Abstract
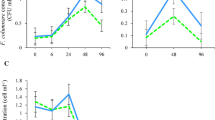
The diatom Nitzschia bizertensis was recently discovered in Bizerte Lagoon (Tunisia, SW Mediterranean Sea), where it was shown to produce the amnesic shellfish poisoning toxin, domoic acid (DA). This diatom was previously found to be widespread in both time and space throughout the lagoon. The present study was carried out to test the hypothesis that N. bizertensis is able to survive after being filtered and ejected as biodeposits (faeces plus pseudofaeces) by mussels. N. bizertensis cultures (strains BD4 and BD8), at natural bloom concentrations, were fed to mussels (Mytilus sp.). The stomach contents (after 1 h) and biodeposits (after 48 h) were observed for the presence and viability of N. bizertensis cells. Light microscopy revealed intact and partial cells of N. bizertensis in both the stomach contents and biodeposits of the mussels. Incubation of the biodeposits resulted in the regrowth of N. bizertensis. This provides evidence that transplanted mussels have the potential to serve as vectors for the transport of N. bizertensis. Furthermore, cells regrown from the biodeposits contained DA, but at levels lower than those in pre-filtered cells (up to 11.9 fg cell−1). Thus, regrown cells remain toxigenic. The results suggest that one must guard against spreading N. bizertensis and other DA producers via the transfer of shellfish from contaminated to pristine areas.





Similar content being viewed by others
References
Anderson DM, Cembella AD, Hallegraeff GM (2012) Progress in understanding harmful algal blooms: paradigm shifts and new technologies for research, monitoring, and management. Annu Rev Mar Sci 4:143–176
Bardouil M, Bohec M, Cormerais M, Bougrier S, Lassus P (1993) Experimental study of a toxic microalgal diet on feeding of the oyster Crassostrea gigas (Thunberg). J Shellfish Res 12:417–422
Barillé L, Cognie B (2000) Revival capacity of diatoms in bivalve pseudofaeces and faeces. Diatom Res 15:11–17
Bates SS, de Freitas ASW, Milley JE, Pocklington R, Quilliam MA, Smith JC, Worms J (1991) Controls on domoic acid production by the diatom Nitzschia pungens f. multiseries in culture: nutrients and irradiance. Can J Fish Aquat Sci 48:1136–1144
Bouchouicha-Smida D, Lundholm N, Kooistra WHCF, Sahraoui I, Ruggiero MV, Kotaki Y, Ellegaard M, Lambert C, Mabrouk HH, Hlaili AS (2014) Morphology and molecular phylogeny of Nitzschia bizertensis sp. nov.—a new domoic acid-producer. Harmful Algae 32:49–63
Bouchouicha-Smida D, Lundholm N, Sahraoui I, Lambert C, Mabrouk HH, Hlaili AS (2015) Detection of domoic acid in Mytilus galloprovincialis and Ostrea edulis linked to the presence of Nitzschia bizertensis in Bizerte Lagoon (SW Mediterranean). Estuar Coast Shelf Sci (in press)
Bricelj VM, Greene M, Cembella AD (1993) Growth of the blue mussel Mytilus edulis on toxic Alexandrium fundyense and effects of gut passage on dinoflagellate cells. In: Smayda TJ, Shimizu Y (eds) Toxic phytoplankton blooms in the sea. Elsevier, New York, pp 371–376
Briski E, Bailey SA, Casas-Monroy O, DiBacco C, Kaczmarska I, Lawrence JE, Leichsenring J, Levings C, MacGillivary ML, McKindsey CW, Nasmith LE, Parenteau M, Piercey GE, Rivkin RB, Rochon A, Roy S, Simard N, Sun B, Way C, Weise AM, MacIsaac HJ (2013) Taxon- and vector-specific variation in species richness and abundance during the transport stage of biological invasions. Limnol Oceanogr 58:1361–1372
Carriker MR (1992) Introductions and transfers of molluscs: risk consideration and implications. J Shellfish Res 11:507–510
Daguin C, Bonhomme F, Borsa P (2001) The zone of sympatry and hybridization of Mytilus edulis and M. galloprovincialis, as described by intron length polymorphism at locus mac-1. Heredity 86:342–354
DGPA, Direction Générale de la Pêche et de l’Aquaculture (2013) Plan Directeur de l’Aquaculture en Tunisie. Programme des Nations-Unies pour le Développement (PNUD), Ministère de l’Agriculture, République Tunisienne
Guéguen M, Lassus P, Laabir M, Bardouil M, Baron R, Séchet V, Truquet P, Amzil Z, Barillé L (2008) Gut passage times in two bivalve molluscs fed toxic microalgae: Alexandrium minutum, A. catenella and Pseudo-nitzschia calliantha. Aquat Living Resour 21:21–29
Guillard RRL (1973) Division rates. In: Stein JR (ed) Handbook of phycological methods. Vol I. Culture methods and growth measurements. Cambridge Univ Press, Cambridge, pp 289–312
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrate animals. Plenum Publ Corp, New York, pp 29–60
Hallegraeff GM (1993) A review of harmful algal blooms and their apparent global increase. Phycologia 32:79–99
Hamer JP, McCollin TA, Lucas IAN (2000) Dinoflagellate cysts in ballast tank sediments: between tank variability. Mar Poll Bull 40:731–733
Harper FM, Hatfield EA, Thompson RJ (2002) Recirculation of dinoflagellate cysts by the mussel, Mytilus edulis L., at an aquaculture site contaminated by Alexandrium fundyense (Lebour) Balech. J Shellfish Res 21:471–477
Hégaret H, Shumway SE, Wikfors GH, Pate S, Burkholder JM (2008) Potential transport of harmful algae via relocation of bivalve molluscs. Mar Ecol Prog Ser 361:169–179
Laabir M, Gentien P (1999) Survival of toxic dinoflagellates after gut passage in the Pacific oyster Crassostrea gigas Thunburg. J Shellfish Res 18:217–222
Laabir M, Amzil Z, Lassus P, Masseret E, Tapilatu Y, De Vargas R, Grzebyk D (2007) Viability, growth and toxicity of Alexandrium catenella and Alexandrium minutum (Dinophyceae) following ingestion and gut passage in the oyster Crassostrea gigas. Aquat Living Resour 20:51–57
Lahbib Y, Abidli S, Trigui El Menif N (2009) Relative growth and reproduction in Tunisian populations of Hexaplex trunculus with contrasting imposex levels. J Shellfish Res 28:891–898
Lilly EL, Kulis DM, Gentien P, Anderson DM (2002) Paralytic shellfish poisoning toxins in France linked to a human introduced strain of Alexandrium catenella from the western Pacific: evidence from DNA and toxin analysis. J Plank Res 24:443–452
Loeblich AR, Smith VE (1968) Chloroplast pigments of the marine dinoflagellate Gyrodinium resplendens. Lipids 8:5–13
Medhioub W, Lassus P, Truquet P, Bardouil M, Amzil Z, Sechet V, Sibat M, Soudant P (2012) Spirolide uptake and detoxification by Crassostrea gigas exposed to the toxic dinoflagellate Alexandrium ostenfeldii. Aquaculture 358–359:108–115
Penna A, Garcés E, Vila M, Giacobbe MG, Fraga S, Lugliè A, Bravoj I, Bertozzini E, Vernesi C (2005) Alexandrium catenella (Dinophyceae), a toxic ribotype expanding in the NW Mediterranean Sea. Mar Biol 148:13–23
Powell E, Ashton-Alcox K (2004) A comparison between a suction dredge and a traditional oyster dredge in the transplantation of oysters in Delaware Bay. J Shellfish Res 23:803–823
Rolland JL, Pelletier K, Masseret E, Rieuvilleneuve F, Savar V, Santini A, Amzil Z, Laabir M (2012) Paralytic toxins accumulation and tissue expression of α-amylase and lipase genes in the Pacific oyster Crassostrea gigas fed with the neurotoxic dinoflagellate Alexandrium catenella. Mar Drugs 10:2519–2534
Rosa M, Holohan BA, Shumway SE, Bullard SG, Wikfors GH, Morton S, Getchis T (2013) Biofouling ascidians on aquaculture gear as potential vectors of harmful algal introductions. Harmful Algae 23:1–7
Ruesink JL, Lenihan HS, Trimble AC, Heiman KW, Micheli F, Byers JE, Kay MC (2005) Introduction of non-native oysters: ecosystem effects and restoration implications. Annu Rev Ecol Evol Syst 36:643–689
Ruiz GM, Fofonoff PW, Carlton JT, Wonham MJ, Hines AH (2000a) Invasion of coastal marine communities in North America: apparent patterns, processes, and biases. Annu Rev Ecol Evol Syst 31:481–531
Ruiz GM, Rawlings TK, Dobbs FC, Drake LA, Mullady T, Huq A, Colwell RR (2000b) Global spread of microorganisms by ships. Nature 408:49–50
Sahraoui I, Bates SS, Bouchouicha D, Mabrouk HH, Hlaili AS (2011) Toxicity of Pseudo-nitzschia populations from Bizerte Lagoon, Tunisia, southwest Mediterranean, and first report of domoic acid production by P. brasiliana. Diatom Res 26:293–303
Sahraoui I, Grami B, Bates SS, Bouchouicha D, Chikaoui MA, Mabrouk HH, Hlaili AS (2012) Response of potentially toxic Pseudo-nitzschia (Bacillariophyceae) populations and domoic acid to environmental conditions in a eutrophied, SW Mediterranean coastal lagoon (Tunisia). Estuar Coast Shelf Sci 102–103:95–104
Scarratt AM, Scarratt DJ, Scarratt MG (1993) Survival of live Alexandrium tamarense cells in mussel and scallop spat under simulated transfer conditions. J Shellfish Res 12:383–388
Tammilehto A, Nielsen TG, Krock B, Møller EF, Lundholm N (2015) Induction of domoic acid production in the toxic diatom Pseudo-nitzschia seriata by calanoid copepods. Aquat Toxicol 159:52–61
Vila M, Garcés E, Maso M, Camp J (2001) Is the distribution of the toxic dinoflagellate Alexandrium catenella expanding along the NW Mediterranean coast? Mar Ecol Prog Ser 222:73–83
Acknowledgments
The authors thank Hansy Haberkorn, Hélène Hégaret, Marcel Koken and Nelly Le Goïc for their technical assistance with carrying out the experiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: E. Briski.
Reviewed by Z. Armi and C. Lafabri.
This article is part of the Topical Collection on Invasive Species.
Rights and permissions
About this article
Cite this article
Bouchouicha-Smida, D., Bates, S.S., Lundholm, N. et al. Viability, growth and domoic acid toxicity of the diatom Nitzschia bizertensis following filtration by the mussel Mytilus sp.. Mar Biol 162, 2513–2519 (2015). https://doi.org/10.1007/s00227-015-2758-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-015-2758-x