Abstract
Novice and expert jugglers employ different visuomotor strategies: whereas novices look at the balls around their zeniths, experts tend to fixate their gaze at a central location within the pattern (so-called gaze-through). A gaze-through strategy may reflect visuomotor parsimony, i.e., the use of simpler visuomotor (oculomotor and/or attentional) strategies as afforded by superior tossing accuracy and error corrections. In addition, the more stable gaze during a gaze-through strategy may result in more accurate movement planning by providing a stable base for gaze-centered neural coding of ball motion and movement plans or for shifts in attention. To determine whether a stable gaze might indeed have such beneficial effects on juggling, we examined juggling variability during 3-ball cascade juggling with and without constrained gaze fixation (at various depths) in expert performers (n = 5). Novice jugglers were included (n = 5) for comparison, even though our predictions pertained specifically to expert juggling. We indeed observed that experts, but not novices, juggled significantly less variable when fixating, compared to unconstrained viewing. Thus, while visuomotor parsimony might still contribute to the emergence of a gaze-through strategy, this study highlights an additional role for improved movement planning. This role may be engendered by gaze-centered coding and/or attentional control mechanisms in the brain.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Research on visual search behavior (i.e., point-of-gaze excursions) has revealed differences between novice and expert performers that may be linked to improvements in the pickup of visual information. In particular, in far-aiming tasks like basketball and rifle shooting, experts exhibit longer target fixations, sometimes referred to as the “quiet eye” period (e.g., de Oliveira et al. 2008; Janelle et al. 2000; Vickers 1996). Indeed, gaze fixation or “anchoring” is a crucial aspect of visuomotor coordination and expertise (Roerdink et al. 2005, 2008). Nevertheless, many activities afford another kind of sensorimotor coordination and visual search behavior. A good example of this is juggling, which requires overt or covert tracking of more objects than hands are available for manipulation. Marked differences in the eye movements of expert and novice jugglers have been documented (e.g., Huys and Beek 2002) that are worth examining further to gain insight into the functional role(s) of gaze fixation in perceptual-motor expertise.
Studies of the visual basis of juggling are of interest to the understanding of the relationship between gaze fixation and information pickup because juggling requires distribution of visual attention and information pickup across the objects juggled. Aspirant jugglers are often instructed to throw a ball when the previously thrown ball passes through its zenith, and thus, by implication, to look at the zeniths of the ball trajectories. By having jugglers look through a wedge-shaped aperture of gradually decreasing size (in vertical dimension), Austin (1976) found that viewing the balls around their zeniths through an aperture of about 25 mm was sufficient to sustain a 3-ball cascade (see Fig. 1). By having experienced participants juggle while wearing liquid–crystal glasses that opened and closed at regular intervals, van Santvoord and Beek (1994) found that a viewing window of 82 ms sufficed to sustain juggling (see also Amazeen et al. 1999; Whiting and Sharp 1974). They also observed adjustments in the juggling pattern aimed at keeping the zeniths of the ball flights in sight. Huys and Beek (2002) reported that novice jugglers switched their gaze between positions close to the zeniths of the ball flights in a frequency-locked fashion, implying that they distribute their visual attention sequentially between the balls. Experts, on the other hand, confined their gaze to a limited area, sometimes referred to by jugglers as a “gaze-through” or “distant stare.” At the same time, however, experts’ smaller gaze excursions were also coupled to the motion of the balls.
One explanation of the emergence of a gaze-through strategy with expertise in juggling can be described in terms of parsimony (e.g., Beek and Lewbel 1995; Huys and Beek 2002), which may arise within the oculomotor or attentional system. Oculomotor parsimony is afforded by experts’ superior toss accuracy, allowing them to sustain the juggling pattern without foveation of the individual balls, with less vision-based error corrections. Attentional parsimony may arise when experts learn to attend all balls at the same time, rather than one at a time (Cavanagh and Alvarez 2005), thus eliminating the need for (covert and overt) attentional shifts. An explanation in terms of parsimony does not imply that the gaze-though strategy would be beneficial for juggling performance, but rather that similar performance is afforded by “simpler” visuomotor strategies.
At the same time, however, there may be beneficial effects of a reduction in eye movements during juggling. If movement planning improves with a more stable gaze, a gaze-through strategy may emerge over time because it would help improve tossing precision. Beneficial effects of gaze fixation on movement planning may arise at different stages of the visuomotor transformation for juggling. Differential sensitivity of the fovea and retinal periphery to visual motion could induce differences in movement planning: during a gaze-through strategy, balls move in the retinal periphery, while the tracking strategy of novices keeps balls within the fovea (e.g., Beek and Lewbel 1995; Huys and Beek 2002). The brain must use such retinal signals to generate movement plans for the catching and tossing movements, which most likely involves a stage “operating” in gaze-centered coordinates (Batista et al. 1999; Dessing et al. 2011; Gardner et al. 2008; Medendorp et al. 2003). Gaze-centered coding should be more accurate with a stable gaze, given that the accompanying gaze-centered updating necessarily adds noise to the representations (Baker et al. 2003; Karn et al. 1997). Besides gaze-centered coding, the brain most likely employs allocentric coding, that is, coding ball position relative to other, most likely stable objects in the environment (Byrne and Crawford 2010). With a stable gaze, allocentric and gaze-centered representations become aligned because the entire visual background is stable; this is likely to improve movement planning. Visual attention is another factor that will influence movement planning in visuomotor tasks like juggling, given the tight links between oculomotor and attentional control (Awh et al. 2006; Itti and Koch 2001; Moore et al. 2003). As has been recognized by several authors, a stable gaze may serve as a “visual pivot” from where attention is distributed across the visual field (Williams and Davids 1998; see also Shulman et al. 1979).
To gain more insight into the factors underlying the emergence of the gaze-through, we assessed the effects of the instruction to look at an explicit visual fixation point. Only the aforementioned “improved movement planning hypothesis” predicts that this manipulation should influence juggling variability. Providing a visual fixation point should improve performance because gaze fixations are more stable with an actual visual reference point (Leigh and Zee 2006; Morisita and Yagi 2001). The visual fixation point in addition provides a visual landmark for allocentric representations, which could further improve movement planning. On the other hand, if gaze-through behavior emerges only out of parsimony, performance improvement is not a driving factor. This explanation requires that similar performance can be achieved using a “simpler” oculomotor strategy; explicit enforcement of this simpler strategy (i.e., gaze fixation) should result in similar juggling performance (i.e., variability).
Importantly, these predictions can only be adequately tested for expert jugglers. Even though novice jugglers could technically benefit from stabilizing their gaze just like experts, gaze fixation would represent a non-preferred strategy, which may affect their performance irrespective of any potential (most likely small) improvements in movement planning. We nevertheless included novices in our study to compare the qualitative effects of fixation between novice and expert jugglers.
We thus examined juggling variability for unconstrained and constrained viewing (fixation) at various distances in both expert and novice jugglers. Variations in fixation distance were included because it is currently unknown at which depth experts prefer to stare during a gaze-through. Moreover, the neutral gaze depth position (i.e., the gaze depth attained in the absence of potential fixation targets, i.e., in the dark) corresponds better with far fixation (Leigh and Zee 2006), which could imply that the associated fixation (and thus possibly juggling) is more stable. We conducted the comparison qualitatively in terms of number of successful trials and quantitatively in terms of toss and catch positions and their variability (cf. Dessing et al. 2007; van Santvoord and Beek 1996; Huys and Beek 2002).
Methods
Participants
Five male expert jugglers (including author JCD) and five novice jugglers (three men and two women) participated in the experiment. Expert jugglers could all stably juggle five balls or more, while novices could only juggle three balls for longer than a minute. Participants were 19–35 years of age (mean 25 years). All experts and three novices were right-handed (Oldfield 1971; laterality quotient > .8); the other two novices were left-handed (laterality quotient < .2). One expert and two novices were left-eye dominant (Miles 1930). All participants had normal or corrected-to-normal vision and a stereoacuity <40 s of arc (Stereo Fly Test, Titmus Optical Co., Inc., Chicago, IL, USA). Participants signed an informed consent form before the experiment, and all were offered a small fee for their participation (which was rejected by two).
Experimental setup
Trials were performed with three white rubber balls (diameter: 74.6 mm; mass: 241 g). To facilitate the video analyses, participants stood in front of two large black curtains and wore black clothes and a black bandana, while their faces and hands were painted with black maquillage to improve data processing (note that this did not prevent vision of the hands, given that the experimentation room was illuminated). A fixation target (a red wooden bead, diameter 10 mm) could be positioned along a translucent vertical fishing line, spun from a lever (iron rod) positioned at 4 m height to a weight placed on the ground. The main experimental manipulation of fixation depth involved fixation targets 0.25, 0.40, and 2.00 m in front of the cyclopean eye. Participants stood with their head directly underneath the rotation axis of the lever (indicated by a plummet bob) facing a wall (at 4 m distance; Fig. 2a). Although not the focus of this study, we also had our participants juggle with left, right, low, and high fixation targets for exploratory purposes (this involved rotation of the lever or repositioning of the bead along the fishing line). Data from these conditions are not included in the present study.
Two cameras were fixed on the wall in front of the participants at a height of 2.50, 1.25 to the left and 1.25 m to the right of the participant. These were high-speed digital cameras (A600f, Basler Vision Technologies, Ahrensburg, Germany; progressive scan CMOS sensor of 1.27 cm; image resolution 656 × 488 pixels) equipped with a 16 mm lens (Pentax C31630, Hamburg, Germany) and an infrared filter (low pass for 750 nm wavelengths). The opening of the shutters of both cameras was timed using a single external signal (100 Hz). The cameras were focused on the participant’s torso. To increase the contrast between the balls and the black clothes, the juggling scene was lit using two sets of four stage lamps (40 Watts, 1610 lumens, F40/T12/HO, Kino Flo, California, USA), placed at ball height on both sides and slightly in front of the participant. During the experiment, two Optotrak 3020 cameras (Northern Digital Inc., Waterloo, Ontario, Canada) registered the position of a rigid body (three markers) attached to each hand between the knuckles of the thumb and index finger. These rigid bodies were used to reconstruct the toss and catch positions (see later). Three additional markers were placed on a head band and were used to track head movements. The 3D video setup was calibrated using a calibration board (1.50 m × 1.25 m) with a checkerboard pattern of which 10 crossing points were used. This board was placed at three different depths (0.25 m between each) along a slide, to cover the entire experimental volume. The 3D position of the crossing points was reconstructed using the board positions measured using an Optotrak system (RMS calibration error < 0.1 mm).
Procedure
Before the experiment, eye height relative to the ground was measured to determine the fixation positions. Participants were asked to juggle at eye height in all experimental conditions. The recordings for a given trial were started when the participant indicated that their juggling pattern was stable. In one condition, participants juggled with unconstrained fixation. All conditions were randomized and repeated twice (successively). When a participant was unable to sustain juggling for 20 s or longer, the trial was rerun. After three consecutive unsuccessful attempts, the condition in question was skipped. In fact, one novice participant only completed ~12.5 s of juggling in his second trial with far fixation. We decided not to exclude this participant and included this trial in the analyses; we confirmed that this decision did not influence our findings. The trial was marked as unsuccessful for calculating the success rates of this condition.
Data analyses
After each measurement, video files (avi format) were compressed with the Indeo 5.0 codec (Intel Corporation 1999). Ball positions were extracted using a custom written program (Matlab, The MathWorks, Inc., Natick, MA). Each video frame was converted to pure black and white images (using a grayscale threshold of 0.65). Subsequently, small white spots (e.g., eye white) and black spots on the balls (i.e., the fixation bead) were eliminated from the image using standard morphological image processing (Gonzalez and Woods 2008). On each image, ball position was defined as the barycenter of the remaining white pixels (about 1,000 pixels per ball); this was done for the upper part of the trajectory (i.e., about 30–40 frames). The 3D ball positions were reconstructed through calibration models generated in WINanalyze (Mikromak, Berlin, Germany; average spatial error: 0.17 mm). Subsequently, 2nd order polynomials were fitted to these 3D positions (average RMS error 0.5 mm); these polynomials were rotated to align the polynomials with gravity (i.e., ensuring that the 2nd order coefficients were zero in x- and y-direction and −9.81 m s−2 in z-direction) to compensate for minor rotational offsets in the placement of the Optotrak calibration cube.
From the first sample, we resampled the fitted ball trajectories to 250 Hz, corresponding with the recording frequency of the Optotrak system. At each sample, hand position was defined as the average position of the markers on the rigid body. The hand and ball trajectories were used to determine tosses (first sample before zenith at which the position of the bottom of the ball became higher than hand position) and catches (first sample after zenith at which the position of the bottom of the ball became lower than hand position). All positions were expressed relative to the average cyclopean eye position during each trail. Its vertical coordinate was defined as 0.05 m beneath the headband markers. Because we positioned each participant such that their eyes were directly underneath the plummet bob (which was also the approximate placement of the origin of our Optotrak calibration cube), we took the horizontal coordinates of the cyclopean eye in each trial to be the horizontal position of the head markers relative to their average position throughout the experiment. From the participant’s perspective, the positive x-, y-, and z-axes pointed rightward, forward, and upward, respectively. We analyzed the within-trial means and standard deviations of the toss and catch positions in three dimensions. These were calculated separately for each hand and then averaged across the two repetitions and the two hands.
Statistics
To assess changes in juggling variability associated with constrained fixation for expert and novice jugglers, we compared the dependent variables for unconstrained viewing with those for fixation at 0.25, 0.40, and 2.00 m using paired-samples t tests. In fact, the first comparison was only possible for the experts, given that none of the novices was able to successfully complete the condition with fixation at 0.25 m. Critical P values for the comparisons were step-down Holm-Sidak adjusted (and will be presented in the text for reference). Novices only completed two of the fixation conditions, but we still corrected for three comparisons within each expertise level to standardize our assessment of the effects of fixation across expertise levels.
Results
Task performance
Table 1 shows the number of drops made by each group in each condition, which illustrates that juggling performance differed considerably between experts and novices. Experts finished all fixation conditions, although some failed to keep juggling during the first trial of the near fixation condition. Most likely, this was due to the fact that this fixation distance is most uncommon to jugglers. Novices seemed to have more difficulty with the enforced (particularly near and far) fixation; only some of them were able to perform certain conditions without drops, whereas none successfully completed the near fixation condition. Because the number of drops depends on our limited requirement of two successful trials, Table 1 does not afford strong conclusions regarding the effects of fixation; this requires analyses of the spatial features of the juggling movement. Figure 3 illustrates front and top views of the ball trajectories for a trial of an expert and novice participant for all viewing conditions. These trajectories are cut off at the toss and catch. For the front views, we also plotted the hand trajectories. Figure 3 suggests that the consistency with which the expert juggler tossed balls might have been greater with fixation. While the crossing point of the left and right ball trajectories varied with fixation depth, this was specific to this particular participant. The novice juggler’s patterns, on the other hand, did not appear to change due to the enforced fixation (that is, for fixation at 0.40 and 2.00 m). In the following, we discuss the statistical analyses of the mean and variability of the toss and catch positions, which confirmed the patterns evident in Fig. 3.
Our analysis assessed whether expert jugglers would reduce juggling variability when they were instructed to gaze at a visual fixation point. For comparison, novice jugglers were also included in our experiment, even though, strictly speaking, the research hypothesis did not apply to these participants. In Fig. 4, for both experts and novices, we depicted the toss and catch positions in three dimensions for unconstrained viewing and for the three fixation distances. Both experts and novices tossed and caught balls closer to the body when they fixated at 0.40 m, compared to unconstrained viewing (toss/experts: t(4) = −5.24; P = 0.0063 [P crit = 0.0253]; toss/novices: t(4) = −6.42; P = 0.0030 [P crit = 0.017]; Fig. 4b; catch/experts: t(4) = −8.71; P = 0.00096 [P crit = 0.017]; catch/novices: t(8) = −4.60; P = 0.010 [P crit = 0.0253]; Fig. 4e). None of the other coordinates were affected significantly by imposed fixation. The analysis of the variability of catch and toss positions did reveal several significant effects, albeit only for the expert jugglers.
Average of the toss positions (X toss, Y toss, and Z toss; top panels) and catch positions (X catch, Y catch, and Z catch; bottom panels) for unconstrained viewing (UV) and for fixation at 0.25, 0.40, and 2.00 m (X: lateral direction, right is positive; Y: depth direction, forward is positive; Z: vertical direction, up is positive). Values for experts and novices are indicated by white and gray bars, respectively. The three columns correspond to the three dimensions. Error bars indicate standard errors. Significant effects are indicated by an asterisk
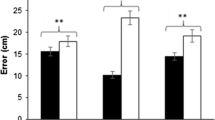
For experts, the spatial variability of the catches was significantly lower in depth when fixating at 0.40 m, compared to unconstrained viewing (t(4) = −4.84; P = 0.0084 [P crit = 0.017]; Fig. 5e), and significantly lower in lateral direction when fixating at 2.00 m, compared to unconstrained viewing (t(4) = −5.93, P = 0.0040 [P crit = 0.017]; Fig. 5d). The former effect could be associated with the shift of the juggling patterns toward the body with fixation at 0.40 m, but the fact that juggling variability of novices was not affected, even though they also shifted their patterns in depth, argues against this. Moreover, it is hard to see why experts would not shift their juggling pattern during unconstrained viewing if this would indeed reduce juggling variability.
Average standard deviations (SD) of the toss positions (X toss, Y toss, and Z toss; top panels) and catch positions (X catch, Y catch, and Z catch; bottom panels) as a function of the experimental conditions and expertise (experts: white bars; novices: gray bars; X: lateral direction, right is positive; Y: depth direction, forward is positive; Z: vertical direction, up is positive). Error bars indicate standard errors. Significant effects are indicated by an asterisk
We aimed to determine the factors contributing to the reduced variability of the catching positions. By definition, the variability of the catch position in any direction reflects a combination of the variability of the toss position and release vector in that direction, and the variability of the flight times. In addition, everything else being equal, horizontal catch variability varies with the variability of vertical toss position. The variability of the toss position was already considered in our initial analyses (Fig. 5a–c). Each follow-up analysis thus involved two additional comparisons: variability of the horizontal release velocity and variability of the flight times (horizontal refers to the lateral direction for effects of fixating at 2.00 m and to the depth direction for effects of fixating at 0.40 m). For all these comparisons, the critical P values were matched to the effects reported above (P crit = 0.017).
As shown in Fig. 5, the variability of toss position was not affected significantly by the fixation; our additional analyses showed this was also the case for the variability of the release vector and flight times. Nevertheless, we noted that the effect of fixation at 0.40 m approached significance for both the toss position and release velocity in depth (P = 0.024 and P = 0.019, respectively [P crit = 0.017]; Fig. 6a). This suggests that the reduced variability of the catch position in depth mainly reflected a combination of these two effects. Our follow-up analysis of the effect of fixation at 2.00 m on the variability of the lateral catch position did not reveal any additional significant effects. The variability of all variables was generally lower with fixation, but none of the effects approached significance (Fig. 6b). It thus appears that the reduced variability of the lateral catch position with fixation at 2.00 m reflects a combination of minor adjustments in various components of the toss and the timing of the catch.
Follow-up analyses of the significant effects of fixation on juggling variability of experts. a Standard deviation (SD) of catch position in depth (Y catch, in m), toss position in depth (Y toss, in m), toss height (Z toss, in m), release velocity in depth (V toss-Y , in m/s), and the flight time (T flight, in s) for unconstrained viewing and fixation at 0.40 m. b SD of lateral catch position (X catch, in m), lateral toss position (X toss, in m), Z toss (in m), lateral release velocity (V toss-X , in m/s), and T flight (in s) for unconstrained viewing and fixation at 2.00 m. P values of the respective effects of fixation are indicated above the bars
Discussion
This study aimed to gain insight into why expert jugglers tend to stare in the middle of the pattern, rather than look at the individual balls. It has been suggested that this gaze-through reflects visuomotor parsimony, that is, the use of “simpler” visuomotor strategies afforded by superior tossing accuracy and error corrections. A more stable gaze, however, may also improve movement planning due to coding and/or attentional mechanisms. To examine the explanatory value of the latter mechanism, we compared the juggling variability of expert jugglers in a 3-ball cascade with and without an explicit visual fixation point. While visuomotor parsimony would not predict any effects of this manipulation, improved movement planning would; the more stable gaze associated with explicit visual fixation (Leigh and Zee 2006; Morisita and Yagi 2001) should stabilize juggling. While this prediction could only be tested explicitly for expert jugglers used to juggling with a stable gaze, we also tested novice jugglers for comparison. While these novice jugglers may show the same positive effects on movement planning, they may also be adversely affected by the fact that explicit fixation deviates from their preferred oculomotor strategy. We tested these predictions using task success rates and the variability of the toss and catch positions in 3D. To control for changes in the juggling patterns that might influence juggling variability irrespective of fixation, we also assessed the within-trial spatial shifts in the juggling patterns.
Experts had some troubles juggling while fixating at 0.25 m, while novices failed altogether in this condition; we interpret this to reflect the fact that near fixation rarely arises in normal juggling. In terms of juggling variability, experts indeed benefited from the enforced fixation (i.e., reduced variability of catching positions), showing a reduced variability of the catching positions in depth when fixating at 0.40 m and in lateral direction when fixating at 2.00 m. They also tossed and caught balls closer to the body when fixating at 0.40 m; therefore, it cannot be excluded that the associated reduced variability was due to the change in the spatial features of the hand movements, rather than the explicit fixation at 0.40 m. Novices showed a similar shift toward the body without the accompanying reduction in variability. If the reduced variability with fixation at 0.40 m for experts is a side effect of the shifted juggling pattern, the question arises why experts would not shift their juggling pattern more forward when viewing was unconstrained. While this makes it unlikely that the reduced variability with fixation at 0.40 m is a side effect of the shifted juggling pattern, future investigations are needed for definitive confirmation.
The lower variability in lateral direction displayed by experts when fixating at 2.00 m was not accompanied by any changes in toss and catch positions, suggesting that this reduction in variability must be due to the explicit fixation instruction. Our follow-up analysis showed that while the variability of all toss parameters was lower with fixation at 2.00 m, this reduction never reached significance. We interpret this finding to suggest that the reduced variability of the catch positions in fact reflects combined effects of minor (i.e., nonsignificant) reductions in the variability of different aspects of the tossing action. For the two closer fixation distances, a quantitatively similar reduction in variability of the lateral catch positions was observed, but these reductions were not significant due to the variability between the participants. As mentioned in the Introduction, particular effects of far fixation might be due to a more stable gaze for the neutral eye position, corresponding to our far fixation (Leigh and Zee 2006). While these findings do not refute visuomotor parsimony as an explanation for the emergence of the gaze-through strategy, they suggest that improved movement planning with a stable gaze is another contributing factor.
In our novice participants, toss and catch variability was not affected by gaze fixation, even though they generally needed more attempts to successfully finish the far fixation condition and failed altogether in the near fixation condition. We hypothesized that gaze fixation, a non-preferred oculomotor strategy, might deteriorate performance. This may be reflected in an increased number of drops with gaze fixation, but our data did not afford strong conclusions in this regard. We considered the possibility that even for novice jugglers, a stable gaze might improve movement planning. Indeed, when novices successfully completed the trial (i.e., those trials included in the analyses of toss and catch parameters), the juggling of novices did not deteriorate due to fixation. This is not inconsistent with the positive effect of fixation on movement planning counterbalancing the negative effect of fixation being a non-preferred oculomotor strategy.
Before discussing our interpretation in more detail, we need to examine the validity of the underlying assumption that the instruction to fixate results in a more stable gaze (Leigh and Zee 2006; Morisita and Yagi 2001). We therefore included three of our expert participants in additional recordings of eye-in-head angles using an EyeLink II eye tracker (250 Hz, pupil-corneal reflection). Supplementary Fig. 1 depicts the relative distribution of these angles and illustrates that fixation generally was more stable in the fixation conditions compared to unconstrained viewing. In each panel, we also depicted saccade parameters (number of saccades × average saccade amplitude); these values reflect our participants’ tendency to show more saccades of larger amplitude with unconstrained viewing, compared to the fixation conditions. In the following, we discuss in more detail the specific mechanisms by which fixation may have improved movement planning.
Within the brain, gray matter in the middle temporal area (Draganski et al. 2004; Driemeyer et al. 2008) and white matter in the posterior parietal cortex (Scholz et al. 2009) has been shown to increase in size when learning to juggle. The middle temporal area is associated with pursuit eye movements and coding of object and background motion (Ilg 2008; Born and Tootell 1992), while the posterior parietal cortex plays an important role in vision-based movement planning (Crawford et al. 2011). The areas appear to code moving objects and movement goals, respectively, predominantly in a gaze-centered reference frame (Batista et al. 1999; Buneo et al. 2002; Beurze et al. 2010; Gardner et al. 2008; Marzocchi et al. 2008; Medendorp et al. 2003; Pitzalis et al. 2010). Areas employing gaze-centered coding must correct for any gaze shifts, a process called gaze-centered updating (Crawford et al. 2011). The inevitability of delays and noise within the brain imply that gaze-centered coding should be more accurate in the absence of gaze shifts (Baker et al. 2003; Karn et al. 1997), that is, when the need for gaze-centered updating is reduced. When possible, the brain forms not only gaze-centered representations. One alternative is to code where an object is relative to some visual landmark, allocentric coding (Byrne and Crawford 2010), which most likely improves when an explicit, stable visual landmark is present (e.g., the fixation bead), which thus would also improve movement plans.
A stable gaze may also improve movement planning through the attentional system. Williams and Davids (1998) and Shulman et al. (1979) proposed that gaze fixations may serve as a “visual pivot” for covertly distributing attention across the visual field. Such a mechanism is likely to be more accurate, the more stable the gaze fixation. The control of attention is tightly linked with the oculomotor system, since one typically attends to what one is looking at (Awh et al. 2006; Itti and Koch 2001; Moore et al. 2003). Indeed, novices look at individual balls in succession (Huys and Beek 2002), suggesting that attention is distributed in time. While experts may have optimized the temporal distribution of attention, the literature on multi-object tracking suggests the use of multifocal attentional control (Cavanagh and Alvarez 2005) or tracking of the “center of mass” (see Fehd and Seiffert 2008, 2010), which at least partly involves decoupling attentional shifts from eye movements. As mentioned in the Introduction, attentional parsimony may arise at this level, but at the same time, parallel, multifocal attention may just be better than sequential attention on single foci, which could result in better movement planning in juggling while fixating.
Optimizing attentional control in juggling most likely may only be attained once rhythmic motor control and visuospatial perception have been automated. In the brain, different networks are thought to subserve these different aspects of skill acquisition. On top of the aforementioned areas critical for visuomotor control, automatization in different tasks has been shown to involve the prefrontal, premotor, somatosensory, supplementary motor, and motor cortices, as well as the putamen, globus pallidus, striatum, and the anterior cerebellum (Floyer-Lea and Matthews 2004, 2005; Gobel et al. 2011; Grafton et al. 2008; Müller et al. 2002; Poldrack et al. 2005; Puttemans et al. 2005; Seidler and Noll 2008; Steele and Penhune 2010). Spatial visual attention most likely involves top-down feedback signals from parietal cortex to the middle temporal area (Herrington and Assad 2010; Saalmann et al. 2007; Womelsdorf et al. 2006).
Huys and Beek (2002) reported that the gaze-through strategy in experts during unconstrained viewing still involves gaze-locking with the balls, albeit of much smaller amplitude (i.e., a small movement to the left when a ball moves leftward). While they interpreted this behavior to be functional, our findings suggest that the low amplitude eye movements reflect an inability to entirely decouple eye movements from ball motion (for a related effect, see Theeuwes et al. 1998). These eye movements may arise with covert attentional shifts; this may be examined through the presence of microsaccades that have been argued to reflect the direction of attention (Hafed and Clark 2002). Alternatively, the small amplitude eye movements may be a form of motion-induced drift (~low gain visual pursuit). To distinguish between the aforementioned options, further investigations of juggling should involve detailed conjoint examinations of ball as well as eye movements, possibly with the addition of a dual task to manipulate attentional load.
Can our results be applied in practice? As mentioned in the Introduction, experts in many sports display explicit fixation strategies (e.g., de Oliveira et al. 2008; Janelle et al. 2000; Vickers 1996). Like in juggling, fixation typically emerges in sports without explicit instructions, and our results are in line with the suggestion that the instruction to fixate at specific landmarks (e.g., front of the rim in basketball) could be beneficial in certain sports situations. In juggling, learning new patterns most likely involves a strategy of looking at the individual balls, for both novices and experts. As a result, explicit fixation may only be useful when fine-tuning patterns that have already been mastered. Whether this indeed leads jugglers to fully stabilize their movements faster with explicit fixation needs to be investigated further.
References
Amazeen EL, Amazeen PG, Post AA, Beek PJ (1999) Timing the selection of information during rhythmic catching. J Mot Behav 31:279–289
Austin HA (1976) A computational theory of a physical skill. Massachusetts Institute of Technology, Boston
Awh E, Armstrong KM, Moore T (2006) Visual and oculomotor selection: links, causes and implications for spatial attention. Trends Cogn Sci 10:124–130
Baker JT, Harper TM, Snyder LH (2003) Spatial memory following shifts of gaze. I. Saccades to memorized world-fixed and gaze-fixed targets. J Neurophysiol 89:2564–2576
Batista AP, Buneo CA, Snyder LH, Andersen RA (1999) Reach plans in eye-centered coordinates. Science 285:257–260
Beek PJ, Lewbel A (1995) The science of juggling. Sci Am 273:92–97
Beurze SM, Toni I, Pisella L, Medendorp WP (2010) Reference frames for reach planning in human parietofrontal cortex. J Neurophysiol 104:1736–1745
Born RT, Tootell RB (1992) Segregation of global and local motion processing in primate middle temporal visual area. Nature 357:497–499
Buneo CA, Jarvis MR, Batista AP, Andersen RA (2002) Direct visuomotor transformations for reaching. Nature 416:632–636
Byrne PA, Crawford JD (2010) Cue reliability and a landmark stability heuristic determine relative weighting between egocentric and allocentric visual information in memory-guided reach. J Neurophysiol 103:3054–3069
Cavanagh P, Alvarez GA (2005) Tracking multiple targets with multifocal attention. Trends Cogn Sci 9:349–354
Crawford JD, Henriques DY, Medendorp WP (2011) Three-dimensional transformations for goal-directed action. Annu Rev Neurosci 34:309–331
de Oliveira RF, Oudejans RRD, Beek PJ (2008) Gaze behaviour in basketball shooting: further evidence for online visual control. Res Q Exerc Sport 79:399–404
Dessing JC, Daffertshofer A, Peper CE, Beek PJ (2007) Pattern stability and error correction during in-phase and antiphase four-ball juggling. J Mot Behav 39:433–446
Dessing JC, Crawford JD, Medendorp WP (2011) Spatial updating across saccades during manual interception. J Vis 11(4):1–18
Draganski B, Gaser C, Busch V, Schuierer G, Bogdahn U, May A (2004) Neuroplasticity: changes in grey matter induced by training. Nature 427:311–312
Driemeyer J, Boyke J, Gaser C, Büchel C, May A (2008) Changes in gray matter induced by learning–revisited. PLoS One 3:e2669
Fehd HM, Seiffert AE (2008) Eye movements during multiple object tracking: where do participants look? Cognition 108:201–209
Fehd HM, Seiffert AE (2010) Looking at the center of the targets helps multiple object tracking. J Vis 10:19.1–19.13
Floyer-Lea A, Matthews PM (2004) Changing brain networks for visuomotor control with increased movement automaticity. J Neurophysiol 92:2405–2412
Floyer-Lea A, Matthews PM (2005) Neuroanatomical correlates of motor acquisition and motor transfer. J Neurophysiol 94:512–518
Gardner JL, Merriam EP, Movshon JA, Heeger DJ (2008) Maps of visual space in human occipital cortex are retinotopic, not spatiotopic. J Neurosci 28:3988–3999
Gobel EW, Parrish TB, Reber PJ (2011) Neural correlates of skill acquisition: decreased cortical activity during a serial interception sequence learning task. Neuroimage 58:1150–1157
Gonzalez RC, Woods RE (2008) Morphological image processing. In: Digital image processing, 3rd edn. Pearson, Upper Saddle River, pp 627–680
Grafton ST, Schmitt P, Van Horn J, Diedrichsen J (2008) Neural substrates of visuomotor learning based on improved feedback control and prediction. Neuroimage 39:1383–1395
Hafed ZM, Clark JJ (2002) Microsaccades as an overt measure of covert attention shifts. Vis Res 42:2533–2545
Herrington TM, Assad JA (2010) Temporal sequence of attentional modulation in the lateral intraparietal area and middle temporal area during rapid covert shifts of attention. J Neurosci 30:3287–3296
Huys R, Beek PJ (2002) The coupling between point-of-gaze and ball movements in three-ball cascade juggling: the effects of expertise, pattern and tempo. J Sports Sci 20:171–186
Ilg UJ (2008) The role of areas MT and MST in coding of visual motion underlying the execution of smooth pursuit. Vis Res 48:2062–2069
Itti L, Koch C (2001) Computational modelling of visual attention. Nat Rev Neurosci 2:194–203
Janelle CM, Hillman CH, Apparies R, Murray NP, Meilli L, Fallon EA, Hatfield BD (2000) Expertise differences in cortical activation and gaze behaviour during rifle shooting. J Sport Exerc Psychol 22:167–182
Karn KS, Møller P, Hayhoe MM (1997) Reference frames in saccadic targeting. Exp Brain Res 115:267–282
Leigh RJ, Zee DS (2006) The neurology of eye movements. Oxford Press, New York
Marzocchi N, Breveglieri R, Galletti C, Fattori P (2008) Reaching activity in parietal area V6A of macaque: eye influence on arm activity or retinocentric coding of reaching movements? Eur J Neurosci 27:775–789
Medendorp WP, Goltz HC, Vilis T, Crawford JD (2003) Gaze-centered updating of visual space in human parietal cortex. J Neurosci 23:6209–6214
Miles WR (1930) Ocular dominance in human adults. J Gen Psychol 3:412–430
Moore T, Armstrong KM, Fallah M (2003) Visuomotor origins of covert spatial attention. Neuron 40:671–683
Morisita M, Yagi T (2001) The stability of human eye orientation during visual fixation and imagined fixation in three dimensions. Auris Nasus Larynx 28:301–304
Müller RA, Kleinhans N, Pierce K, Kemmotsu N, Courchesne E (2002) Functional MRI of motor sequence acquisition: effects of learning stage and performance. Brain Res Cogn Brain Res 14:277–293
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Pitzalis S, Sereno MI, Committeri G, Fattori P, Galati G, Patria F, Galletti C (2010) Human V6: the medial motion area. Cereb Cortex 20:411–424
Poldrack RA, Sabb FW, Foerde K, Tom SM, Asarnow RF, Bookheimer SY, Knowlton BJ (2005) The neural correlates of motor skill automaticity. J Neurosci 25:5356–5364
Puttemans V, Wenderoth N, Swinnen SP (2005) Changes in brain activation during the acquisition of a multifrequency bimanual coordination task: from the cognitive stage to advanced levels of automaticity. J Neurosci 25:4270–4278
Roerdink M, Peper CE, Beek PJ (2005) Effects of correct and transformed visual feedback on rhythmic visuo-motor tracking: tracking performance and visual search behaviour. Hum Mov Sci 24:379–402
Roerdink M, Ophoff ED, Peper CE, Beek PJ (2008) Visual and musculoskeletal underpinnings of anchoring in rhythmic visuo-motor tracking. Exp Brain Res 184:143–156
Saalmann YB, Pigarev IN, Vidyasagar TR (2007) Neural mechanisms of visual attention: how top-down feedback highlights relevant locations. Science 316:1612–1615
Scholz J, Klein MC, Behrens TE, Johansen-Berg H (2009) Training induces changes in white-matter architecture. Nat Neurosci 12:1370–1371
Seidler RD, Noll DC (2008) Neuroanatomical correlates of motor acquisition and motor transfer. J Neurophysiol 99:1836–1845
Shulman GL, Remington RW, Mclean JP (1979) Moving attention through visual space. J Exp Psychol: Hum Percept Perf 5:522–526
Steele CJ, Penhune VB (2010) Specific increases within global decreases: a functional magnetic resonance imaging investigation of five days of motor sequence learning. J Neurosci 30:8332–8341
Theeuwes J, Kramer AF, Hahn S, Irwin DE (1998) Our eyes do not always go where we want them to go: capture of the eyes by new objects. Psychol Sci 9:379–385
van Santvoord AAM, Beek PJ (1994) Phasing and the pickup of optical information in cascade juggling. Ecol Psychol 6:239–263
van Santvoord AAM, Beek PJ (1996) Spatiotemporal variability in cascade juggling. Acta Psychol 91:131–151
Vickers JN (1996) Visual control when aiming at a far target. J Exp Psychol: Hum Percept Perf 22:342–354
Whiting HTA, Sharp RH (1974) Visual occlusion factors in a discrete ball-catching task. J Mot Behav 6:11–16
Williams AM, Davids K (1998) Visual search strategy, selective attention, and expertise in soccer. Res Q Exerc Sport 69:111–128
Womelsdorf T, Anton-Erxleben K, Pieper F, Treue S (2006) Dynamic shifts of visual receptive fields in cortical area MT by spatial attention. Nat Neurosci 9:1156–1160
Acknowledgments
We thank Hans Agricola for assisting in preparing the experimental setup, Dr. Berry den Brinker for developing the ball tracking software and Irénée Hottier for preparing Fig. 2.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
221_2011_2967_MOESM1_ESM.eps
Supplementary Fig. 1 Distribution of eye-in-head angles (horizontal: θEIH; vertical: ψEIH) during juggling, recorded at 250 Hz using an EyeLink II system (SR Research, Inc., Missassauga, Ontario, Canada) in pupil–corneal model. Slow drift and noise were filtered out using a 4th order recursive Butterworth band-pass filter (0.75Hz-60Hz). We determined the distribution of these angles (normalized to the total number of samples in the trial) for three expert jugglers (rows) for all experimental conditions (columns). For each position (θref,ψref), we determined the number of recorded eye-in-head angles that were less than 1 degree from this position (i.e., \( \sqrt {\left( {\theta_{\text{EIH}} - \theta_{\text{ref}} } \right)^{2} + \left( {\psi_{\text{EIH}} - \psi_{\text{ref}} } \right)^{2} } < 1 \)). This number was used for color coding (a continuous color scale from dark blue via green, yellow, and orange to dark red representing a scale from zero to the maximal number of gaze positions). As such, a stable gaze would be represented by a single red peak around (0,0). In addition, we calculated two saccade parameters (number and mean amplitude of saccades) that were inserted in each panel. Both the distributions as well as the saccade parameters confirm that the tested expert jugglers tended to have a less stable gaze (i.e., wide distributions and more/larger saccades) with unconstrained viewing compared to the fixation conditions. Interestingly, Expert 2 actually did not display a gaze-through during unconstrained viewing (i.e., he tracked each ball around its zenith). In addition, the fixation of Expert 5 appeared to be slightly less variable for unconstrained viewing than for fixation at 0.25 m or 0.40 m; interestingly, of the three tested participants, the latter expert showed the smallest effects of fixation on the variability of the catch positions. (EPS 20592 kb)
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Dessing, J.C., Rey, F.P. & Beek, P.J. Gaze fixation improves the stability of expert juggling. Exp Brain Res 216, 635–644 (2012). https://doi.org/10.1007/s00221-011-2967-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-011-2967-6