Abstract
Factors related to mortality after osteoporotic hip fracture (HF) have been investigated intensively, except for proximal femoral bone mineral density (BMD), which is also the primary cause of osteoporosis. In this study, we aimed to investigate the association of hip BMD with mortality risk after HF. Four hundred and eleven elderly patients with HF in Beijing, China, were included and prospectively followed up with a median time of 3 years. At baseline, quantitative CT technique (QCT) was used to measure areal BMD (aBMD) of the unaffected hip. Areal BMDs of the total hip (TH), femoral neck (FN), trochanter (TR), and intertrochanter were analyzed with postoperative mortality as the primary outcome. A total of 394 patients (78.59 ± 7.59 years, 75.4% female) were included in our final analysis, with 86 (82.23 ± 7.00 years, 81.4% female) dead. All hip bone densities demonstrated a significant association with mortality risks in the unadjusted model, but only TR aBMD remained significantly correlated after adjusting for all covariates. Compared to the lower TR aBMD group, the higher TR aBMD group yielded significantly lower death risks (HR 0.21 95% CI 0.05–0.9, P = 0.036). Higher survival probabilities were observed for higher TH and TR aBMD in survival analysis (P < 0.001). Hip BMD, especially TR BMD assessed by QCT, is an independent risk factor for postoperative mortality following HF. QCT may present a promising avenue for opportunistic analysis in immobilized patients, providing valuable information for early detection and personalized interventions to enhance patient outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporotic hip fracture (HF) is a common and severe injury among older people, most of whom suffer from low-energy trauma, imposing a huge burden on patients and health systems [1]. According to the World Health Organization, the number of osteoporotic HF will triple over the next 50 years, from 1.7 million in 1990 to 6.3 million in 2050 worldwide [2]. The injured elderly are usually frail, and the associated comorbidities are known to be implicated in the postoperative risk of death [3, 4]. The one-year mortality rate for HF patients was reported to be up to 20–24%, and the mortality risk may persist beyond 5 years [5]. Thus, it is crucial to identify those patients at high risk of death, which precedes the subsequent interventions and strategies for improvement.
Osteoporosis, the primary cause of osteoporotic HF, is characterized by low bone density, which might be a non-specific marker for aging and weakness. Multiple studies have found that bone mineral density (BMD) is inversely associated with adverse events, including death [6,7,8,9]. Lower BMD was further revealed to contribute to higher refracture and death risk after fragility fractures [10], although the evidence was quite limited. Perhaps due to the difficulty for HF elderly patients to examine BMD, few studies focused on the impact of baseline BMD on future mortality after HF, which is still unclear.
Nowadays, to better evaluate the injury pattern and plan the operation, preoperative computed tomography (CT) scans of the proximal femur have become routine in HF patients. The advent of the quantitative CT (QCT) technique further enables radiologists and orthopedists opportunity to explore the hip structure and bone densities [11]. Different parts of the proximal femur could be separated through QCT, and the BMD of some parts (like trochanter (TR) and intertrochanter (IT)) were suggested to be better parameters in clinical prediction than the femoral neck (FN) or total hip (TH) BMD [12,13,14], which is most often used as a representative measure of proximal femoral BMD. To our knowledge, no studies have explored the association of BMD levels in different parts of the proximal femur with mortality risk after osteoporotic HF.
In this study, using QCT, we first aimed to investigate the association between hip BMD and mortality risk after HF, and second whether the role of bone density in predicting death varies according to different parts of the proximal femur. We hypothesized that higher BMD was associated with lower death risk, and further, TR BMD was a stronger predictor of the mortality risk for HF patients than FN or TH BMD.
Materials and Methods
Design
The present study was performed in a single tertiary hospital in Beijing, China, with a co-managed orthogeriatric hip fracture care path. Ethics approval was obtained from the Institutional Review Board of the hospital (201,807-II) before the initiation of this study. The present post hoc analysis used baseline data prospectively collected in our previous observational study [15], exploring the effect of a co-management care model on older hip fracture patients in China (Clinical Trials.gov Identifier: NCT03184896). For our specific purposes, from November 2018 to November 2019, a total of 565 hip fracture subjects over 65 with eligible QCT imaging were screened. The clinical approach has been described previously [15]. All included subjects were followed up by telephone for a median time of 3.5 years (from 2018–2019 to 2021–2022).
During the screening, we ruled out the subjects presented over 48 h from injury to ensure immediate CT imaging after a fracture. We excluded those who cannot fully ambulate and sustained non-ground level falls. Those who underwent conservative therapy, those with contralateral hip lesions (like fractures or bone necrosis) that may disturb imaging measurements, and those with severe diseases that may interfere with bone metabolism (like parathyroid diseases) were also excluded. Further exclusion criteria were pathological fractures or terminal malignancies. All patients gave written informed consent.
QCT Scans and Bone Mineral Density Measurement
To avoid the bias caused by CT machine, two same Toshiba Aquilion 64-slices CT scanners (Toshiba Medical Systems Corp., Tokyo, Japan) with a solid phantom (Mindways Software Inc., Austin, TX, USA) were used. The subjects were scanned in the supine position, with the phantom beneath the hip. Hips were scanned from the top of the acetabulum to a level of 3 cm inferior to the lesser trochanter or longer to cover the fractured bone. The scan parameters were the same: 120 Kvp, 125 mAs, 1 mm thickness, 50 cm field of view, and 512 × 512 matrix in spiral reconstruction and standard reconstruction. Subsequently, the acquired CT images were transferred to a QCT workstation and analyzed using the computed tomography X-ray absorptiometry function (CTXA, version 4.2.3) of Mindways QCT pro software (Mindways Software Inc., Austin, TX, USA). The measurement procedure was previously described, and the areal BMD (aBMD, g/cm2) was obtained from CTquals DXA [16]. Briefly, two-dimensional projections were generated from the three-dimensional CT dataset after image segmentation and proper manipulation. All subjects’ aBMD of the femoral neck (FN), greater trochanter (TR), intertrochanter (IT), and total hip (TH) were then measured on the unaffected side.
Data Collection and Outcome Measures
The demographic information and perioperative records were prospectively collected. The demographic data included age, sex, weight, height, bone mass index (BMI), drinking or smoking habits, education level, and living status. Baseline medical situations, such as hypertension, diabetes, and cognitive and visual impairment, were also investigated and documented. Mini-Mental State Examination—China (MMSE) [17] was further used to quantify cognitive ability, and participants with an MMSE score of 23 or lower were defined as having cognitive impairment. The Charlson comorbidity index (CCI) [18] was then calculated to further represent the overall medical situation. Perioperative variables included fracture pattern, side, American Society of Anesthesiologists (ASA) scores, type of anesthesia and operation, length of stay (LOS), and rehabilitation or not. Albumin level was also recorded to indicate nutrition status while falling times in the past year indicate the tendency to be injured. The educational level was composed of five states, ranging from illiterate to university or higher. Concerning operations, intertrochanteric fractures were treated by intramedullary nailing, while femoral neck fractures were treated by cannulated screw fixation or arthroplasty. The operation type was categorized into internal fixation (intramedullary nailing and screw fixation) and arthroplasty.
All patients received telephone visits by orthopaedists after 3 years. Our primary outcome was the postoperative three-year mortality from all causes. Secondary outcomes included the living place alteration (institutionalized or not) and health-related quality of life (HRQoL) at the final follow-up. Institutionalized sites refer to hospitals, rehabilitation centers, or nursing homes. Euro-Qol 5 Dimensions Score (EQ-5D 5L) [18], a generic health-utility instrument, was used to measure HRQoL. It consists of a five-level response (no problems, slight problems, moderate problems, severe problems, extreme problems) for five health domains related to daily activities: mobility; self-care; usual activities; pain and discomfort; and anxiety and depression. We then converted the responses into an overall score using a published utility model for the China population [19].
Statistical Analysis
Data are described as means and standard deviations for parametric data or as medians and interquartile ranges when the data are not normally distributed. Categorical data are presented using frequencies and numerical distributions. The Chi-squared test was used to assess the differences between the two groups for categorical variables and Student’s t test or the Mann–Whitney U test for continuous variables, as appropriate (parametric vs non-parametric data, respectively).
In our present study, considering the inherent differences in BMD between males and females, we first employed sex-specific Z-score normalization to facilitate further analysis and interpretation. The baseline variables that were considered clinically relevant or showed a univariate relationship with outcome were considered covariates. Based on that, multivariate Cox proportional-hazards models were used to estimate the association between the baseline BMDs and risk of death, without and with adjustments for age, sex, BMI, CCI, albumin, MMSE, housebound, fracture type, anesthesia type, and time from injury to operation. To ensure parsimony of the final model, we have transformed the latter seven covariates into a propensity score for death, calculated from a multivariate logistic regression model, given the number of death events available. Kaplan–Meier survival plots were produced using TH aBMD and FN aBMD stratified into standardized score categories: Z < − 1; Z ≥ − 1 and Z < 1; and Z ≥ 1. Subgroup analysis were also performed according to age group (above and below 80 years), sex, BMI (above and below 24), and fracture type.
All the analyses were performed with the statistical software packages R 4.1.1 (http://www.R-project.org, The R Foundation). A two-tailed test was performed, and P < 0.05 was considered statistically significant.
Results
Population and Baseline Characteristics
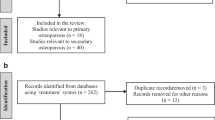
A total of 394 patients were ultimately included in our final analysis after 154 were excluded and 17 dropped out. At the end of the follow-up, 86 (21.8%) of these patients had died, including 28 (7.11%) deaths that occurred in the first year. Figure 1 presents a flowchart of the study.
The baseline characteristics of all available subjects are listed in Table 1. The mean age was 78.59 ± 7.59 years; 297 (75.4%) were women, and 189 (48%) were ITF. The median time from injury to operation was around 3 days, and the median length of stay (LOS) was around 5 days. As shown in Table 1, compared with the groups who died, all bone densities were statistically significantly higher in surviving patients (P < 0.001). More patients with cognitive impairment and lower level of albumin were also revealed in the died (26 (30.6%) vs 57 (18.6%); 39.40 ± 3.68 vs 41.36 ± 2.86, respectively).
Primary Outcome
In the unadjusted model, all hip bone densities were found to be significantly associated with the risk of death following hip fractures, as shown in Table 2. However, after adjusting for age, sex, and BMI in Model 1, only TH aBMD (HR 0.75, 95% CI 0.59–0.96, P = 0.024) and TR aBMD (HR 0.7, 95% CI 0.54–0.9, P = 0.006) remained significantly correlated with mortality risk. Further adjustment for all covariates in Model 2 revealed that TR aBMD was still significantly linked to postoperative mortality. Compared to the lower TR aBMD group (Z < − 1), the higher TR aBMD group (Z ≥ 1) yielded significantly lower death risks (HR 0.21 95% CI 0.05–0.9, P = 0.036). Kaplan–Meier curve also demonstrated lower survival probability in lower TH and TR aBMD groups (Fig. 2). Subgroup analysis examined the interaction and verified the robust associations (Supplementary Fig. 3).
Secondary Outcome
In terms of the HRQoL and living place alteration, higher BMD was associated with higher EQ-5D and a lower risk of institutionalization (Table 3). However, the significant association faded away to varying degrees after adjustments.
Discussion
In the proximal femur, trochanter (TR) BMD performs better in representing femoral strength and predicting second hip fracture than FN or TH BMD [12, 14]. However, few studies have focused on its potential in forecasting risks of death after a first HF. As far as we know, this is the first study to explore the different roles of proximal femoral BMD in predicting the mortality risks after HF occurrence in the elderly. The study results demonstrated hip BMD as a risk factor for postoperative mortality and revealed that TR aBMD was the most relevant parameter in predicting mortality risks. Higher TR aBMD and TH aBMD yielded much lower risks of death during three-year follow-up after a first HF in the elderly.
Our study found an inverse association between hip bone densities and postoperative mortality probabilities, which aligns with many previous researches [10, 20, 21]. Little explanations were given, and the intrinsic connections were still unclear. A recent systematic review [22] stated that bisphosphonates reduce all-cause mortality after osteoporotic fractures, which indirectly reflected the effect of bone density on postoperative mortality. Generally, skeletal health is a pretty good marker for overall health status, and low BMD is related to multiple factors, like estrogen metabolism [23], nutrition [24], as well as genetic factors [25]. Indeed, a range of risk factors was shared in the pathophysiological mechanisms of bad habits, chronic diseases, and low BMD, such as aging, smoking, and lack of physical activity. The albumin level relating to the overall nutrition status and the MMSE score relating to the cognitive ability were also lower in the dead group in our study, which is consistent with other studies [26, 27]. The potential metabolic association between the comorbidities, adverse events, and BMD might suggest that the bone parameters could give a better hint or predict the prognosis after surgery, as in our present study.
Moreover, we found that the TR BMD plays a prominent role in hip BMD predicting mortality. The greater trochanter is the primary attachment of posterior hip muscles (gluteus), which are strong muscles that help maintain gait stability and standing balance. The constant mechanical loading and physical forces created by the gluteus contractions could partly explain that TR BMD might better represent the proximal femur strength, as Cheng demonstrated in his in-vitro study [12]. Yin et al. [28] found a significant association between trochanter BMD and gluteus maximus muscle area through QCT imaging. In addition, TR aBMD was discovered to be the only bone parameter tightly associated with handgrip strength [28], which has become a critical indicator for screening sarcopenia [29]. The development of osteosarcopenia has suggested an indissoluble underlying crosstalk between bone and muscle metabolism [30]. It is reasonable to hypothesize that TR aBMD could be an optimal parameter predicting overall health and physical performance, which needs to be verified in well-powered studies. Furthermore, lower TR aBMD was linked to a higher probability of intertrochanteric fracture (ITF) [13], which was also a risk factor for death [4]. However, following adjustment for key confounding factors, including fracture type, CCI, nutrition level, cognitive status, and anesthesia type [4, 31], TR aBMD remained a strong and independent risk factor for postoperative death.
Few studies have explored the relationship between bone density and mortality in the context of postoperative death after HF. As described in some studies [32,33,34,35], this may be due to the challenges of obtaining dual-energy X-ray absorptiometry (DXA) scans in the acute setting for HF patients. Radiological parameters (such as cortical thickness [34, 35], Singh index, and Dorr classification [32]) were revealed to be associated with osteoporosis or mortality risks. Despite this, lots of BMD information from clinical CT imaging of HF has not been fully utilized. The findings in our study demonstrated that TR BMD assessed by QCT may serve as a potential indicator for death after HF using opportunistic analysis of routine clinical CT scans. QCT has its advantages in providing body information for patients with difficulty moving after injury, such as those with HF. We believe that the extensive information provided by routine CT scans can add tremendous value to patient care in the future [11, 36].
One of the potential strengths of our study was its comprehensive, prospectively collected data, ensuring minimal recall bias. During inclusion, we only selected those who could fully ambulate and excluded those who presented more than 48 h after injury to minimize the influence of immobilization on BMD. Besides, the well-documented medical information and adjusted confounding bias further helped us to draw a more robust relationship between bone densities and mortality. This study also has several limitations. First, the mortality rate over 3 years in our study was found to be 21.8%, which is notably lower than that reported in the previous studies [5, 37]. This could be attributed to the implementation of our co-management care model, which may effectively mitigate the mortality risks [15, 38]. Also, our study participants were relatively younger and exhibited better mobility [5]. Therefore, caution should be taken when comparing our findings with others. Second, the sample size of the cohort was relatively small, which also imposed restrictions on our analysis. Even so, significant associations between BMD and mortality were detected. Further well-designed studies with a larger sample size may dig deeper into the role of bone parameters in predicting HF patients’ prognosis.
In conclusion, hip BMD, especially TR BMD assessed by QCT, is an independent risk factor for postoperative mortality following HF. QCT may present a promising avenue for opportunistic analysis in immobilized patients, providing valuable information for early detection and personalized interventions to enhance patient outcomes.
References
Papadimitriou N, Tsilidis KK, Orfanos P et al (2017) Burden of hip fracture using disability-adjusted life-years: a pooled analysis of prospective cohorts in the CHANCES consortium. Lancet Public Health 2(5):e239–e246. https://doi.org/10.1016/S2468-2667(17)30046-4
Amarilla-Donoso FJ, Lopez-Espuela F, Roncero-Martin R et al (2020) Quality of life in elderly people after a hip fracture: a prospective study. Health Qual Life Outcomes 18(1):71. https://doi.org/10.1186/s12955-020-01314-2
Paterni S, Okoye C, Calabrese AM et al (2021) Prognostic value of glycated hemoglobin in frail older diabetic patients with hip fracture. Front Endocrinol (Lausanne) 12:770400. https://doi.org/10.3389/fendo.2021.770400
Yong EL, Ganesan G, Kramer MS et al (2020) Risk factors and trends associated with mortality among adults with hip fracture in Singapore. JAMA Netw Open 3(2):e1919706. https://doi.org/10.1001/jamanetworkopen.2019.19706
Xu BY, Yan S, Low LL, Vasanwala FF, Low SG (2019) Predictors of poor functional outcomes and mortality in patients with hip fracture: a systematic review. BMC Musculoskelet Disord 20(1):568. https://doi.org/10.1186/s12891-019-2950-0
Veronese N, Stubbs B, Crepaldi G et al (2017) Relationship between low bone mineral density and fractures with incident cardiovascular disease: a systematic review and meta-analysis. J Bone Miner Res 32(5):1126–1135. https://doi.org/10.1002/jbmr.3089
Qu X, Huang X, Jin F et al (2013) Bone mineral density and all-cause, cardiovascular and stroke mortality: a meta-analysis of prospective cohort studies. Int J Cardiol 166(2):385–393. https://doi.org/10.1016/j.ijcard.2011.10.114
Browner WS, Seeley DG, Vogt TM, Cummings SR (1991) Non-trauma mortality in elderly women with low bone mineral density. Study of osteoporotic fractures research group. Lancet 338(8763):355–358. https://doi.org/10.1016/0140-6736(91)90489-c
Zhu B, Yang J, Zhou Z et al (2022) Total bone mineral density is inversely associated with stroke: a family osteoporosis cohort study in rural China. QJM 115(4):228–234. https://doi.org/10.1093/qjmed/hcaa339
Bliuc D, Alarkawi D, Nguyen TV, Eisman JA, Center JR (2015) Risk of subsequent fractures and mortality in elderly women and men with fragility fractures with and without osteoporotic bone density: the Dubbo osteoporosis epidemiology study. J Bone Miner Res 30(4):637–646. https://doi.org/10.1002/jbmr.2393
Engelke K (2017) Quantitative computed tomography-current status and new developments. J Clin Densitom 20(3):309–321. https://doi.org/10.1016/j.jocd.2017.06.017
Cheng XG, Lowet G, Boonen S et al (1997) Assessment of the strength of proximal femur in vitro: relationship to femoral bone mineral density and femoral geometry. Bone 20(3):213–218. https://doi.org/10.1016/s8756-3282(96)00383-3
Su YB, Wang L, Wu XB et al (2019) The spatial differences in bone mineral density and hip structure between low-energy femoral neck and trochanteric fractures in elderly Chinese using quantitative computed tomography. Bone 124:62–68. https://doi.org/10.1016/j.bone.2019.04.007
Wang L, Yin L, Yang M et al (2022) Muscle density is an independent risk factor of second hip fracture: a prospective cohort study. J Cachexia Sarcopenia Muscle. https://doi.org/10.1002/jcsm.12996
Zhang J, Yang M, Zhang X et al (2022) The effectiveness of a co-management care model on older hip fracture patients in China—a multicentre non-randomised controlled study. Lancet Reg Health West Pac 19:100348. https://doi.org/10.1016/j.lanwpc.2021.100348
Cheng X, Wang L, Wang Q, Ma Y, Su Y, Li K (2014) Validation of quantitative computed tomography-derived areal bone mineral density with dual energy X-ray absorptiometry in an elderly Chinese population. Chin Med J (Engl) 127(8):1445–1449
Katzman R, Zhang MY, OuangYa Q et al (1988) A Chinese version of the mini-mental state examination; impact of illiteracy in a Shanghai dementia survey. J Clin Epidemiol 41(10):971–978. https://doi.org/10.1016/0895-4356(88)90034-0
Herdman M, Gudex C, Lloyd A et al (2011) Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res 20(10):1727–1736. https://doi.org/10.1007/s11136-011-9903-x
Luo N, Liu G, Li M, Guan H, Jin X, Rand-Hendriksen K (2017) Estimating an EQ-5D-5L value set for China. Value Health 20(4):662–669. https://doi.org/10.1016/j.jval.2016.11.016
Ganhao S, Guerra MG, Lucas R et al (2022) Predictors of mortality and refracture in patients older than 65 years with a proximal femur fracture. J Clin Rheumatol 28(1):e49–e55. https://doi.org/10.1097/RHU.0000000000001581
Cai S, Fan J, Zhu L et al (2020) Bone mineral density and osteoporosis in relation to all-cause and cause-specific mortality in NHANES: a population-based cohort study. Bone 141:115597. https://doi.org/10.1016/j.bone.2020.115597
Hsu YH, Li CC, Liang FW et al (2022) Reduced all-cause mortality with bisphosphonates among post-fracture osteoporosis patients: a nationwide study and systematic review. Clin Pharmacol Ther. https://doi.org/10.1002/cpt.2645
Lambert MNT, Thybo CB, Lykkeboe S et al (2017) Combined bioavailable isoflavones and probiotics improve bone status and estrogen metabolism in postmenopausal osteopenic women: a randomized controlled trial. Am J Clin Nutr 106(3):909–920. https://doi.org/10.3945/ajcn.117.153353
Lu JX, Pan H, Hu XQ, Huang ZW, Zhang Q (2019) Effects of milk powder intervention on bone mineral density and indicators related to bone metabolism in Chinese adolescents. Osteoporos Int 30(11):2231–2239. https://doi.org/10.1007/s00198-019-05105-4
Scalco R, Trarbach E, Albuquerque EVA et al (2019) ESR1 polymorphism (rs2234693) influences femoral bone mass in patients with turner syndrome. Endocr Connect 8(11):1513–1519. https://doi.org/10.1530/EC-19-0398
Cabrerizo S, Cuadras D, Gomez-Busto F, Artaza-Artabe I, Marín-Ciancas F, Malafarina V (2015) Serum albumin and health in older people: review and meta analysis. Maturitas 81(1):17–27. https://doi.org/10.1016/j.maturitas.2015.02.009
Liu H-Y, Shyu Y-IL, Chou Y-C et al (2022) Combined effects of cognitive impairment and nutritional trajectories on functional recovery for older patients after hip-fracture surgery. J Am Med Dir Assoc 23(12):1962.e1915-1962.e1920. https://doi.org/10.1016/j.jamda.2022.08.012
Yin L, Xu Z, Wang L et al (2020) Associations of muscle size and density with proximal femur bone in a community dwelling older population. Front Endocrinol (Lausanne) 11:503. https://doi.org/10.3389/fendo.2020.00503
Cruz-Jentoft AJ, Bahat G, Bauer J et al (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48(1):16–31. https://doi.org/10.1093/ageing/afy169
Simon A, Schafer HS, Schmidt FN, Sturznickel J, Amling M, Rolvien T (2022) Compartment-specific effects of muscle strength on bone microarchitecture in women at high risk of osteoporosis. J Cachexia Sarcopenia Muscle 13(5):2310–2321. https://doi.org/10.1002/jcsm.13044
Neuman MD, Feng R, Carson JL et al (2021) Spinal anesthesia or general anesthesia for hip surgery in older adults. N Engl J Med 385(22):2025–2035. https://doi.org/10.1056/NEJMoa2113514
Okkaoglu MC, Ozdemir E, Yaradilmis U, Altay M (2022) Is radiographic osteoporotic hip morphology a predictor for high mortality following intertrochanteric femur fractures?: osteoporotic hip morphology & mortality. Injury 53(6):2184–2188. https://doi.org/10.1016/j.injury.2021.01.033
Nyholm AM, Palm H, Sandholdt H, Troelsen A, Gromov K, Dfdb C (2020) Risk of reoperation within 12 months following osteosynthesis of a displaced femoral neck fracture is linked mainly to initial fracture displacement while risk of death may be linked to bone quality: a cohort study from Danish fracture database. Acta Orthop 91(1):1–75. https://doi.org/10.1080/17453674.2019.1698503
Nguyen BN, Hoshino H, Togawa D, Matsuyama Y (2018) Cortical thickness index of the proximal femur: a radiographic parameter for preliminary assessment of bone mineral density and osteoporosis status in the age 50 years and over population. Clin Orthop Surg 10(2):149–156. https://doi.org/10.4055/cios.2018.10.2.149
Sah AP, Thornhill TS, LeBoff MS, Glowacki J (2007) Correlation of plain radiographic indices of the hip with quantitative bone mineral density. Osteoporos Int 18(8):1119–1126. https://doi.org/10.1007/s00198-007-0348-6
Pickhardt PJ (2022) Value-added opportunistic CT screening: state of the art. Radiology 303(2):241–254. https://doi.org/10.1148/radiol.211561
Boutin RD, Bamrungchart S, Bateni CP et al (2017) CT of patients with hip fracture: muscle size and attenuation help predict mortality. Am J Roentgenol 208(6):W208–W215. https://doi.org/10.2214/ajr.16.17226
Handoll HHG, Cameron ID, Mak JCS, Panagoda CE, Finnegan TP (2021) Multidisciplinary rehabilitation for older people with hip fractures. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD007125.pub3
Funding
This work is supported in part by Capital’s Funds for Health Improvement and Research (Code: 2022-1-2071), Beijing Jishuitan Hospital Nova Program (Code: XKXX2021), and the National Natural Science Foundation of China (Grant Nos. 82072445).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, and data interpretation. MY had full access to the data, take responsibility for the content, and guarantee the integrity and accuracy of the work undertaken. YG, JZ, YC, and BL performed the data collection and analysis. YG, XW, and GL did the literature search. YC, SZ, and MT did the measurements. YG wrote the manuscript, and review editing was performed by YG, MY, and GL. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
YG, YC, GL, SZ, BL, MT, JZ, WX, and MY declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
Approval was obtained from the institutional review boards of the Jishuitan Hospital, Beijing, China, and all procedures used adhere to the tenets of the Declaration of Helsinki. All participants gave written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ge, Y., Chen, Y., Liu, G. et al. Association Between Hip Bone Mineral Density and Mortality Risk After Hip Fracture: A Prospective Cohort Study. Calcif Tissue Int 113, 295–303 (2023). https://doi.org/10.1007/s00223-023-01109-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-023-01109-9