Abstract
In this sub-analysis of a comprehensive meta-analysis, we aimed to determine the effect of different types of exercise on (areal) bone mineral density (BMD) in postmenopausal women. A systematic review of the literature according to the PRISMA statement included (a) controlled trials, (b) with at least one exercise and one control group, (c) intervention ≥ 6 months, (d) BMD assessments at lumbar spine (LS), femoral neck (FN) or total hip (TH), (e) in postmenopausal women. Eight electronic databases were scanned without language restrictions up to March 2019. The present subgroup analysis was conducted as a mixed-effect meta-analysis with “type of exercise” as the moderator. The 84 eligible exercise groups were classified into (a) weight bearing (WB, n = 30) exercise, (b) (dynamic) resistance exercise (DRT, n = 18), (c) mixed WB&DRT interventions (n = 36). Outcome measures were standardized mean differences (SMD) for BMD-changes at LS, FN and TH. All types of exercise significantly affect BMD at LS, FN and TH. SMD for LS average 0.40 (95% CI 0.15–0.65) for DRT, SMD 0.26 (0.03–0.49) for WB and SMD 0.42 (0.23–0.61) for WB&DRT. SMD for FN were 0.27 (0.09–0.45) for DRT, 0.37 (0.12–0.62) for WB and 0.35 (0.19–0.51) for WB&DRT. Lastly, SMD for TH changes were 0.51 (0.28–0.74) for DRT, 0.40 (0.21–0.58) for WB and 0.34 (0.14–0.53) for WB&DRT. In summary, we provided further evidence for the favorable effect of exercise on BMD largely independent of the type of exercise. However, in order to generate dedicated exercise recommendations or exercise guideline, meta-analyses might be a too rough tool.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Exercise is considered a highly relevant component in the prevention and treatment of osteoporosis and fracture reduction [1, 2]. Consequently numerous exercise studies (review in [3]) aim to increase bone strength, predominately assessed by (areal) bone mineral density (BMD) in postmenopausal women, as the most prominent and largest cohort at risk for osteoporosis. However, although there are some evidence-based recommendations for exercise protocols [1, 4, 5], the most promising exercise to address BMD still remains unsettled [2]. Apart from exercise parameters and principles, even basic decisions, for example about the type of exercise that should be applied, is still (or once again) controversial [6, 7]. In a recent meta-analysis, Rahimi et al. [6] reported the absence of effects of resistance exercise and negative effects of weight bearing aerobic exercise on BMD at lumbar spine (LS) and femoral neck (FN) in postmenopausal cohorts 60 years and older (n = 16). Provided that these data are reliable and generalizable to the entire cohort of postmenopausal women, all the current exercise recommendations (e.g., [1, 4, 5, 8, 9]. and—even more importantly—the exercise effect on BMD in general are rendered questionable. In order to verify the findings of Rahimi et al. [6], and to estimate the effects of different roughly classified types of exercise on BMD at different regions of interest (ROI), we conducted a sub-analysis based on a recent comprehensive meta-analysis on exercise effects on BMD in postmenopausal women [3]. Similarly to Rahimi et al. [6], we roughly categorized exercises into (dynamic) resistance exercise (DRT), weight bearing (WB) exercise and combined WB&DRT exercise. Our hypotheses were that all types of exercise significantly affect BMD at (1) LS, (2) FN and (3) total hip (TH) (4), albeit without significant differences between the exercise categorizations at any BMD-ROI.
Material and Methods
The present study is based on a comprehensive systematic review of the effect of exercise on (areal) BMD in postmenopausal women [3] to which the reader is kindly referred for details.
Data Sources and Search Strategy
We strictly followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [10]; and fully registered the study in PROSPERO (CRD42018095097). Briefly eight databases (PubMed, Scopus, Web of Science, Cochrane, Science Direct, Eric, ProQuest and Primo) were searched for articles published up to March 1, 2019 without language restrictions.
The search strategy comprised a combination of population, intervention, and outcomes. Databases were systematically searched around the following combination of terms: “Bone Mineral Density”, “Exercise”, and “Postmenopausal”. Following the primary search and duplicate exclusion, the same reviewer (MS) screened studies by title and abstracts according to the eligibility criteria. A manual search in the reference lists of all included articles was carried out in an attempt to find new relevant studies. Authors of trials that were potentially eligible were contacted by e-mail for any missing data (e.g., mean change of BMD or SD) or clarification of data presented.
Inclusion and Exclusion Criteria
We included studies/study arms with (1) randomized and non-randomized controlled protocols with at least one exercise group versus one control group with sedentary/habitual active lifestyle or placebo exercise; (2) women who were postmenopausal at study start; (3) ≥ 6 months intervention duration; (4) areal BMD of the LS, femoral neck (FN) and/or total hip (tH) region at baseline and follow-up assessment as determined by (5) dual-energy X-ray absorptiometry (DXA) or dual-photon absorptiometry (DPA); (6) ≤ 10% of women on osteoanabolic/antiresorptive, or osteocatabolic (glucocorticoids) pharmaceutic agents; albeit only when the number of subjects was comparable between exercise and control.
We further excluded studies with (1) mixed gender or mixed pre- and postmenopausal cohorts without separate BMD analyses; (2) women undergoing chemo- and/or radiotherapy and (3) women with diseases that relevantly affect bone metabolism. (4) Duplicates from one study and (5) review articles, case reports, editorials, conference abstracts, and letters were not considered. Lastly, exercise study groups (see below) that cannot be classified on the intended type of exercise were also excluded from the present analysis.
Data Extraction
We designed a pre-piloted extraction form to extract relevant data. The form asked for details with respect to publication characteristics, methodology, participant characteristics, exercise characteristics, risk assessment and outcome characteristics. Two reviewers (SvS and MS) independently evaluated full-text articles and extracted data from the included studies, in case of inconsistency, a third reviewer decided (WK).
Outcome Measures
The primary outcome was change of (areal) BMD at LS-, FN- and TH-ROI as assessed by DXA or DPA between baseline and follow-up. In cases of multiple BMD assessments, we considered only changes between the baseline and final BMD assessments.
Quality Assessment
All studies included were independently assessed for risk of bias by two independent raters (WK and MV) using the Physiotherapy Evidence Database (PEDro) scale [11]. In case of inconsistency, a third reviewer decided (SvS).
Data Synthesis
For the detailed procedure to impute missing standard deviations (SD) the reader is kindly referred to the comprehensive meta-analysis of Shojaa et al. [3]. Briefly, if the studies presented a confidence interval (CI) or standard errors (SE), they were converted to SD. In cases of missing CI or SE data we first contacted authors (n = 11) to provide corresponding information. When no reply was received or data were not available, the exact p-value of the absolute change of BMD was obtained to compute the SD of the change. In the case of unreported p-value, we calculated the SDs using pre and post SDs.
In order to determine the effects of different types of exercise we categorized the studies according to the following approach: (a) dynamic resistance exercise, i.e., any kind of resistance exercise that involves joint movement to develop musculoskeletal strength. We focus on studies that applied isolated DRT without any adjuvant exercise component and without bone-specific warm ups (e.g., running, hopping, aerobic dance) with validated effect on bone [1, 4, 5], (b) weight bearing exercise that involved any kind of aerobic and anaerobic loading of axial skeletal sites due to gravity, i.e., Tai Chi, walking, running, dancing, movement games, heel drops, hopping, jumping and (c) exercise studies that combined weight bearing and DRT exercise, even if WB exercise was applied only shortly during warm up. The latter approach was selected due to the observation that only few cycles with high strain rates may induce positive effects on bone [12, 13]. Two raters (WK and MV) independently categorized the data, in case of inconsistency, a third reviewer decided (SvS).
Statistical Analysis
The statistical analysis was performed using the statistical software R (R Development Core Team) [14]. Effect size (ES) value was considered as the standardized mean differences (SMDs) combined with the 95% confidence interval (95% CI). Random-effects meta-analysis was performed by applying the metafor package [15]. Heterogeneity for between-study variability was determined using the Cochran Q test, as with other statistical analyses a P-value < 0.05 was considered significant. The level of heterogeneity was analyzed with the I2 statistic. For those studies with two different intervention groups, the control group was proportionally split into two groups for comparison against each intervention group [16]. Sensitivity analysis was conducted to check whether the overall result of the analysis is robust regarding the use of the imputed SDs. Funnel plots with regression test and the rank correlation between effect estimates and their standard errors, using the t-test and Kendall’s τ statistic respectively, were applied to explore potential publication bias. To adjust the results for possible publication bias, we also conducted a trim and fill analysis using the L0 estimator proposed by Duval et al. [17]. The present subgroup analysis was conducted as a mixed-effect meta-analysis with “type of exercise” as the moderator. A P-value of < 0.05 was considered as significant for all tests.
Results
Study Characteristics
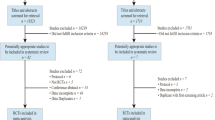
In total, our search identified 74 eligible studies with 84 exercise groups (Fig. 1), categorized into 18 DRT groups [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32], 30 weight bearing (WB) type exercise groups [31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57] and 36 study groups that scheduled combined exercise protocols [33, 48, 58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88].
Flow diagram of search process according to PRISMA [10]
The pooled number of participants was 2793 in the exercise and 2319 in the control group respectively. In detail, the number of participants in exercise and control was 1344 and 1175 women in the combined WB DRT group, 1045 and 815 women in the WB and 404 and 329 women in the DRT group. Table 1 gives the anthropometric participant characteristics of the included studies.
Sample sizes in the exercise arms ranged from 5 [28] to 125 participants [58] per group (CG: 2 to 125 women). Thirteen studies [22, 33, 41, 45, 47, 61, 63, 68, 73, 75, 77, 87, 89] included women with osteopenia/osteoporosis (DRT: n = 1 vs. WB: n = 4 vs. WB&DRT: n = 8); (Table 2). Average age varied among the studies between 51 ± 2 years [18] and 77 ± 3 years [81]. Twelve studies with fourteen exercise groups [18, 24, 28, 38, 55, 57, 69, 72, 75] focused on cohorts of “early postmenopausal women” (1 to ≤ 8 years post). Thirty studies included participants with sedentary/habitually active lifestyles, 28 trials involved participants with exercise activities presumably with minor effects on bone and 16 studies did not provide the corresponding information (Table 2).
Intervention Characteristics
Cholecalciferol, Calcium Supplementation
Vitamin-D and/or calcium supplementation for the exercise and control groups were provided in 21 studies [18, 19, 21, 29, 33,34,35, 40, 41, 43, 44, 49, 51, 65, 71, 73, 76, 77, 79, 80, 84].
Exercise Intervention
Table 2 gives exercise characteristics of the included studies categorized according the type of exercise.
Dynamic Resistance Exercise (DRT)
Table 2 (midsection) specifies the exercise protocols of included 18 included DRT study groups.
Apart from one exception [30], all studies focused on a supervised group exercise protocol [18,19,20,21,22,23,24,25,26,27,28,29, 31, 32, 60]. Studies ranged from six [18, 20, 22, 26] to 24 months [30]. Except for three studies/study groups [24, 30, 32], the studies focused on all or most main muscle groups. Besides two studies that did not provide sufficient information for the LS-site [23, 32], all the other studies provided exercises for their specified BMD-ROI (i.e., LS and proximal femur). Most studies prescribed a training frequency of three sessions per week (Table 2); with a session length that varied from about 1–2 min (i.e., 10 × back extension [30]) to ≈ 120 min (39 sets × 20 reps, 2–3 min of rest [23]). Most studies applied a multiple set approach ( [18, 19, 21,22,23,24,25, 27, 29, 43, 60]. Relative exercise intensity ranged between 80% 1RM [18, 23, 25, 27, 29, 43, 60] and ≤ 30% 1RM [26, 30]. Five studies prescribed either work to repetition maximum [27, 28, 80] or work to muscular fatigue [21, 43], another study [20] referred to 5–6 (i.e., strong-strong +) on the Borg CR10 scale. Reviewing the repetition number and relative exercise intensity (% 1RM), some studies [30] or study arms [18, 29] might have exercised with (too) low effort.Footnote 1 None of the studies reported an explosive movement in the concentric or eccentric phase. Progression or at least regular adjustments of exercise intensity were realized by all but one DRT study. Periodization models [90] were not applied by any of the studies.
Weight Bearing Exercise
Table 2 (upper part) lists the exercise protocol of the weight bearing type exercise studies. By nature, the specific exercise was much more heterogeneous compared with DRT. Studies specified (brisk) walking including walking with additional load (n = 11), walking/running (n = 3), Tai Chi (n = 4), jumping or rope skipping (n = 3), heel drops (n = 1), stepping (n = 1), standing on one leg (n = 1) and combined weight bearing types (e.g., heel drops, jumping skipping; stairclimbing, n = 6) (Table 2). Duration of the studies varied between 6 (e.g., [33].) and 30 months [45]. Twelve of 30 study groups applied a supervised group exercise program, 12 study groups specified non-supervised individual exercise [36, 37, 39, 41, 42, 44, 47, 53, 55, 89] or additional [34, 35, 45, 51, 89] to the supervised group exercise, for 6 study groups this information was not listed (Table 2). Site specificity at the LS might be realized by direct muscular insertion of exercises applied in the Tai Chi studies [31, 32, 38, 47] and studies that applied higher ground reaction forces (i.e., jumping, drop-jumps and potentially jogging in older cohorts) [33,34,35, 40, 41, 43,44,45,46, 52, 55, 56, 83]. Net training frequency (considering attendance rate) varied between ≥ 10 sessionsFootnote 2 and about 2 sessions/week [55]; corresponding net exercise volume/week vary between ≥ 240 [89] and about 10–15 min/week [55]. Progression of exercise intensity was consciously considered by about half of the WB type exercise studies [40, 41, 43,44,45,46, 49, 52, 56, 83] (Table 2). Periodized exercise models were not applied.
Combined WB and DRT Studies
Most combined WB DRT studies applied a combination of walking, running, stepping, movement games, dancing either as single session, session component or during warm up and a DRT on machines or with free weights (Table 2, lower part). At least nine study arms [63, 69, 72, 76, 77, 79, 82, 84, 91] specified exercises with higher GRF (e.g., jumping variations, heel drops) during the WB&DRT sessions. With few exceptions [73], the studies scheduled either a consistently supervised exercise protocol or a mix of supervised (DRT) sessions and non-supervised walking/home training sessions (Table 2). Duration of the studies varied between 6 (e.g., [33, 58].) and 26 months [77]. Training frequency varied from ≈ 8 [58] to < 2 sessions/week [70, 82, 83]; net training volume ranged from about 6 h/week [58] to 67 min/week [70]. Due to the overall DRT (i.e., all or most main muscle groups) that was applied by all, but 3 studies [58, 72, 88], most studies mechanically addressed the LS-ROI by muscular tension. Apart from one study [66], all studies that adequately described their exercise protocols applied multiple set approaches. Peak relative exercise intensity varied between 90% 1RM [77] and 60–65% 1RM [76]. Progression of exercise intensity was applied by the vast majority of the studies [48, 62, 63, 65,66,67,68,69,70,71, 73, 74, 76,77,78,79,80,81, 83,84,85, 88].
Methodologic Quality
The Pedro scores of the included studies are listed in Table 3. Methodologic quality of the trials ranges from 3 to 9 score points (Table 3), with a mean and SD of 5.44 ± 1.32 score points. Methodologic quality of the DRT studies was on average (6.24 ± 1.30 points) significantly higher (P = 0.024) compared with the other groups.
Outcomes
Apart from two studies [28, 30] that applied DPA, all the others used DXA. Furthermore, all the other studies except two ( [23]: hip only; [30]: LS only) determined both, BMD at LS and proximal femur regions of interest.
Effect of Different Types of Exercise on LS-BMD
Sixteen DRT exercise groups, 26 WB exercise groups and 33 combined WB&DRT exercise groups evaluated the effect of exercise on LS-BMD. In summary, the pooled estimate of random effect analysis for DRT was SMD: 0.40, 95% CI 0.15–0.65 (P = 0.009), for WB exercise SMD: 0.26, 95% CI: 0.03–0.49 (P = 0.037) and SMD: 0.42, 95% CI 0.23–0.61 (P = 0.001) for the combined WB&DRT exercise. No significant differences between the types of exercise were observed (P = 0.508). All types of exercise revealed a similarly high level of heterogeneity between their trials (I2 = 76.3–76.5%)) (Fig. 2).
Effect of Different Types of Exercise on FN-BMD
Fifteen DRT exercise groups, 23 WB exercise groups and 25 combined WB&DRT exercise groups evaluated the effect of exercise on femoral neck-BMD. In summary, the pooled estimate of random effect analysis for DRT was SMD: 0.27, 95% CI 0.09–0.45 (P = 0.003), for WB exercise SMD: 0.37, 95% CI 0.12–0.62 (P = 0.004) and SMD: 0.35, 95% CI 0.19–0.51 (P = 0.001) for the combined WB&DRT exercise. No significant differences between the types of exercise were observed (P = 0.822). Heterogeneity level of included trials in the WB and WB&DRT group was considerable (I2: 82.1) or substantial (I2: 63.6); but was negligible (I2: 16.5) in the DRT group (Fig. 3).
Effect of Different Types of Exercise on TH-BMD
Ten DRT exercise groups, seven WB exercise groups and 12 combined WB&DRT exercise groups evaluated the effect of exercise on total hip-BMD. In summary, the pooled estimate of random effect analysis for DRT was SMD: 0.51, 95% CI 0.28–0.74 (P < 0.001), for WB exercise SMD: 0.40, 95% CI 0.21–0.58 (P < 0.001) and SMD: 0.34, 95% CI 0.14–0.53 (P < 0.001) for the combined WB&DRT exercise. No significant differences between the types of exercise were observed (P = 0.554). Heterogeneity level of included trials in the WB or DRT group was negligible (I2 < 10%) and moderate (I2: 43.8%) in the WB&DRT group (Fig. 4).
Funnel plots for LS, FN and TH did not suggest positive evidence of publication bias. The regression and rank correlation test for funnel plot asymmetry did not indicate significant asymmetry for LS or TH, but did for TH with missing studies to the right (positive difference/effects). The trim and fill analysis that correspondingly imputed three studies results in a slightly higher total SMD (0.43; 95% CI 0.31–0.54) than the non-adjusted results listed in Fig. 4.
Discussion
In this sub-analysis of a comprehensive meta-analysis, we clearly confirmed the significant positive effects of different types of exercise on BMD at LS, FN and TH in postmenopausal women. Further, WB type exercises, DRT and a combination of both types of exercise revealed at least no significant groups differences for LS, FN or TH-BMD. Thus, we verified all our hypothesis and in turn now question the data of Rahimi et al. [6]. One possible explanation for the diverging results of the present analysis and the data of Rahimi et al. [6] might be the focus on studies with women 60 years + , i.e., the advanced postmenopausal status in the latter study. Considering that menopausal transition and early menopausal status is related to considerably increased bone turnover [92, 93], there is some evidence that exercise might be more effective during early than in late post-menopause, at least with respect to trabecular bone loss [76, 94]. The meta-analysis of Shojaa et al. [3] on this issue observed only slight, non-significant differences between exercise during the early vs. late postmenopausal years,Footnote 3 be it for LS (SMD “early”: MV = 0.64, 95% CI 0.33–0.95 vs. “late”: 0.39, 0.14–0.55) or total hip ROI (SMD: 0.51, 0.27–75 vs. 0.38, 0.20–0.56). Apart from age, both meta-analyses also differ with respect to eligibility criteria, i.e., randomization, language, publication type, medication and diseases, while the limitation on studies ≥ 6 months with healthy postmenopausal women without hormone replacement therapy and previous DRT are common to both studies. The most striking difference, on the other hand, is the low amount of studies classified into the exercise categories by Rahimi et al. [6]. Considering that only two studies were analyzed to determine the effect of WB aerobic exercise on LS-BMD (vs. n = 23 in the present study), one should draw definite conclusions from that data with extreme caution.
Although we consistently determined significant positive exercise effects on BMD-ROIs, (SMD: 0.26–0.51), SMDs of the single exercise trial vary substantially, particularly for the LS (I2 = 76–77%). Even in the DRT group, which can be considered as the most homogeneous group with respect to exercise type classification (see above), the heterogeneity level for LS-BMD effects was substantial (i.e., I2 > 75%). This is understandable, however, since considerable differences can be observed between the trials or study groups (Table 2) particularly with respect to exercise parameters (i.e., strain magnitude, rate [5]) and training principles (e.g., progression, periodization [5, 95]).
Revisiting the effects of different types of exercise, it is noteworthy that the effect of the WB type interventions at the LS was considerably less pronounced compared with the DRT group (SMD: 0.26 versus SMD: 0.40). This is not necessarily related to higher effects of DRT-induced direct muscular impact on LS in general, however, but to the large number of WB studies that applied low ground reaction forces (e.g., walking: n = 11) with corresponding axial impact loading that might not (longer) reach the LS area. Two meta-analyses [96, 97] that reported significant positive “walking effects” at FN-BMD without effects at LS-BMD support this estimation. Another surprising result is that the combined effect of WB&DRT group failed to generate relevantly higher BMD effects compared with DRT (…or apart from LS-BMD, WB type exercise). Recent evidence-based guidelines that focus on bone development [1, 4, 5] consistently recommended exercise protocols that included impact activities and progressive resistance training applied with high strain magnitude and rate. However, at this point at the latest, we have to acknowledge and discuss the limited ability of meta-analyses to derive exercise recommendations [98], largely independent of the outcome [99]. Selecting the adequate type of exercise to address a given training aim is only the first, rough decision within the training process [5, 95]. Much more challenging, particularly when addressing bone, is the consideration how to optimally specify the type of exercise in the light of the large variety of exercise parameters (e.g., strain magnitude, rate, duration, frequency, cycle number, rest periods) [5, 95]. Another modifying aspect within the exercise process is the inclusion of exercise principles [5, 95]. Applying, e.g., progression and periodization might not be important within a 10-week exercise intervention; however, considering that studies included in the present analysis on BMD average between ≥ 6 months and 30 months their relevance becomes obvious. The fact that even slight differences in exercise parameters, e.g., movement velocity of the concentric phase during DRT, significantly modify the effect on BMD [100] suggests that high complexity of exercise effects on BMD could conflict with the comprehensive meta-analytic approach. One may assume that the rather high number of study groups included in the present subgroups might even out differences at individual study levels, but this assumption is frequently wide of the mark. This might be confirmed by the considerably higher effects of DRT versus WB for TH-BMD (SMD: 0.51, 95% CI 0.28–0.74 vs. 0.34, 0.14–0.52), however, not for BMD at the adjacent FN-region (SMD-DRT: 0.27, 0.09–0.45 versus SMD-WB: 0.37, 0.12–0.62, Table 2), a constellation for which no serious explanationFootnote 4 can be provided.
Furthermore, some limitations and study features of the present analysis may decrease the evidence and generality of our finding. (1) Although we placed high emphasis on eligibility and reliable classification of the exercise types, some decisions are certainly debatable. This may be the case for the exclusion of the study of Rhodes et al. [101]Footnote 5 that combined non-weight bearing exercise (however only as a warm up) and DRT, while still including others (e.g., [66, 67, 87]. that applied a mixed weight bearing/non-weight bearing & DRT intervention. However, in our defense it should be noted that some studies were very lapse in their standards of exercise reporting, and so extracting the relevant information was sometimes challenging. (2) We conducted funnel plots with trim and fill analysis for the entire cohort of included studies for LS, FN and TH (not given). However, it might have been better to conduct separate funnel plots for the effects of the isolated exercise group for each ROI. On the other hand, reviewing the three funnel plots in detail, we did not observe relevant differences between the different exercise groups that might have significantly changed the present result. (3) We failed to generate reliable scores/categories for exercise intensity/strain magnitude across the different types of exercises, in order to conduct a sub-analysis for this crucial exercise parameters. A sub-analysis of our outcome adjusted for “exercise intensity/strain magnitude” might have resulted in more sophisticated results and higher overall treatment effects. (4) The present literature search was conducted up to March 1, 2019, i.e., some more studies might have been published in the meantime. However, considering the large amount of studies included in this systematic review and meta-analysis, we feel that the few additional exercise studies will not considerably modify our finding.
In conclusion, we do not share the enthusiasm for basing exercise recommendations or exercise guidelines on meta-analyses – at least in the area of “bone strengthening”. Nonetheless, at least uncritical acceptance of the acquired data should be avoided if this is done. Accurately designed randomized controlled exercise trials that manipulate a dedicated single aspect while maintaining all other exercise parameters and confounders will be more qualified to generate reliable exercise recommendations.
Data Availability
The data that support the findings of this study are available from the corresponding author (WK), upon reasonable request.
Notes
Or two sessions of 3 min/day, 7 days/week [53].
Early: ≤ 8 years vs. late postmenopausal > 9 years postmenopausal for LS-BMD (LS-BMD: 40, total hip: 20 exercise groups).
Considering site specificity of most DRT-exercises, we would expect the opposite effect, however, differences were not significant.
…or Watson et al. [102]. This highly effective and time-efficient trial had to be excluded by protocol due to the number of participants (< 10%) on osteoporosis medication.
References
Beck BR, Daly RM, Singh MA, Taaffe DR (2016) Exercise and Sports Science Australia (ESSA) position statement on exercise prescription for the prevention and management of osteoporosis. J Sci Med Sport 20:438–445
Tarantino U, Iolascon G, Cianferotti L et al (2017) Clinical guidelines for the prevention and treatment of osteoporosis: summary statements and recommendations from the Italian Society for Orthopaedics and Traumatology. J Orthop Traumatol 18:3–36
Shojaa N, von Stengel S, Schoene D et al (2020) Effect of exercise training on bone mineral density in postmenopausal women: a systematic review and meta-analysis of intervention studies. Front Physiol 11:1427–1444
Daly RM, Dalla Via J, Duckham RL, Fraser SF, Helge EW (2019) Exercise for the prevention of osteoporosis in postmenopausal women: an evidence-based guide to the optimal prescription. Braz J Phys Ther 23:170–180
Kemmler W, Stengel V (eds) (2019) The role of exercise on fracture reduction and bone strengthening. Avademic Press, London
Rahimi GRM, Smart NA, Liang MT et al (2020) The impact of different modes of exercise training on bone mineral density in older postmenopausal women: a systematic review and meta-analysis research. Calcif Tissue Int 106(6):577–590
Shojaa M, von Stengel V, Kohl M, Schoene D, Kemmler W (2020) Effects of dynamic resistance exercise on bone mineral density in postmenopausal women—a systematic review and meta-analysis with special emphasis to exercise parameters. Osteo Int 31:1427–1444
Felsenberg D, Dietzel R, Pthy M, et al. (2008) Leitlinie physiotherapie und bewegungstherapie bei osteoporose. Guideline long version:
Kohrt WM, Bloomfield SA, Little KD, Nelson ME, Yingling VR (2004) American College of Sports Medicine position stand: physical activity and bone health. Med Sci Sports Exerc 366:1985–1996
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(264–269):W264
Sherrington C, Herbert RD, Maher CG, Moseley AM (2000) PEDro. A database of randomized trials and systematic reviews in physiotherapy. Man Ther 5:223–226
Kato T, Terashima T, Yamashita T, Hatanaka Y, Honda A, Umemura Y (2006) Effect of low-repetition jump training on bone mineral density in young women. J Appl Physiol 100:839–843
Umemura Y, Ishiko T, Tsujimoto H, Miura H, Mokushi N, Suzuki H (1995) Effects of jump training on bone hypertrophy in young and old rats. Int J Sports Med 16:364–367
R_Development_Core_Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor package. J Stat Softw 36:1–48
Cochrane (2016) Cochrane handbook for systematic reviews of interventions. In Higgins J, Green S (eds). The Cochrane Collaboration
Duval SJ, Tweedie RL (2000) A nonparametric "trim and fill" method of accounting for publication bias in meta-analysis. JASA 95:89–98
Bemben DA, Fetters NL, Bemben MG, Nabavi N, Koh ET (2000) Musculoskeletal responses to high- and low-intensity resistance training in early postmenopausal women. Med Sci Sports Exerc 32:1949–1957
Chilibeck PD, Davison KS, Whiting SJ, Suzuki Y, Janzen CL, Peloso P (2002) The effect of strength training combined with bisphosphonate (etidronate) therapy on bone mineral, lean tissue, and fat mass in postmenopausal women. Can J Physiol Pharmacol 80:941–950
de Oliveira LC, de Oliveira RG, de Almeida Pires-Oliveira DA (2019) Effects of whole-body vibration versus pilates exercise on bone mineral density in postmenopausal women: a randomized and controlled clinical trial. J Geriatr Phys Ther 42:E23–E31
Duff WR, Kontulainen SA, Candow DG et al (2016) Effects of low-dose ibuprofen supplementation and resistance training on bone and muscle in postmenopausal women: a randomized controlled trial. Bone Rep 5:96–103
Hartard M, Haber P, Ilieva D, Preisinger E, Huber JC (1996) Systematic strength training as a model of therapeutic intervention. Arch Phys Med Rehabil 75:21–28
Kerr D, Morton A, Dick I, Prince R (1996) Exercise effects on bone mass in postmenopausal women are site-specific and load-dependent. J Bone Miner Res 11:218–225
Maddalozzo GF, Widrick JJ, Cardinal BJ, Winters-Stone KM, Hoffman MA, Snow CM (2007) The effects of hormone replacement therapy and resistance training on spine bone mineral density in early postmenopausal women. Bone 40:1244–1251
Nelson ME, Fiatarone MA, Morganti CM, Trice I, Greenberg RA, Evans WJ (1994) Effects of high-intensity strength training on multiple risk factors for osteoporotic fractures: a randomized controlled trial. JAMA 272:1909–1914
Nicholson VP, McKean MR, Slater GJ, Kerr A, Burkett BJ (2015) Low-load very high-repetition resistance training attenuates bone loss at the lumbar spine in active post-menopausal women. Calcif Tissue Int 96:490–499
Orsatti F, Petri-Nahas E, Nahas-Neto J, Orsatti C, Teixeira A (2013) Effects of isoflavone and counter-resistance training on bone mineral density in postmenopausal women. Rev Bras Cineantropom Desempenho Hum 15:726–736
Pruitt LA, Jackson RD, Bartels RL, Lehnhard HJ (1992) Weight-training effects on bone mineral density in early postmenopausal women. J Bone Miner Res 7:179–185
Pruitt LA, Taaffe DR, Marcus R (1995) Effects of a one-year high-intensity versus low-intensity resistance training program on bone mineral density in older women. J Bone Miner Res 10:1788–1795
Sinaki M, Wahner HW, Offord KP, Hodgson SF (1989) Efficacy of nonloading exercises in prevention of vertebral bone loss in postmenopausal women: a controlled trial. Mayo Clin Proc 64:762–769
Wang H, Yu B, Chen W, Lu Y, Yu D (2015) Simplified Tai Chi resistance training versus traditional Tai Chi in slowing bone loss in postmenopausal women. Evid Based Complement Alternat Med 2015:379451
Woo J, Hong A, Lau E, Lynn H (2007) A randomised controlled trial of Tai Chi and resistance exercise on bone health, muscle strength and balance in community-living elderly people. Age Ageing 36:262–268
Basat H, Esmaeilzadeh S, Eskiyurt N (2013) The effects of strengthening and high-impact exercises on bone metabolism and quality of life in postmenopausal women: a randomized controlled trial. J Back Musculoskelet Rehabil 26:427–435
Bassey EJ, Ramsdale SJ (1995) Weight-bearing exercise and ground reaction forces: a 12-month randomized controlled trial of effects on bone mineral density in healthy postmenopausal women. Bone 16:469–476
Bassey EJ, Rothwell MC, Littlewood JJ, Pye DW (1998) Pre- and postmenopausal women have different bone mineral density responses to the same high-impact exercise. J Bone Miner Res 13:1805–1813
Brooke-Wavell K, Jones PR, Hardman AE (1997) Brisk walking reduces calcaneal bone loss in post-menopausal women. Clin Sci (Lond) 92:75–80
Brooke-Wavell K, Jones PR, Hardman AE, Tsuritan YY (2001) Commencing, continuing and stopping brisk walking: effects on bone mineral density, quantitative ultrasound of bone and markers of bone metabolism in postmenopausal women. Osteoporos Int 12:581–587
Chan K, Qin L, Lau M et al (2004) A randomized, prospective study of the effects of Tai Chi Chun exercise on bone mineral density in postmenopausal women. Arch Phys Med Rehabil 85:717–722
Ebrahim SB, Thompson PW, Baskaran V (1997) Randomized placebo controlled trial of brisk walking in the prevention of postmenopausal osteoporosis. Age Aging 26:252–260
Evans EM, Racette SB, Van Pelt RE, Peterson LR, Villareal DT (2007) Effects of soy protein isolate and moderate exercise on bone turnover and bone mineral density in postmenopausal women. Menopause 14:481–488
Hans D, Genton L, Drezner MK et al (2002) Monitored impact loading of the hip: initial testing of a home-use device. Calcif Tissue Int 71:112–120
Hatori M, Hasegawa A, Adachi H et al (1993) The effects of walking at the anaerobic threshold level on vertebral bone loss in postmenopausal women. Calcif Tissue Int 52:411–414
Kohrt WM, Ehsani AA (1997) Effects of exercise involving predominantly either joint-reaction or ground-reaction forces on bone mineral density in older women. J Bone Miner Res 12:1253–1261
Kohrt WM, Snead DB, Slatopolsky E, Birge SJ Jr (1995) Additive effects of weight-bearing exercise and estrogen on bone mineral density in older women. J Bone Miner Res 10:1303–1311
Korpelainen R, Keinanen-Kiukaanniemi S, Heikkinen J, Vaananen K, Korpelainen J (2006) Effects of impact exercise on bone mineral density in elderly women with low BMD: a population based randomized controlled 30-month intervention. Osteoporos Int 17:109–118
Lau EM, Woo J, Leung PC, Swaminathan R, Leung D (1992) The effects of calcium supplementation and exercise on bone density in elderly Chinese women. Osteoporos Int 2:168–173
Liu BX, Chen SP, Li YD et al (2015) The effect of the modified eighth section of eight-section brocade on osteoporosis in postmenopausal women: a prospective randomized trial. Medicine (Baltimore) 94:e991
Marques EA, Wanderley F, Machado L et al (2011) Effects of resistance and aerobic exercise on physical function, bone mineral density, OPG and RANKL in older women. Exp Gerontol 46:524–532
Martin D, Notelovitz M (1993) Effects of aerobic training on bone mineral density of postmenopausal women. J Bone Miner Res 8:931–936
Nelson ME, Fisher EC, Dilmanian FA, Dallal GE, Evans WJ (1991) A 1-y walking program and increased dietary calcium in postmenopausal women: effects on bone. Am J Clin Nutr 53:1304–1311
Prince RL, Devine A, Dick I et al (1995) The effect of calcium supplementation (milk powder or tablets) and exercise on bone density in postmenopausal women. J Bone Miner Res 10:1068–1075
Ryan AS, Nicklas BJ, Dennis KE (1998) Aerobic exercise maintains regional bone mineral density during weight loss in postmenopausal women. J Appl Physiol 84:1305–1310
Sakai A, Oshige T, Zenke Y, Yamanaka Y, Nagaishi H, Nakamura T (2010) Unipedal standing exercise and hip bone mineral density in postmenopausal women: a randomized controlled trial. J Bone Miner Metab 28:42–48
Silverman NE, Nicklas BJ, Ryan AS (2009) Addition of aerobic exercise to a weight loss program increases BMD, with an associated reduction in inflammation in overweight postmenopausal women. Calcif Tissue Int 84:257–265
Sugiyama T, Yamaguchi A, Kawai S (2002) Effects of skeletal loading on bone mass and compensation mechanism in bone: a new insight into the "mechanostat" theory. J Bone Miner Metab 20:196–200
Tartibian B, Hajizadeh Maleki B, Kanaley J, Sadeghi K (2011) Long-term aerobic exercise and omega-3 supplementation modulate osteoporosis through inflammatory mechanisms in post-menopausal women: a randomized, repeated measures study. Nutr Metab (Lond) 8:71
Wu J, Oka J, Tabata I et al (2006) Effects of isoflavone and exercise on BMD and fat mass in postmenopausal Japanese women: a 1-year randomized placebo-controlled trial. J Bone Miner Res 21:780–789
Adami S, Gatti D, Braga V, Bianchini D, Rossini M (1999) Site-specific effects of strength training on bone structure and geometry of ultradistal radius in postmenopausal women. J Bone Miner Res 14:120–124
Bello M, Sousa MC, Neto G, Oliveira L, Guerras I, Mendes R, Sousa N (2014) The effect of a long-term, community-based exercise program on bone mineral density in postmenopausal women with pre-diabetes and type 2 diabetes. J Hum Kinet 43:43–48
Bemben DA, Palmer IJ, Bemben MG, Knehans AW (2010) Effects of combined whole-body vibration and resistance training on muscular strength and bone metabolism in postmenopausal women. Bone 47:650–656
Bergström I, Landgren BM, Brinck J, Freyschuss B (2008) Physical training preserves bone mineral density in postmenopausal women with forearm fractures and low bone mineral density. Osteoporos Int 19:177–183
Bocalini DS, Serra AJ, dos Santos L, Murad N, Levy RF (2009) Strength training preserves the bone mineral density of postmenopausal women without hormone replacement therapy. J Aging Health 21:519–527
Bolton KL, Egerton T, Wark J et al (2012) Effects of exercise on bone density and falls risk factors in post-menopausal women with osteopenia: a randomised controlled trial. J Sci Med Sport 15:102–109
Caplan GA, Ward JA, Lord SR (1993) The benefits of exercise in postmenopausal women. Aust J Public Health 17:23–26
Chilibeck PD, Vatanparast H, Pierson R et al (2013) Effect of exercise training combined with isoflavone supplementation on bone and lipids in postmenopausal women: a randomized clinical trial. J Bone Miner Res 28:780–793
Choquette S, Riesco E, Cormier E, Dion T, Aubertin-Leheudre M, Dionne IJ (2011) Effects of soya isoflavones and exercise on body composition and clinical risk factors of cardiovascular diseases in overweight postmenopausal women: a 6-month double-blind controlled trial. Br J Nutr 105:1199–1209
Chuin A, Labonte M, Tessier D et al (2009) Effect of antioxidants combined to resistance training on BMD in elderly women: a pilot study. Osteoporos Int 20:1253–1258
de Matos O, Lopes da Silva DJ, Martinez de Oliveira J, Castelo-Branco C (2009) Effect of specific exercise training on bone mineral density in women with postmenopausal osteopenia or osteoporosis. Gynecol Endocrinol 25:616–620
Deng S (2009) Effects of exercise therapy on bone mineral density in early postmenopausal women: a controlled trial. Front Med China 3:323–329
Englund U, Littbrand H, Sondell A, Pettersson U, Bucht G (2005) A 1-year combined weight-bearing training program is beneficial for bone mineral density and neuromuscular function in older women. Osteoporos Int 16:1117–1123
Going S, Lohman T, Houtkooper L et al (2003) Effects of exercise on bone mineral density in calcium-replete postmenopausal women with and without hormone replacement therapy. Osteoporos Int 14:637–643
Grove KA, Londeree BR (1992) Bone density in postmenopausal women: high impact vs low impact exercise. Med Sci Sports Exerc 24:1190–1194
Iwamoto J, Takeda T, Ichimura S (2001) Effects of exercise training and detraining on bone mineral density in postmenopausal women with osteoporosis. J Orthop Sci 6:128–132
Jessup JV, Horne C, Vishen RK, Wheeler D (2003) Effects of exercise on bone density, balance, and self-efficacy in older women. Biol Res Nurs 4:171–180
Karakiriou S, Douda H, Smilios I, Volaklis KA, Tokmakidis SP (2012) Effects of vibration and exercise training on bone mineral density and muscle strength in post-menopausal women. Eur J Sport Sci 12:81–88
Kemmler W (1999) Einfluß unterschiedlicher Lebensabschnitte auf die belastungsabhängige Reaktion ossärer Risikofaktoren einer Osteoporose. Dtsch Z Sportmed 50:114–119
Kemmler W, Lauber D, Weineck J, Hensen J, Kalender W, Engelke K (2004) Benefits of 2 years of intense exercise on bone density, physical fitness, and blood lipids in early postmenopausal osteopenic women: results of the Erlangen Fitness Osteoporosis Prevention Study (EFOPS). Arch Intern Med 164:1084–1091
Kemmler W, von Stengel S, Engelke K, Haberle L, Kalender WA (2010) Exercise effects on bone mineral density, falls, coronary risk factors, and health care costs in older women: the randomized controlled senior fitness and prevention (SEFIP) study. Arch Intern Med 170:179–185
Kemmler W, Bebenek M, von Stengel S, Engelke K, Kalender WA (2013) Effect of block-periodized exercise training on bone and coronary heart disease risk factors in early post-menopausal women: a randomized controlled study. Scand J Med Sci Sports 23:121–129
Kerr D, Ackland T, Maslen B, Morton A, Prince R (2001) Resistance training over 2 years increases bone mass in calcium-replete postmenopausal women. J Bone Miner Res 16:175–181
Kwon Y, Park SK, Kim E, Park J (2008) The effects of multi-component exercise training on VO2max, muscle mass, whole bone mineral density and fall risk in community-dwelling elderly women. Jpn J Phys Fit Sport 57:339–348
Lord SR, Ward JA, Williams P, Zivanovic E (1996) The effect of a community exercise program on fracture risk factors in older women. Osteoporos Int 6:361–367
Marques EA, Mota J, Machado L, Sousa F, Coelho M, Moreira P, Carvalho J (2011) Multicomponent training program with weight-bearing exercises elicits favorable bone density, muscle strength, and balance adaptations in older women. Calcif Tissue Int 88:117–129
Milliken LA, Going SB, Houtkooper LB et al (2003) Effects of exercise training on bone remodeling, insulin-like growth factors, and bone mineral density in postmenopausal women with and without hormone replacement therapy. Calcif Tissue Int 72:478–484
Nichols JF, Nelson KP, Peterson KK, Satoris DJ (1995) Bone Mineral density responses to high-intensity strength training in older women. J Age Phys Activity 3:26–38
Park H, Kim KJ, Komatsu T, Park SK, Mutoh Y (2008) Effect of combined exercise training on bone, body balance, and gait ability: a randomized controlled study in community-dwelling elderly women. J Bone Miner Metab 26:254–259
Tolomio S, Lalli A, Travain G, Zaccaria M (2009) Effects of a combined weight- and non weight-bearing (water) exercise program on bone mass and quality in postmenopausal women with low bone mineral density. Clin Ter 160:105–109
Verschueren SM, Roelants M, Delecluse C, Swinnen S, Vanderschueren D, Boonen S (2004) Effects of 6-month whole body vibration training on hip density; muscle strength, and postural control in postmenopausal women: a randomized controlled pilot study. J Bone Min Res 19:352–359
Yamazaki S, Ichimura S, Iwamoto J, Takeda T, Toyama Y (2004) Effect of walking exercise on bone metabolism in postmenopausal women with osteopenia/osteoporosis. J Bone Miner Metab 22:500–508
Hoover DL, VanWye WR, Judge LW (2016) Periodization and physical therapy: bridging the gap between training and rehabilitation. Phys Ther Sport 18:1–20
Marques EA, Mota J, Carvalho J (2011) Exercise effects on bone mineral density in older adults: a meta-analysis of randomized controlled trials. Age 34:1493–1515
Tella SH, Gallagher JC (2014) Prevention and treatment of postmenopausal osteoporosis. J Steroid Biochem Mol Biol 142:155–170
Zaidi M, Turner CH, Canalis E et al (2009) Bone loss or lost bone: rationale and recommendations for the diagnosis and treatment of early postmenopausal bone loss. Curr Osteoporos Rep 7:118–126
Beck BR, Snow CM (2003) Bone health across the lifespan–exercising our options. Exerc Sport Sci Rev 31:117–122
Kemmler W, von Stengel S (2011) Exercise and osteoporosis-related fractures: perspectives and recommendations of the sports and exercise scientist. Phys Sportmed 39:142–157
Ma D, Wu L, He Z (2013) Effects of walking on the preservation of bone mineral density in perimenopausal and postmenopausal women: a systematic review and meta-analysis. Menopause 20:1216–1226
Martyn-St James M, Carroll S (2008) Meta-analysis of walking for preservation of bone mineral density in postmenopausal women. Bone 43:521–531
Gentil P, Arruda A, Souza D, Giessing J, Paoli A, Fisher J, Steele J (2017) Is there any practical application of meta-analytical results in strength training? Front Physiol 8:1
Kemmler W (2013) Meta-analysis and exercise related sports medicine [Meta-Analysen im trainingswissenschaftlichen und sportmedizinischen Spannungsfeld]. Dt Ztschr Sportmedizin 64:96–98
von Stengel S, Kemmler W, Lauber D, Weineck J, Kalender WA, Engelke K (2005) Power training is more effective than strength training to maintain bone mineral density in postmenopausal woman. J Appl Physiol 99:181–188
Rhodes EC (1995) Effect of one year of resistance training on strength and bone density in elderly women. Med Sci Sports Exerc 27:S21
Watson SL, Weeks BK, Weis LJ, Harding AT, Horan SA, Beck BR (2018) High-intensity resistance and impact training improves bone mineral density and physical function in postmenopausal women with osteopenia and osteoporosis: the LIFTMOR randomized controlled trial. J Bone Miner Res 33:211–220
Acknowledgements
This study is one of the intellectual outputs of the project “ACTLIFE-Physical activity the tool to improve the quality of life in osteoporosis people” and had grant support from the European Union’s Erasmus plus sport program under Grant Agreement No. 2017-2128/001-001.
Funding
Open Access funding provided by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
WK, MS, MK, and SvS completed data analysis and interpretation and drafted the manuscript. All the authors contributed to study conception and design and revised the manuscript. WK accepts responsibility for the integrity of the data sampling, analysis and interpretation.
Corresponding author
Ethics declarations
Conflict of interest
Wolfgang Kemmler (WK), Mahdieh Shojaa (MS), Matthias Kohl (MK), and Simon von Stengel (SvS) declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kemmler, W., Shojaa, M., Kohl, M. et al. Effects of Different Types of Exercise on Bone Mineral Density in Postmenopausal Women: A Systematic Review and Meta-analysis. Calcif Tissue Int 107, 409–439 (2020). https://doi.org/10.1007/s00223-020-00744-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-020-00744-w