principally been noted in the North Atlantic (Newson et al. 2008; Pollet and Shutler 2018; Wilhelm et al. 2019; Deakin et al. 2021), are still debated. Trophic ecology could play a role, however there is a paucity of baseline information on diet, especially during non-breeding periods. Spatial shifts in prey are suspected to contribute to ongoing declines as well, highlighting the need to better understand their diet (Pollet et al. 2023). During breeding, Leach’s storm-petrels from colonies in eastern Canada forage mostly in deep, pelagic waters to the E-SE of the colonies (Pollet et al. 2014; Hedd et al. 2018), whereas after breeding, birds from these populations also make stopovers SE of the Grand Banks before undertaking trans-equatorial migrations to wintering quarters in the South Atlantic (Pollet et al. 2019). Observations of oceanic foraging are consistent with results of traditional diet studies based on the identification of undigested prey items, which indicate that breeding Leach’s storm-petrels tend to feed on mesopelagic lantern fish (Family Myctophidae) and crustaceans (Steele and Montevecchi 1994; Hedd and Montevecchi 2006; Hedd et al. 2009; Frith et al. 2020). In the W Pacific, however, breeding Leach’s storm-petrels shift from taking squid early in the season to fish during chick-rearing, towards the end of summer (Watanuki 1985).
Molecular techniques that rely on the extraction and identification of DNA from digested items in diet samples have improved our ability to detect and identify seabird prey enormously, frequently with high taxonomic detail (Medeiros-Mirra 2010; Waap et al. 2017; Alonso et al. 2018; Carreiro et al. 2020; Alho et al. 2022). As such, they are improving the characterization of trophic interactions in marine communities (Deagle et al. 2009). So far, molecular diet studies on storm-petrels have revealed that these species subsist mostly on mesopelagic fish, but also frequently consume deep-water cephalopods and crustaceans (Wapp 2015, Alho et al. 2020, Carreiro et al. 2020). This is surprising considering that storm-petrels feed by taking prey at or very near the surface of the ocean as they hover and patter on the sea surface (Warham 1990). Although there are no records of diving behaviour in Leach’s storm-petrels (Pollet et al. 2021), dive depths in closely related H. castro tend not to exceed 1 m (Bried 2005). Indeed, it has been suggested that this foraging behaviour explains why storm-petrels ingest small plastic particles relatively frequently (Bond and Lavers 2013). Inferences about the diet of these birds, such as the trophic position and spatial origin of prey, during not only the breeding but also non-breeding periods, can be made by analysing stable isotope ratios in tissues, such as feathers and blood, which are synthesized during different time periods (Hedd and Montevecchi 2006; Pollet et al. 2014; Fairhurst et al. 2015; Halpin et al. 2018). The ratio of the stable isotopes 13C:12C (hereafter δ13C) can be used to infer the oceanic regime from which prey originate (e.g. neritic vs. oceanic), and those of 15N:14N (hereafter δ15N) can be used to infer trophic position (Cherel et al. 2005; Bond and Jones 2009). We expect that these ratios in growing rectrices and in whole blood to reflect diet over the past few days to few weeks, respectively (Hobson 2008).
Here, we aim to use at-sea sampling, stable isotopes and molecular analyses to characterize the diet of Leach’s storm-petrels foraging in the summer in the vicinity of the North Atlantic Current and Evlanov Seamount Marine Protected Area (NACES MPA; Fig. 1). Sampled birds were all thought to be non-breeders, so our study provides the first detailed dietary information in an otherwise very data deficient species, life history stage, and geographical area.
Materials and methods
Study area
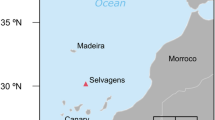
The sampling area was centred on the Sub-Polar Frontal Zone of the oceanic (i.e., off-continental shelf) North Atlantic, in the vicinity of the Charlie Gibbs Fracture Zone of the Mid-Atlantic Ridge (Wakefield 2018; Wakefield et al. 2021) (Fig. 1). The area is characterized by a complex physical oceanography influenced by the convergence of the main NW Atlantic currents (Wakefield et al. 2021). Over recent years, tracking data (Egevang et al. 2010; Hedd et al. 2012; Dias et al. 2013) have revealed that millions of seabirds from disparate breeding populations aggregate in the region. To protect this assemblage, the NACES MPA (Fig. 1) was recently established (Davies et al. 2021). A systematic ship-based survey suggested that seabird distribution in the region was driven ultimately by the effects of physical forcing, mediated via fronts and eddies, on primary and presumably secondary production (Browning et al. 2020, Wakefield et al. 2021).
The study area is shown by the black rectangle. The blue polygon delineates the North Atlantic Current and Evlanov Seamount Marine Protected Area. Individuals were caught between 45º − 50ºN and 30º − 41º W (exact locations provided in the Online Resource 1)
Field data collection
Storm-petrels were caught onboard the RSS Discovery, cruise DY080, between the 14th and 28th of June 2017 (Wakefield 2018). Analysis of data collected during this cruise showed that, in this area, most Leach’s storm-petrels occurred south of the Sub-polar Front, in relatively warm waters (Wakefield et al. 2021). All individuals were caught at night, most (68%) opportunistically after they stranded accidentally at the aft of the ship, presumably having been attracted to or disorientated by the ship´s lights (Miles et al. 2010) (exact location of where the birds were caught provided in the Online Resource 1). The remainder were attracted to the ship by playing calls over a loudspeaker at the bow and were caught either in a mist-net or by hand after landing near the loudspeaker. Upon capture, individuals were ringed and right flight feathers were scored for moult following Alonso et al. (2008) (complete record of birds captured provided in the Online Resource 1). A small blood sample (∼500 µl) was taken for further analysis. Four drops were stored at -80ºC for molecular sexing and the remainder was air dried at 60ºC for subsequent stable isotope analysis (SIA). A distal section of a feather tissue clipped from a growing or new rectrix, when available, was also taken for SIA. Regurgitate samples to study diet were taken by two gastric lavages (Wilson 1984; Neves et al. 2006) and faecal samples were also collected from the same birds, when available. These samples were kept in 100% ethanol and stored at -80ºC until laboratory analyses. After data collection, birds were released immediately or, if fog was present, early the following morning to minimise the risk of the birds colliding with the ship.
Sex determination
Sex was determined using molecular methods adapted from Fridolfsson and Ellegren (1999). In brief, DNA was extracted from the blood samples using a DNeasy Blood and Tissue kit (Qiagen, Manchester, UK). PCR reactions were performed in 10 µl volumes on a MJ research PTC-200 machine, using GoTaq G2 Flexi DNA polymerase (Promega, Madison, WI, USA). Final reaction concentrations were 200 µM dNTP’s, 2 mM MgCl2, 0.4 U GoTaq and 80 ng of the following primers: 2550F (5’ GTT ACT GAT TCG TCT ACG AGA 3’); 2718R (5’ ATT GAA ATG ATC CAG TGC TTG 3’) (Fridolfsson and Ellegren 1999). The thermal profile comprised an initial denaturing step of 94 °C for 2 min, followed by 30 cycles of 48 °C for 60 s, 72 °C for 60 s and 94 °C for 45 s, followed by 48 °C for 60 s and a final extension at 72 °C for 5 min. PCR products were separated in 2% agarose gels in 1X TBE, and visualised via Sybersafe (Invitrogen, Carlsbad, CA, USA) staining.
Microplastics
On the return to the laboratory, regurgitate samples were examined under a stereo microscope (total amplification 20X) for the presence of plastic particles larger than 1 mm. Floating microplastics, all in the form of small pellets, were isolated from the sample with metal tweezers, washed and dried at room temperature, counted and measured by a single observer with a scaled eye piece calibrated with a micrometre slide, and the colour (scored by eye) recorded.
Diet based on regurgitate and faecal samples
DNA isolation
DNA was isolated from regurgitate and faecal samples with a Norgen Stool DNA isolation kit (Norgen Biotek, Canada) following the manufacturer’s protocol. Specifically, the tubes were centrifuged for 1 min at 13,000 rpm and ethanol was removed by aspiration with a micropipette before transferring the solid phase into the bead tubes. Samples were incubated in lysis buffer with gentle vortex for 1–2 h at room temperature before horizontal bead beating in a vortex at full speed. DNA was eluted in 65 µL of pre-heated (70ºC) elution buffer with 30 min of incubation at room temperature. The quality of DNA was assessed by A260/230 and A260/280 ratios measured with a NanoDrop Spectrophotometer. DNA concentration was measured with a Qubit 2.0 dsDNA HS Assay kit (Thermo Fisher Scientific Inc.). DNA samples with low concentrations were concentrated by sample evaporation with a SpeedVac to a final volume of 20 µl. Samples with DNA concentration < 1ng/µl were not processed further.
DNA metabarcoding
DNA metabarcoding library preparation and sequencing were carried out by AllGenetics & Biology SL (www.allgenetics.eu). Libraries were prepared to target the main expected prey groups (fish, cephalopods and crustaceans) with primers previously used to study the diet of other North Atlantic pelagic seabirds (Waap 2015; Waap et al. 2017): For fish, Chord_16S_F (5’ GATCGAGAAGACCCTRTGGAGCT 3’)/Chord_16S_R (5’ GGATTGCGCTGTTATCCCT 3’); for cephalopods, Ceph_16S_F (5’ GACGAGAAGACCCTAWTGAGCT 3’)/ Ceph_16S_R (5’ AAATTACGCTGTTATCCCT) (Deagle et al. 2009); and Crust_16S_F1 (5’ GACGATARGACCCTATAA 3’)/ Crust_16S_R1 (5’ TCTGTTATCCCTARAG 3’) developed specifically for crustaceans (Waap 2015). Amplifications with fish and cephalopod primers were performed in multiplex, in the same reaction. DNA was amplified with a 2-step PCR approach, as in Alho et al. (2022). Details are included in Online Resources. Negative controls were used in every PCR reaction. A blocking primer to prevent host DNA amplification was designed by AllGenetics based on 16 S sequences from GenBank of Pelagodroma marina, Hydrobates leucorhous and Puffinus lherminieri using Geneious 11.1.5 and following Vestheim and Jarman (2008): 5’ CCTGTGGAACTTAAAAATYARCGRCCAC 3’. Libraries were purified with Mag-Bind RXNPure Plus magnetic beads (Omega Biotek), pooled in equimolar amounts, and sequenced in two separate runs: the chordata-cephalopod in 1 GB of NovaSeq PE250 (Illumina), and the Crustacea pool was sequenced in 0.6 GB of an Illumina MiSeq PE300 run. The read output of the two sequencing runs is provided in the Online Resource 2.
Sequence analysis and taxonomic assignment
FASTQ data were processed with the Qiime2-2021.4 pipeline (Bolyen et al. 2019) with the DADA2 plugin (Callahan et al. 2016) to denoise and dereplicate paired-end sequences. Trimming thresholds were set to cut the primers at the 5’ end and low- quality bases at the 3’ end. Reads were truncated at the first base with a Phred quality score ≤ 20. A minimum overlap of 50 bases was set for pairing reads. Sequences were then classified with Qiime2 classify-consensus-vsearch (Bokulich et al. 2018; Rognes et al. 2016) using the 16 S Midori UNIQ-NUC_GB244 database as a reference (Machida el al. 2017), setting 0.8 as the minimum identity and 0.7 as the minimum cover (full list of commands available from BioStudies). Taxa assignment by vsearch were confirmed with online NCBI blastN (https://blast.ncbi.nlm.nih.gov/Blast.cgi). A species was assigned if the Amplicon Sequence Variants (ASVs) had a 98–100% match to the best hit in blastN, otherwise the assignments were adjusted to the least common ancestor when other taxa were assigned with similar identity or if the taxon had no documented occurrence in the North Atlantic. Taxonomic classification of sequence variants was also tested with Qiime classify-consensus-blast as in Waap (2015), which did not outperform the vsearch classification. For both libraries, the resulting sequence variants were collapsed per sample according to taxonomy to construct a table of frequency of occurrence.
Stable isotope analysis
The feather tissues were washed in an ultrasonic bath with deionized water, air dried them for 24 h at 60 °C, rinsed them in 2:1 chloroform: methanol, then air dried them again (Paritte and Kelly 2009). Feathers were then cut into small pieces with scissors. We homogenized whole dried blood in a pestle and mortar. We then weighed 0.7 mg of the respective samples into tin combustion cups and measured stable isotope ratios by continuous flow mass spectrometry using an Elementar (Hanau, Germany) Pyrocube elemental analyser and Thermo (Bremen, Germany) Delta XP mass spectrometer at the NERC National Environmental Isotope Facility (formerly Life Sciences Mass Spectrometry Facility), East Kilbride, U.K. The δ15NAir values are represented in parts per thousand in relation to atmospheric Air, and δ13CVPDB values are in relation to Vienna Pee Dee Belemnite. Differences between sexes on stable isotopic profiles in blood and feather samples were analysed with ANOVA. A Pearson correlation was used to test for a relationship between δ13C and δ15N with latitude and longitude.
Results
Nineteen storm-petrels were captured in the second half of June 2017 - six females, twelve males and one bird whose sex could not be determined. Sixteen individuals were in active tail feather moult which was progressing descendantly (R6 to R1) (Online Resource 1, Fig. 2). In three individuals none of the tail feathers had been moulted (score 0), and six individuals already had new outer tail feathers (score 5). In contrast, while eleven birds had initiated primary moult, most were just in beginning as they were missing only P1 (Online Resource 1, Fig. 2). As most individuals were moulting, all sampled individuals were considered non-breeders (see Discussion).
Microplastic ingestion
Of the sixteen birds from which regurgitate samples were obtained, 50% contained small plastic pellets (diameter 1–3 mm). The mean number of pellets per individual was 2.3 (± 3.7), range 0–13. Lighter colours were prevalent with seven individuals regurgitating at least one white particle, and three also regurgitating yellow or grey particles. Of the total 35 plastic particles found, only 5 were dark coloured, these being dark blue, red and orange. There was no difference in plastic occurrence between sexes (Chi-square test, χ2 (1,16) = 0.285, P > 0.05).
Diet
Denoising of the Fish/Cephalopod library retrieved 33 different sequence variants, which were classified as fish (n = 24 sequences) and cephalopods (n = 4). Non-prey (n = 4) or unclassified sequences (n = 1) were discarded from further analyses. Denoising of the Crustacean library retrieved 26 sequence variants, all subsequently classified as the target group in four of the six samples that originally amplified with the crustacean primers (Online Resource 2).
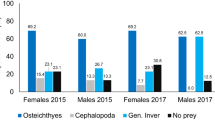
Prey species were successfully detected in 11 regurgitates and 4 faecal samples (no ASVs were retained for two faecal samples after denoising). Two faecal and five regurgitate samples were initially discarded because they did not have sufficient starting material for DNA extraction. Fish, cephalopods and crustacean species were all detected in regurgitates whereas only fish species were detected in faecal samples (Table 1). The average number of species per regurgitate was 3.1, varying between one (n = 1, containing one cephalopod) and five (n = 2, both containing fish and crustaceans). The number of species varied between one and three in faecal samples. Only once was a prey species detected in the faecal sample but not in the regurgitate sample from the same individual. Almost all regurgitate samples contained fish (frequency of occurrence (FO) = 91%, Table 1) whereas cephalopods were much rarer (FO = 18%). Myctophidae (lanternfishes) and Sternoptychidae (hatchetfishes) were the most frequently occurring families (FO = 73%), the former represented by four species and the latter by three (Table 1; Fig. 2). The most frequently occurring species (FO = 73%) was the Glacier lantern fish (Benthosema glaciale). Only two cephalopods species (Gonatus fabricii and Histioteuthis reversa) from two families were identified. In nine of the 15 samples, amplification with the crustacean primers failed but it was unclear if this was due to an absence of crustaceans or technical failure of the reaction. Of the six samples successfully assayed with the crustacean primers, only four (67%) were considered to contain crustaceans. These were from four families of two orders, including Oplophoridae represented by Acantephyra sp. (slightly higher homology with A. pelagica) which was the most frequently detected family. All taxa identified to the species level occur in the mesopelagic zone and perform diel vertical migrations, approaching the surface at night (Table 1; Fig. 2).
Frequency of occurrence (%) of prey families in the diet of Leach’s storm-petrels revealed by DNA metabarcoding. Bar colours represent the main oceanic depth zones where the different families occur according to FishBase (www.fishbase.org). M and F indicate whether the family was found in the diet of males or females, respectively
Tissue stable isotopes
In storm-petrel whole blood samples, values of δ15N varied between 11.4 and 12.8 ‰, and between − 19.3 and − 19.8 ‰ for δ13C. In feathers, isotopic values varied slightly more, ranging between 10.0 and 13.3 ‰ for δ15N and − 18.7 and − 19.4 ‰ for δ13C (Fig. 3). Stable isotopes ratio values did not differ significantly between sexes, for either whole blood: δ15N (F1,16 = 2.15, P > 0.05) or δ13C (F1,16=3.05, P > 0.05), or feathers: δ15N (F1,15 = 0.38, P > 0.05) or δ13C (F1,15=0.08, P > 0.05), suggesting no large differences in resource use between males and females. In blood, there was a positive correlation between δ13C and latitude (r = 0.59, P = 0.012, n = 17) but no correlation between δ15N and latitude.
Discussion
This study is the first to characterize the diet of one of the most commonly occurring seabirds in the highly productive sub-polar frontal zone of the North Atlantic (Boertmann et al. 2011, Wakefield et al. 2021), an area recently accorded international protection due to its importance to seabirds and other higher predators (Davies et al. 2021). Despite its relevance, there is almost no direct information regarding the diet of seabirds in the region. Although based on a small sample taken over a short period, our results suggest that within this area, Leach’s storm-petrels, and possibly other species, may subsist largely on mesopelagic fish, highlighting the importance of this group in the pelagic food web of the North Atlantic. Moreover, our results, along those of previous diet, tracking, and at-sea studies (Watanuki 1985; Vermeer and Devito 1988; Steele and Montevecchi 1994; Hedd and Montevecchi 2006; Hedd 2009, Pollet et al. 2014; Hedd et al. 2018) suggest that Leach’s storm-petrels are specialists of the shelf-break to oceanic zones.
Flight feather moult is energy demanding and so most small Procellariiformes tend to not moult large flight feathers during the breeding season to avoid competing energy requirements (Bridge 2006). Sampling in our study occurred during the second half of June, a time when breeders in the Atlantic colonies are incubating eggs (Money et al. 2008; Pollet et al. 2021). Successful breeders from these populations start flight feather moult in October, with most feathers being replaced in the non-breeding areas (Cramp and Simmons 1977; Warham 1990; Pollet et al. 2021). Most storm-petrels sampled in this study were in active flight feather moult, hence they were most likely immature or failed breeders (Boertmann et al. 2011, Wakefield et al. 2021). This is supported by tracking, which shows that the core area used by breeding adults from the nearest colonies (in Newfoundland), extends eastwards to around 42° W (Hedd et al. 2018). In contrast, the birds in our study were caught between 30.2 and 40.6ºW, with the majority being caught east of 36.5ºW. Tracking also shows that post-breeding adults from North America colonies stopover in the latter area (Pollet et al. 2019). Presumably, non- and post-breeding Leach’s petrels and other seabirds are attracted to this area by its abundant and diverse mesopelagic community (Cook et al. 2013; Sutton et al. 2013, 2017).
In accordance with the oceanic regime in which they were captured, Leach’s storm-petrels fed mainly on mesopelagic species, particularly fishes of the Orders Myctophiformes and Stomiiformes, which are among the most abundant mesopelagic fishes of the North Atlantic (Sutton et al. 2013; Carmo et al. 2015). These results are consistent with other studies showing that Leach’s and other storm-petrels, despite feeding by seizing prey from the surface of the ocean, mostly consume deep water prey, particularly fish (Hedd and Montevecchi 2006, 2009, Carreiro et al. 2020; Alho et al. 2022). The frequent capture of mesopelagic and bathypelagic species in epipelagic waters (above 200 m) along the Mid-Atlantic Ridge during trawling surveys suggests that many taxa in the area undertake diel vertical migrations (Cook et al. 2013). This is a key feature of pelagic ecosystems for top predators that do not dive or are shallow divers, such as most petrels, because prey species, including fish, cephalopods and crustaceans, become available when they ascend to shallower depths at night (Cook et al. 2013).
Lanternfishes (family Myctophidae) are among the most abundant fishes from the mesopelagic zone (Catul et al. 2011; Irigoien et al. 2014) and play a key role in marine pelagic food webs by transferring energy from zooplankton to higher trophic levels (Cherel et al. 2010; Watanuki and Thiebot 2018). Benthosema glaciale, which is widespread in the North Atlantic and Mediterranean (Knutsen et al. 2023), was not only the myctophid most frequently found in the diet of non-breeding Leach’s storm-petrels, but also in the diets of breeding adults and their offspring from Baccalieu Island, Newfoundland (Hedd and Montevecchi 2006). Although it may not be the most abundant (numerically or in biomass) deep-water fish species recorded in the North Atlantic upper water column, it has a clear pattern of diel vertical migration (Cook et al. 2013; Sutton et al. 2013), possibly making it the most abundant species available to surface-feeding storm-petrels. This supports the previous suggestion that storm-petrels feed on the most locally abundant prey (Medeiros-Mirra 2010; Carreiro et al. 2020). Hatchetfishes (genus Maurolicus) which also occurred frequently in the diet of the birds in our study, also undertake vertical migration (Cook et al. 2013; Christiansen et al. 2019). It is known from analysis of stomach contents of freshly dead birds (Spear and Ainley 2007) that Leach’s storm-petrels mostly feed nocturnally, perhaps during twilight hours. Indeed, combined activity and light-level geolocation loggers have shown that Leach’s storm-petrels are among the most nocturnal seabirds, at least during the non-breeding period (Bonnet-Lebrun et al. 2021). Together, these lines of evidence indicate a reliance on vertically migrating mesopelagic prey, captured at night or during the crepuscular periods.
Although not common, we also detected cephalopods in the diet of the storm-petrels, including Histioteuthis reversa, a deep-water species frequently recorded in western North Atlantic surveys (Vecchoine and Pohle 2002). Crustaceans have been found to be an important prey group of breeding Leach’s (Hedd et al. 2009), British (Medeiros-Mirra et al. 2010) and Madeiran storm-petrels (Waap 2015), but we amplified crustaceans in only 27% of the samples (see also Alho et al. 2022). It is unclear whether this is representative of the true diet of non-breeding Leach’s storm-petrels or whether it was due to a methodological failure in, for example, DNA isolation and/or amplification. The fact that one of the samples that did not amplify contained a macroscopic, unidentified mysid suggests a problem with the optimization of the primers used to build the crustacean library (Waap 2015). However, the blood and feather δ15N values observed in birds in our study, which are consistent with those reported for Leach’s storm-petrels in other North Atlantic studies (Hedd and Montevecchi 2006; Pollet et al. 2014; Fairhurst et al. 2015), indicate a diet dominated by relatively high trophic-level prey (e.g., fish), rather than crustaceans (wintering birds in the Pacific have more variable δ15N values (Halpin et al. 2018), possibly reflecting a wider dietary trophic range). Moreover, blood stable isotope profiles are comparable to those of other petrels feeding mostly on mesopelagic fish and squid south of the mid-Atlantic sub-polar frontal system (Fig. 4), suggesting that, despite their size, storm-petrels feed at similar trophic levels to much larger pelagic seabirds in the oceanic North Atlantic. Hence, as in other storm-petrels, mesopelagic fish, which are secondary to tertiary consumers in pelagic ecosystems (Cherel et al. 2010), are a key component of storm-petrel diet.
Stable isotope ratios of nitrogen (δ15N) and carbon (δ13C) (mean ± SD) in blood of pelagic seabirds feeding south of the Sub-polar frontal zone of the North Atlantic during the boreal summer. Bulwer’s petrels (Bulweria bulwerii, n = 15 from Desertas Is.), Madeiran Storm-petrels (Hydrobates castro, n = 16 from Desertas Is.) (Online Resource 1), Zino’s Petrels (Pterodroma madeira, n = 17) and Desertas Petrels (Pterodroma deserta, n = 17) (Ventura et al. 2024) were all sampled between June and September 2019. Cory’s Shearwater (Calonectris borealis) from Ceia et al. (2018)
It remains unclear the extent to which current declines in Leach’s storm petrels relate to diet. Potential hypotheses for declines mediated by diet include the effect of anthropogenic contaminants and shifts in prey abundance or distribution (Pollet et al. 2021). In either case, our data point to the prey taxa that should be considered to investigate these hypotheses further.
Another potential threat which our results have a bearing on is plastics. Plastic ingestion by Leach’s storm-petrels has been recorded in the Atlantic and Pacific oceans since the 1970s (Bond and Lavers 2013). These studies found variable frequencies of occurrence, ranging between 6.5% (Watanuki 1985) and 59% (Furness 1985), possibly related to the foraging areas of the populations sampled. More recently, Bond and Lavers (2013) found that ∼50% of the individuals sampled from a colony in Newfoundland had at least one piece of plastic debris in their gastrointestinal tract, and an average number per bird similar to what we found. Furthermore, if we had considered plastic particles of smaller size, we might have found an even greater contamination level. Unfortunately, many pelagic seabirds are at risk of plastic exposure (Clark et al. 2023), and the smaller species, such as storm-petrels, incur disproportionately greater risks of plastic ingestion (Senko et al. 2020). In petrels, adults likely offload plastic particles primarily when regurgitating to feed chicks (Rodríguez et al. 2012), so immatures or non-breeders may suffer additional impacts due to potential higher retention rates and increased exposure to damage. The preponderance of lighter-coloured microplastic particles suggests these might be more easily detected at the surface of the water, particularly during the dusk or darkness, when Leach’s storm petrels forage.
Metagenomics is becoming an extremely efficient tool to characterize the diet of top predators, particularly in aquatic ecosystems, often resolving a high level of taxonomic detail from very small amounts of sample material (Sousa et al. 2019). However, technical improvements are needed to guarantee that all prey groups are amplified with equal probability. Moreover, a better understanding of the relationship between the number of reads per prey taxa and their relative abundance in the consumer’s diet would greatly improve our ability to characterise trophic relationships and understand ecosystem dynamics. The recovery of DNA from faecal samples, particularly those of pelagic seabirds, needs to be improved, including sample collection, storage and subsequent extraction (McInnes et al. 2017). Although the collection of faecal samples can be much less invasive than stomach contents, in the present study regurgitates allowed the identification of a higher number of prey species, likely due to the recovery of DNA of better quality and quantity.
This study and similar ones (e.g., Powers et al. 2020) show that non-lethal at-sea sampling has the potential to provide large amounts of information on the diet of pelagic seabirds during the non-breeding stages which occupy most of their life histories. Characterizing the drivers of pelagic seabird distribution, particularly during less well-known periods of their annual cycle, is critical to identify and manage responses to predicted oceanic environmental changes, particularly those likely to affect the distribution and abundance of preferred prey.
Data availability
The datasets generated and analysed during the current study will be available in BioStudies (https://www.ebi.ac.uk/biostudies/), accession number S-BSST1220, or from the authors upon request.
References
Alho M, Catry P, Silva MC, Nunes V, Granadeiro JP (2022) Revealing the foraging movements and diet of the White-faced storm Petrel Pelagodroma marina in the Atlantic. Mar Biol 169:91. https://doi.org/10.1007/s00227-022-04078-z
Alonso H, Matias R, Granadeiro JP, Catry P (2008) Moult strategies of Cory’s Shearwaters Calonectris diomedea borealis: the influence of colony location, sex and individual breeding status. J Ornith 150:329–337. https://doi.org/10.1007/s10336-008-0354-2
Alonso H, Granadeiro JP, Waap S, Xavier J, Symondson WOC et al (2018) An holistic ecological analysis of the diet of Cory’s shearwaters using prey morphological characters and DNA barcoding. Mol Ecol 23:3719–3733. https://doi.org/10.1111/mec.12785
Barrett RT, Chapdelaine G, Anker-Nilssen T, Mosbech A, Montevecchi WA et al (2006) Seabird numbers and prey consumption in the North Atlantic. Ices J Mar Sci 63:1145–1158. https://doi.org/10.1093/icesjms/fsm152
Barrett RT, Camphuysen K, Anker-Nilssen T, Chardine JW, Furness RW et al (2007) Diet studies of seabirds: a review and recommendations. Ices J Mar Sci 64:1675–1691
BirdLife International (2023) Species factsheet: Hydrobates leucorhous. Downloaded from http://datazone.birdlife.org/species/factsheet/leachs-storm-petrel-hydrobates-leucorhous on 06/09/2023
Boertmann D (2011) Seabirds in the Central, North Atlantic, September 2006: further evidence for an oceanic seabird aggregation area. Mar Ornithol 39:183–188
Bokulich NA, Kaehler BD, Rideout JR, Dillon M, Boylen E et al (2018) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90. https://doi.org/10.1186/s40168-018-0470-z
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Bond AL, Jones IL (2009) A practical introduction to stable-isotope analysis for seabird biologists: approaches, cautions and caveats. Mar Ornithol 37:183–188
Bond AL, Lavers JL (2013) Effectiveness of emetics to study plastic ingestion by Leach’s storm-petrel (Hydrobates Leucorhoa). Mar Pollut Bull 70:171–175. https://doi.org/10.1016/j.marpolbul.2013.02.030
Bonnet-Lebrun A-S, Dias MP, Phillips RA, Granadeiro JP, Brooke ML et al (2021) Seabird Migration Strategies: Flight budgets, Diel activity patterns and Lunar Influence. Front Mar Sci 8:683071. https://doi.org/10.3389/fmars.2021.683071
Bridge ES (2006) Influences of morphology and behaviour on wing moult strategies in seabirds. Mar Ornithol 34:7–19
Bried J (2005) The diving ability of the Madeiran Storm Petrel. Waterbirds 28:162–166. http://www.jstor.org/stable/4132522
Brooke M (2004) Albatrosses and Petrels across the World. Oxford University Press, Oxford
Browning TJ, Al-Hashem AA, Hopwood MJ, Engel A, Wakefield ED et al (2020) Nutrient regulation of late spring phytoplankton blooms in the mid-latitude North Atlantic. Limnol Oceanog 65:1136–1148. https://doi.org/10.1002/lno.11376
Browning TJ, Al-Hashem AA, Achterberg EP, Carvalho PC, Catry P et al (2023) The role of seabird guano in maintaining North Atlantic summertime productivity. Sci Total Environ 897:165309. https://doi.org/10.1016/j.scitotenv.2023.165309
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJA et al (2016) Dada2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13(7):581. https://doi.org/10.1038/nmeth.3869
Carmo V, Sutton T, Menezes G, Falkenhaug T, Bergstad OA (2015) Feeding ecology of the Stomiiformes (Pisces) of the northern Mid-atlantic Ridge. 1. The Sternoptychidae and Phosichthyidae. Prog Oceanogr 130:172–187. https://doi.org/10.1016/j.pocean.2014.11.003
Carreiro AR, Paiva VH, Medeiros R, Franklin KA, Oliveira N et al (2020) Metabarcoding, stable isotopes and tracking: unravelling the trophic ecology of a winter breeding storm petrel (Hydrobates castro) with a multi-method approach. Mar Biol 167:14. https://doi.org/10.1007/s00227-019-3626-x
Catul V, Gauns M, Karuppasamy PK (2011) A review on mesopelagic fishes belonging to family Myctophidae. Rev Fish Biol 21:339–354. https://doi.org/10.1007/s11160-010-9176-4
Ceia FR, Cherel Y, Paiva VH, Ramos JA (2018) Stable Isotope Dynamics (d13C and d15N) in Neritic and Oceanic Waters of the North Atlantic inferred from GPS-Tracked Cory’s shearwaters. Front Mar Sci 5:377. https://doi.org/10.3389/fmars.2018.00377
Cherel Y, Hobson KA, Weimerskirch H (2005) Using stable isotopes to study resource acquisition and allocation in procellariiform seabirds. Oecologia 145:533–540
Cherel Y, Fontaine C, Richard P, Labat J-P (2010) Isotopic niches and trophic levels of myctophid fishes and their predators in the Southern Ocean. Limnol Oceanogr 55:324–332. https://doi.org/10.1007/s00442-005-0156-7
Christiansen S, Titelman J, Kaartvedt S (2019) Night-time swimming behavior of a mesopelagic fish. Front Mar Sci 6:787. https://doi.org/10.3389/fmars.2019.00787
Clark B, Carneiro APB, Pearmain EJRouyer M-M, Clay TA et al (2023) Global assessment of marine plastic exposure risk for oceanic birds. Nat Commun 14:3665. https://doi.org/10.1038/s41467-023-38900-z
Cook AB, Sutton TT, Galbraith JK, Vecchione M (2013) Deep-pelagic (0-3000m) fish assemblage structure over the Mid-atlantic Ridge in the area of the Charlie-Gibbs Fracture Zone. Deep-Sea Res II 98:279–291. https://doi.org/10.1016/j.dsr2.2012.09.003
Cramp S, Simmons KEL (1977) Handbook of the birds of Europe, the Middle East and North Africa: the birds of the Western Palearctic. Ostrich to ducks, vol 1. Oxford University Press, Oxford, London and New York
Cury PM, Boyd IL, Bonhommeau S, Anker-Nilssen T, Crawford RJM et al (2011) Global seabird response to forage fish depletion—one-third for the birds. Science 334:1703–1706. https://doi.org/10.1126/science.1212928
Davies TE, Carneiro APB, Tarzia M, Wakefield E, Hennicke JC et al (2021) Multispecies tracking reveals a major seabird hotspot in the North Atlantic. Conserv Lett 14:1–14. https://doi.org/10.1111/conl.12824
Deagle BE, Kirkwood R, Jarman SN (2009) Analysis of Australian fur seal diet by pyrosequencing prey DNA in faeces. Mol Ecol 18:2022–2038. https://doi.org/10.1111/j.1365-294X.2009.04158.x
Deakin Z, Hansen ES, Luxmoore R, Thomas RJ, Wood MJ et al (2021) Decline of Leach’s storm petrels hydrobates leucorhous at the largest colonies in the northeast Atlantic. Seabird 33:74–106. http://www.seabirdgroup.org.uk/seabird-33-74
Dias MP, Granadeiro JP, Catry P (2013) Individual variability in the migratory path and stopovers of a long-distance pelagic migrant. Ani Behav 86:359–364. https://doi.org/10.1016/j.anbehav.2013.05.026
Egevang C, Stenhouse IJ, Phillips RA, Petersen A, Fox JW et al (2010) Tracking of Arctic terns Sterna paradisaea reveals longest animal migration. Proc Natl Acad Sci 107:2078–2081. https://doi.org/10.1073/pnas.0909493107
Fairhurst GD, Bond AL, Hobson KA, Ronconi RA (2015) Feather-based measures of stable isotopes and corticosterone reveal a relationship between trophic position and physiology in a pelagic seabird over a 153-year period. Ibis 157:273–283. https://doi.org/10.1111/ibi.12232
Fridolfsson A-K, Ellegren H (1999) A simple and universal method for Molecular Sexing of Non-ratite Birds. J Avian Biol 30:116–121. https://doi.org/10.2307/3677252
Frith R, Krug D, Ronconi R, Wong SNP, Mallory M et al (2020) Diet of Leach’s storm-petrels (Hydrobates Leucorhous) among three colonies in Atlantic Canada. Northeast Nat 27(4):612–630. https://doi.org/10.1656/045.027.0402
Furness RW (1985) Plastic at Scottish colonies Mar Pol Bull 6: 103–106. https://doi.org/10.1016/0025-326X(85)90531-4
Greene CH, Pershing AJ (2007) Climate Drives Sea Change. Science 315:1084–1085
Grémillet D, Boulinier T (2009) Spatial ecology and conservation of seabirds facing global climate change: a review. Mar Ecol Prog Ser 391:121–137. https://doi.org/10.3354/meps08212
Halpin LR, Pollet IL, Lee C, Morgan KH, Carter HR (2018) Year-round movements of sympatric fork-tailed (Oceanodroma furcata) and Leach’s (O. Leucorhoa) storm-petrels. J Field Ornith 89:207–220. https://doi.org/10.1111/jofo.12255
Hedd A, Montevecchi WA (2006) Diet and trophic position of Leach’s storm-petrel Oceanodroma leucorhoa during breeding and moult, inferred from stable isotope analysis of feathers. Mar Ecol Prog Ser 322:291–301. https://doi.org/10.3354/meps322291
Hedd A, Montevecchi WA, Davoren GK, Fifield DA (2009) Diet and distributions of Leach’s storm-petrel (Oceanodroma leucorhoa) before and after an ecosystem shift in the Northwest Atlantic. Can J Zool 87:787–801. https://doi.org/10.1139/Z09-060
Hedd A, Montevecchi WA, Otley H, Phillips RA, Fifield DA (2012) Trans-equatorial migration and habitat use by sooty shearwaters Puffinus griseus from the South Atlantic during the nonbreeding season. Mar Ecol Prog Ser 449:277–290. https://doi.org/10.3354/meps09538
Hedd A, Pollet IL, Mauck RA, Burke CM Mallory ML et al. (2018) Foraging areas, offshore habitat use, and colony overlap by incubating Leach’s storm-petrels Oceanodroma leucorhoa in the Northwest Atlantic. PLoS ONE 13:e0194389. https://doi.org/10.1371/journal.pone.0194389
Herbert-Read JE, Thornton A, Amon DJ, Birchenough SNR, Côté IM et al (2022) A global horizon scan of issues impacting marine and coastal biodiversity conservation. Nat Ecol Evol 1–9. https://doi.org/10.1038/s41559-022-01812-0
Hobson KA (2008) Applying isotopic methods to tracking animal movements. In: Hobson KA, Wassenaar LI (eds) Tracking animal migration with stable isotopes. Academic, London. https://doi.org/10.1016/S1936-7961(07)00003-6
Hunt GL Jr, McKinnell S (2006) Interplay between top-down, bottom-up, and wasp-waist control in marine ecosystems. Prog Oceanogr 68:115–124. https://doi.org/10.1016/j.pocean.2006.02.008
Irigoien X, Klevjer TA, Rostad A, Martinez U, Boyra G et al (2014) Large mesopelagic fishes biomass and trophic efficiency in the open ocean. Nat Commun 5:3271. https://doi.org/10.1038/ncomms4271
Karnovsky NJ, Hobson KA, Iverson SJ (2012) From lavage to lipids: estimating diets of seabirds. Mar Ecol Prog Ser 451:263–284. http://www.jstor.org/stable/24875935
Knutsen T, Strand E, Klevjer TA, Salvanes AGV, Broms C al (2023) Feeding ecology of Benthosema glaciale across the North Atlantic. Front Mar Sci 10:1086607. https://doi.org/10.3389/fmars.2023.1086607
Lewison R, Oro D, Godley BJ, Underhill L, Bearhop S et al (2012) Research priorities for seabirds: improving conservation and management in the 21st century. Endanger Species Res 17:93. https://doi.org/10.3354/esr00419
Machida RJ, Leray M, Ho SL, Knowlton N (2017) Metazoan mitochondrial gene sequence reference datasets for taxonomic assignment of environmental samples. Scient Data 4:170027. https://doi.org/10.1038/sdata.2017.27
Mangolte I, Lévy M, Clément H, Ohman MD (2023) Sub-frontal niches of plankton communities driven by transport and trophic interactions at ocean fronts. Biogeosciences 20:3273–3299. https://doi.org/10.5194/bg-20-3273-2023
McInnes JC, Alderman R, Deagle BE, Lea MA, Raymond B et al (2017) Optimised scat collection protocols for dietary DNA metabarcoding in vertebrates. Meth Ecol Evol 8:192–202. https://doi.org/10.1111/2041-210X.12677
Medeiros-Mirra R (2010) The migration strategy, diet and foraging ecology of a small seabird in a changing environment. Cardiff University, Cardiff
Miles W, Money S, Luxmoore R, Furness RW (2010) Effects of artificial lights and moonlight on petrels at St Kilda. Bird Study 57:244–251. https://doi.org/10.1080/00063651003605064
Money S, Sohle I, Parsons M (2008) A pilot study of the phenology and breeding success of Leach’s storm-petrel Oceanodroma leucorhoa on St. Kilda, Western Isles. Seabird 21:98–101
Neves VC, Bolton M, Monteiro LR (2006) Validation of the water offloading technique for diet assessment: an experimental study with Cory’s shearwaters (Calonectris diomedea). J Ornith 147:474–478. https://doi.org/10.1007/s10336-005-0040-6
Newson SE, Mitchell PI, Parsons M, O’Brien SH, Austin GE et al (2008) Population decline of Leach’s storm-petrel Oceanodroma leucorhoa within the largest colony in Britain and Ireland. Seabird 21:77–84
Paritte JM, Kelly JF (2009) Effect of cleaning Regime on stable-isotope ratios of feathers in Japanese quail (Coturnix japonica). Auk 126:165–174. https://doi.org/10.1525/auk.2009.07187
Pollet IL, Shutler D (2018) Leach’s storm Petrel Oceanodroma leucorhoa population trends on Bon Portage Island, Canada. Seabird 31:75–83. https://doi.org/10.61350/sbj.31.75
Pollet I, Ronconi RA, Jonsen ID, Leonard ML, Taylor PD, Shutler D (2014) Foraging movements of Leach’s storm-petrels Oceanodroma leucorhoa during incubation. J Avian Biol 45:305–314. https://doi.org/10.1111/jav.00361
Pollet IL, Ronconi RA, Leonard ML, Shutler D (2019) Migration routes and stopover areas of Leach’s storm-petrels Oceanodroma leucorhoa. Mar Ornith 47:55–65
Pollet IL, Bond AL, Hedd A, Huntington CE, Butler RG et al (2021) Leach’s storm-petrel (Hydrobates Leucorhous), version 1.1. Birds of the World (Editor not available). Cornell Lab of Ornithology, Ithaca, NY, USA
Powers KD, Wiley DN, Robuck AR, Olson ZH, Welch LJ et al (2020) Spatio-temporal characterization of non-breeding great shearwaters Ardenna gravis within their wintering range. Mar Ornith 48:215–229
Ramírez F, Afán I, Davies LS, Chaaradia A (2017) Climate impacts on global hot spots of marine biodiversity. Sci Adv 3:e1601198. https://doi.org/10.1126/sciadv.1601198
Rodríguez A, Rodríguez B, Carrasco MN (2012) High prevalence of parental delivery of plastic debris in Cory’s shearwaters (Calonectris diomedea). Mar Pol Bull 64:2219–2223. https://doi.org/10.1016/j.marpolbul.2012.06.011
Rodríguez A, Arcos JM, Bretagnolle V, Dias MP, Holmes ND et al (2019) Future directions in Conservation Research on petrels and shearwaters. Front Mar Sci 6. https://doi.org/10.3389/fmars.2019.00094
Rognes T, Flouri T, Nichols B, Quince C, Mahé F (2016) Vsearch: a versatile open source tool for metagenomics. PeerJ 4:e2584. https://doi.org/10.7717/peerj.2584
Senko JF, Nelms SE, Reavis JL, Witherington B, Godley BJ et al (2020) Understanding individual and population-level effects of plastic pollution on marine megafauna. Endanger Species Res 43:234–252. https://doi.org/10.3354/esr01064
Sousa LL, Silva SM, Xavier R (2019) DNA metabarcoding in diet studies: unveiling ecological aspects in aquatic and terrestrial ecosystems. Environm DNA 1:199–214. https://doi.org/10.1002/edn3.27
Spear LB, Ainley DG, Walker WA (2007) Foraging dynamics of seabirds in the eastern tropical Pacific Ocean. Stud Avian Biol 35:1–99
Steele DH, Montevecchi WA (1994) Leach’s storm-petrels prey on lower mesopelagic (Mysidacea and Decapoda) crustaceans: possible implications for crustacean and avian distributions. Crustaceana 66:212–218. https://www.jstor.org/stable/20104940
Sutton TT, Letessier TB, Bardarson B (2013) Midwater fishes collected in the vicinity of the Sub-polar Front. Mid-North Atlantic Ocean, during ECOMAR pelagic sampling, Deep-Sea Res II Top Stud Oceanogr 98: 292–300. https://doi.org/10.1016/j.dsr2.2013.08.001.
Sutton TT, Clark MR, Dunn DC, Halpin PN, Rogers AD et al (2017) A global biogeographic classification of the mesopelagic zone. Deep-Sea Res I 126:85–102. https://doi.org/10.1016/j.dsr.2017.05.006
Vecchione M, Pohle G (2002) Midwater cephalopods in the western north Atlantic Ocean off Nova Scotia. Bull Mar Sci 71(2):883–892
Ventura F, Granadeiro JP, Catry P, Gjerdrum C, De Pascalis F et al (2024) Allochrony is shaped by foraging niche segregation rather than adaptation to the windscape in long-raging seabirds. Mov Ecol 12:27. https://doi.org/10.1186/s40462-024-00463-z
Vermeer K, Devito K (1988) The importance of Paracallisoma coecus and myctophid fishes to nesting fork-tailed and Leach’s storm-petrels in the Queen Charlotte Islands, British Columbia. J Plank Res 10:63–75. https://doi.org/10.1093/plankt/10.1.63
Vestheim H, Jarman SN (2008) Blocking primers to enhance PCR amplification of rare sequences in mixed samples - a case study on prey DNA in Antarctic krill stomachs. Front Zool 5:12. https://doi.org/10.1186/1742-9994-5-12
Waap S (2015) Trophic relationships among pelagic predators of the deep seas of the Madeira islands. PhD thesis, Cardiff University
Waap S, Symondson WOC, Granadeiro JP, Alonso H, Serra-Gonçalves C et al (2017) The diet of a nocturnal pelagic predator, the Bulwer’s petrel, across the lunar cycle. Sci Rep 7:1384. https://doi.org/10.1038/s41598-017-01312-3
Wakefield ED (2018) Distribution and Ecology of Seabirds in the Sub-Polar Frontal Zone of the Northwest Atlantic - Cruise Report. University of Glasgow. http://eprints.gla.ac.uk/153574. Accessed 20 August 2023
Wakefield ED, Miller DL, Bond SL, le Bouard F, Carvalho PC et al (2021) The summer distribution, habitat associations and abundance of seabirds in the sub-polar frontal zone of the Northwest Atlantic. Prog Oceanogr 198:102657. https://doi.org/10.1016/j.pocean.2021.102657
Warham J (1990) The petrels. Academic, San Diego
Watanuki Y (1985) Food of breeding Leach’s storm-petrels (Oceanodroma leucorhoa). Auk 102:884–886. https://www.jstor.org/stable/4086731
Watanuki Y, Thiebot JB (2018) Factors affecting the importance of myctophids in the diet of the world’s seabirds. Mar Biol 165:79. https://doi.org/10.1007/s00227-018-3334-y
Wilhelm SI, Hedd A, Robertson GJ, Mailhiot J, Regular PM et al (2019) The world’s largest breeding colony of Leach’s storm-petrel Hydrobates Leucorhous has declined. Bird Conserv Intl 30:40–57. https://doi.org/10.1017/S0959270919000248
Wilson RP (1984) An improved stomach pump for penguins and other Seabirds. J Field Ornith 55:109–112
Young JW, Hunt BPV, Cook TR, Llopiz JK, Hazen EL et al (2015) The trophodynamics of marine top predators: current knowledge, recent advances and challenges. Deep-Sea Res II Top Stu Oceanogr 113:170–187. https://doi.org/10.1016/j.dsr2.2014.05.015
Acknowledgements
This study was financed by the Fundação para a Ciência e a Tecnologia, Portugal, through project PTDC/BIA-EVL/28565/2017, grants awarded to MARE ((UIDB/04292/2020 and UIDP/04292/2020) and cE3c (UIDB/BIA/00329/2020) and project LA/P/0069/2020 granted to the Associate Laboratory ARNET. Cruise DY080 and EDW were funded by the UK Natural Environmental Research Council (NERC) (grant number NE/M017990/1). MCS is funded by FCT through a contract foreseen in DL 57/2016, changed by Law 57/2017. We are very grateful to Captain Antonio Gatti and the crew of the RRS Discovery for facilitating sample collection, to Elizabeth Kilbride for molecular sex determination, and to José Pedro Granadeiro and Maria Dias for statistical expertise.
Funding
This study was financed by the Fundação para a Ciência e a Tecnologia, Portugal, through financing awarded to MARE (UIDB/04292/2020 and UIDP/04292/2020) and cE3c (UIDB/BIA/00329/2020), project PTDC/BIA-EVL/28565/2017, and project LA/P/0069/2020 granted to the Associate Laboratory ARNET. Cruise DY080 and EDW were funded by the UK Natural Environmental Research Council (NERC) (grant number NE/M017990/1).
Open access funding provided by FCT|FCCN (b-on).
Author information
Authors and Affiliations
Contributions
PC and EW contributed to the study conception and design. They also conducted all field work. Material preparation, data collection and analysis were performed by MCS, VN, JN. The first draft of the manuscript was written by MCS, and PC and EW commented on earlier versions. All authors agreed with the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
The work reported in this paper complies with the standards and procedures laid down by national legislations from UK and Portugal.
Competing interests
All authors declare that they have no conflict of interests and have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by V. Paiva.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Silva, M.C., Catry, P., Newton, J. et al. Diet of non-breeding leach’s storm-petrels (Hydrobates leucorhous) in the sub-polar frontal zone of the North Atlantic. Mar Biol 171, 148 (2024). https://doi.org/10.1007/s00227-024-04469-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04469-4