Abstract
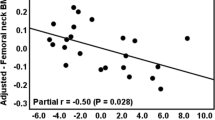
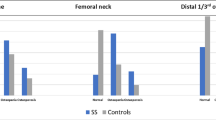
Due to disturbances in hormones and long-term glucocorticoid replacement therapy (GRT), congenital adrenocortical hyperplasia (CAH) patients are at risk of impaired bone structure and metabolism. This cross-sectional, case–control study aims to investigate for the first time bone microarchitecture features in 21-hydroxylase deficiency (21OHD; N = 38) and 17α-hydroxylase deficiency (17OHD; N = 16) patients using high-resolution peripheral quantitative computed tomography (HR-pQCT) by matching the same sex and similar age [21OHD vs. control: 29.5 (24.0–34.3) vs. 29.6 (25.9–35.2) years; 17OHD vs. controls: 29.0 (21.5–35.0) vs. 29.7 (24.6–35.3) years] with healthy controls (1:3). All patients underwent HR-pQCT scans of the nondominant radius and tibia, and had received GRT. Compared with corresponding controls, 17OHD cases had higher height (P < 0.001), weight (P = 0.013) and similar body mass index (BMI), while 21OHD had lower height (P < 0.001), similar weight and higher BMI (P < 0.001). 17OHD and 21OHD patients demonstrated various significant bone differences in most HR-pQCT indices, suggesting abnormalities in bone microarchitectures from healthy people. Further correlation analyses revealed that some characteristics, such as height and hormones, may contribute to the bone differences in HR-pQCT indices between two diseases. However, treatment dosage and time were not correlated, indicating that the current glucocorticoid doses may be within safety limits for bone impairment. Overall, our study for the first time revealed changes of bone microarchitecture in CAH patients and their potential relations with clinical characteristics. Further longitudinal researches are required to confirm these findings.



Similar content being viewed by others
References
Claahsen-van der Grinten H, Stikkelbroeck N, Falhammar H, Reisch N (2021) Management of endocrine disease: gonadal dysfunction in congenital adrenal hyperplasia. Eur J Endocrinol 184:R85–R97
Merke D, Bornstein S (2005) Congenital adrenal hyperplasia. Lancet (London, England) 365:2125–2136
Marsh C, Auchus R (2014) Fertility in patients with genetic deficiencies of cytochrome P450c17 (CYP17A1): combined 17-hydroxylase/17,20-lyase deficiency and isolated 17,20-lyase deficiency. Fertil Steril 101:317–322
Hardy R, Zhou H, Seibel M, Cooper M (2018) Glucocorticoids and bone: consequences of endogenous and exogenous excess and replacement therapy. Endocr Rev 39:519–548
Warriner A, Saag K (2013) Glucocorticoid-related bone changes from endogenous or exogenous glucocorticoids. Curr Opin Endocrinol Diabetes Obes 20:510–516
Messina O, Vidal L, Vidal M, Bultink I, Raterman H, Lems W (2021) Management of glucocorticoid-induced osteoporosis. Aging Clin Exp Res 33:793–804
Zavatta G, Clarke B (2021) Glucocorticoid- and transplantation-induced osteoporosis. Endocrinol Metab Clin North Am 50:251–273
Arisaka O, Hoshi M, Kanazawa S, Numata M, Nakajima D, Kanno S, Negishi M, Nishikura K, Nitta A, Imataka M, Kuribayashi T, Kano K (2001) Preliminary report: effect of adrenal androgen and estrogen on bone maturation and bone mineral density. Metabolism 50:377–379
De Oliveira D, Fighera T, Bianchet L, Kulak C, Kulak J (2012) Androgens and bone. Minerva Endocrinol 37:305–314
Halper A, Sanchez B, Hodges J, Dengel D, Petryk A, Sarafoglou K (2019) Use of an aromatase inhibitor in children with congenital adrenal hyperplasia: impact of anastrozole on bone mineral density and visceral adipose tissue. Clin Endocrinol 91:124–130
Espinosa Reyes T, Leyva González G, Domínguez Alonso E, Falhammar H (2021) Bone mass in young patients with congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Horm Res Paediatr 94:1–8
Auer M, Paizoni L, Hofbauer L, Rauner M, Chen Y, Schmidt H, Huebner A, Bidlingmaier M, Reisch N (2020) Effects of androgen excess and glucocorticoid exposure on bone health in adult patients with 21-hydroxylase deficiency. J Steroid Biochem Mol Biol 204:105734
Cameron FJ, Kaymakci B, Byrt EA, Ebeling PR, Warne GL, Wark JD (1995) Bone mineral density and body composition in congenital adrenal hyperplasia. J Clin Endocrinol Metab 80:2238–2243
de Almeida Freire PO, de Lemos-Marini SH, Maciel-Guerra AT, Morcillo AM, Matias Baptista MT, de Mello MP, Guerra G Jr (2003) Classical congenital adrenal hyperplasia due to 21-hydroxylase deficiency: a cross-sectional study of factors involved in bone mineral density. J Bone Miner Metab 21:396–401
Girgis R, Winter JS (1997) The effects of glucocorticoid replacement therapy on growth, bone mineral density, and bone turnover markers in children with congenital adrenal hyperplasia. J Clin Endocrinol Metab 82:3926–3929
King JA, Wisniewski AB, Bankowski BJ, Carson KA, Zacur HA, Migeon CJ (2006) Long-term corticosteroid replacement and bone mineral density in adult women with classical congenital adrenal hyperplasia. J Clin Endocrinol Metab 91:865–869
Paganini C, Radetti G, Livieri C, Braga V, Migliavacca D, Adami S (2000) Height, bone mineral density and bone markers in congenital adrenal hyperplasia. Horm Res 54:164–168
Sciannamblo M, Russo G, Cuccato D, Chiumello G, Mora S (2006) Reduced bone mineral density and increased bone metabolism rate in young adult patients with 21-hydroxylase deficiency. J Clin Endocrinol Metab 91:4453–4458
Zimmermann A, Sido PG, Schulze E, Al Khzouz C, Lazea C, Coldea C, Weber MM (2009) Bone mineral density and bone turnover in Romanian children and young adults with classical 21-hydroxylase deficiency are influenced by glucocorticoid replacement therapy. Clin Endocrinol 71:477–484
Falhammar H, Filipsson H, Holmdahl G, Janson P, Nordenskjöld A, Hagenfeldt K, Thorén M (2007) Fractures and bone mineral density in adult women with 21-hydroxylase deficiency. J Clin Endocrinol Metab 92:4643–4649
dos Santos CV, Vieira Neto L, Madeira M, Alves Coelho MC, de Mendonca LM, Paranhos-Neto Fde P, Lima IC, Gadelha MR, Farias ML (2015) Bone density and microarchitecture in endogenous hypercortisolism. Clin Endocrinol 83:468–474
Xu L, Yu J, Wang O, Hou Y, Li W, Zhang H, Ping F, Xu Q, Li Y, Xia W (2021) Comparison of differences in bone microarchitecture in adult- versus juvenile-onset type 1 diabetes Asian males versus non-diabetes males: an observational cross-sectional pilot study. Endocrine 71:87–95
Yang H, Yan K, Yuping X, Zhang Q, Wang L, Gong F, Zhu H, Xia W, Pan H (2019) Bone microarchitecture and volumetric bone density impairment in young male adults with childhood-onset growth hormone deficiency. Eur J Endocrinol 180:145–153
Gao Y, Yu B, Mao J, Wang X, Nie M, Wu X (2020) The prevalence of heterozygous CYP21A2 deficiency in patients with idiopathic acne, hirsutism, or both. Endocrine 67:665–672
Gao Y, Wang O, Guan W, Wu X, Mao J, Wang X, Yu W, Nie M (2021) Bone mineral density and trabecular bone score in patients with 21-hydroxylase deficiency after glucocorticoid treatment. Clin Endocrinol 94:765–773
Nicks K, Amin S, Atkinson E, Riggs B, Melton L, Khosla S (2012) Relationship of age to bone microstructure independent of areal bone mineral density. J Bone Miner Res 27:637–644
Geusens P, Chapurlat R, Schett G, Ghasem-Zadeh A, Seeman E, de Jong J, van den Bergh J (2014) High-resolution in vivo imaging of bone and joints: a window to microarchitecture. Nat Rev Rheumatol 10:304–313
Wu C, Fan S, Qian Y, Zhou Y, Jin J, Dai Z, Jiang L (2017) 17α-hydroxylase/17, 20-lyase deficiency: clinical and molecular characterization of eight Chinese patients. Endocr Pract 23:576–582
Elnecave R, Kopacek C, Rigatto M, Keller Brenner J, Sisson de Castro J (2008) Bone mineral density in girls with classical congenital adrenal hyperplasia due to CYP21 deficiency. J Pediatr Endocrinol Metab 21:1155–1162
Ganesh R, Suresh N, Janakiraman L (2018) Bone mineral content and density in Indian children with congenital adrenal hyperplasia. Indian Pediatr 55:880–882
Metwalley K, El-Saied A (2014) Bone mineral status in Egyptian children with classic congenital adrenal hyperplasia. A single-center study from Upper Egypt. Indian J Endocrinol Metab 18:700–704
Rangaswamaiah S, Gangathimmaiah V, Nordenstrom A, Falhammar H (2020) Bone mineral density in adults with congenital adrenal hyperplasia: a systematic review and meta-analysis. Front Endocrinol 11:493
Jääskeläinen J, Kiekara O, Hippeläinen M, Voutilainen R (2000) Pituitary gonadal axis and child rate in males with classical 21-hydroxylase deficiency. J Endocrinol Invest 23:23–27
Ceccato F, Barbot M, Albiger N, Zilio M, De Toni P, Luisetto G, Zaninotto M, Greggio NA, Boscaro M, Scaroni C, Camozzi V (2016) Long-term glucocorticoid effect on bone mineral density in patients with congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Eur J Endocrinol 175:101–106
Hardy R, Cooper MS (2010) Adrenal gland and bone. Arch Biochem Biophys 503:137–145
Zhang D, Sun J, Xu J, Xing Y, Zheng M, Ye S, Zhu J (2021) 17α-hydroxylase/17,20 carbon chain lyase deficiency caused by p.Tyr329fs homozygous mutation: three case reports. World J Clin Cases 9:1923–1930
New M (2004) An update of congenital adrenal hyperplasia. Ann N Y Acad Sci 1038:14–43
Dalle Carbonare L, Arlot M, Chavassieux P, Roux J, Portero N, Meunier P (2001) Comparison of trabecular bone microarchitecture and remodeling in glucocorticoid-induced and postmenopausal osteoporosis. J Bone Miner Res 16:97–103
Dalle Carbonare L, Bertoldo F, Valenti M, Zenari S, Zanatta M, Sella S, Giannini S, Cascio V (2005) Histomorphometric analysis of glucocorticoid-induced osteoporosis. Micron 36:645–652
Tsugeno H, Tsugeno H, Fujita T, Goto B, Sugishita T, Hosaki Y, Ashida K, Mitsunobu F, Tanizaki Y, Shiratori Y (2002) Vertebral fracture and cortical bone changes in corticosteroid-induced osteoporosis. Osteoporos Int 13:650–656
Acknowledgements
We thank American Journal Experts for the service with the grammar editing.
Funding
This study was supported by CAMS Innovation Fund for Medical Sciences (No. 2021-I2M-C&T-B-001), Center for Rare Diseases Research, Chinese Academy of Medical Sciences, Beijing, China (No. 2016ZX310174-4), and Natural Science Foundation of Sichuan Province (No. 2023NSFSC1885).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosure
Xu Sun, Yijun Wu, Lin Lu, Weibo Xia, Li Zhang, Shi Chen, Min Nie, Guangyao Zheng, Wan Su, Huijuan Zhu and Zhaolin Lu declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This study was carried out in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of PUMCH. All patients and controls signed written informed consent forms.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
223_2023_1132_MOESM1_ESM.jpg
Supplementary file1 (JPG 5677 kb)—Supplemental Figure 1. HR-pQCT images of bone microarchitecture in patients with congenital adrenocortical hyperplasia (17OHD and 21OHD) and healthy controls. There were obvious differences in bone microarchitecture between each patient and his or her corresponding control. Compared with controls or 17OHD group, the 21OHD group had higher Ct.Th (P<0.001). Other specific differences are shown in the results. A: Control of 21OHD (20-year-old female); B: Patient with 21OHD (20-year-old female); C: Control of 17OHD (20-year-old female); D: Patient with 17OHD (20-year-old female). R: radius; T: tibia. A1-D1 and A4-D4 for cortical bone; A2-D2 and A5-D5 for trabecular bone; A3-D3 and A6-D6 for total bone
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, X., Wu, Y., Lu, L. et al. Bone Microarchitecture and Volumetric Mineral Density Assessed by HR-pQCT in Patients with 21- and 17α-Hydroxylase Deficiency. Calcif Tissue Int 113, 515–525 (2023). https://doi.org/10.1007/s00223-023-01132-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-023-01132-w