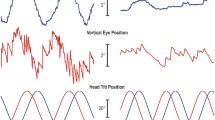
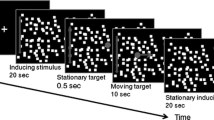
Abstract
Orientation and movement relies on both visual and vestibular information mapped in separate coordinate systems. Here, we examine how coordinate systems interact to guide eye movements of rabbits. We exposed rabbits to continuous horizontal optokinetic stimulation (HOKS) at 5°/s to evoke horizontal eye movements, while they were statically or dynamically roll-tilted about the longitudinal axis. During monocular or binocular HOKS, when the rabbit was roll-tilted 30° onto the side of the eye stimulated in the posterior → anterior (P → A) direction, slow phase eye velocity (SPEV) increased by 3.5–5°/s. When the rabbit was roll-tilted 30° onto the side of the eye stimulated in the A → P direction, SPEV decreased to ~2.5°/s. We also tested the effect of roll-tilt after prolonged optokinetic stimulation had induced a negative optokinetic afternystagmus (OKAN II). In this condition, the SPEV occurred in the dark, “open loop.” Modulation of SPEV of OKAN II depended on the direction of the nystagmus and was consistent with that observed during “closed loop” HOKS. Dynamic roll-tilt influenced SPEV evoked by HOKS in a similar way. The amplitude and the phase of SPEV depended on the frequency of vestibular oscillation and on HOKS velocity. We conclude that the change in the linear acceleration of the gravity vector with respect to the head during roll-tilt modulates the gain of SPEV depending on its direction. This modulation improves gaze stability at different image retinal slip velocities caused by head roll-tilt during centric or eccentric head movement.






Similar content being viewed by others
References
Angelaki DE, Hess BJ (1994) Inertial representation of angular motion in the vestibular system of rhesus monkeys. I. Vestibulo-ocular reflex. J Neurophysiol 71:1222–1249
Angelaki DE, Hess BJ (1995) Inertial representation of angular motion in the vestibular system of Rhesus monkeys. II. Otolith-controlled transformation that depends on intact cerebellar Nodulus. J Neurophysiol 73:1729–1751
Barmack NH (1981) A comparison of the horizontal and vertical vestibulo-ocular reflexes of the rabbit. J Physiol 314:547–564
Barmack NH, Nelson BJ (1987) Influence of long term optokinetic stimulation on eye movements of the rabbit. Brain Res 437:111–120
Barmack NH, Errico P, Ferraresi A, Fushiki H, Pettorossi VE, Yakhnitsa V (2002) Cerebellar nodulectomy impairs spatial memory of vestibular and optokinetic stimulation in rabbits. J Neurophysiol 87:962–975
Benson AJ (1974) Modification of the response to angular accelerations by linear accelerations, vestibular system. Part 2—psychophysics, applied aspects, and general interpretations. Springer, Berlin, pp 281–320
Boyle R, Buttner U, Markert G (1985) Vestibular nuclei activity and eye movements in the alert monkey during sinusoidal optokinetic stimulation. Exp Brain Res 57:362–369
Clément G (2003) A review of the effects of space flight on the asymmetry of vertical optokinetic and vestibule-ocular reflexes. J Vest Res 13:255–263
Curthoys IS, Wearne SL, Dai M, Halmagyi GM, Holden JR (1992) Linear acceleration modulates the nystagmus induced by angular acceleration stimulation of the horizontal canal. Ann NY Acad Sci 656:716–724
Dai M, Raphan T, Cohen B (1991) Spatial orientation of the vestibular system. Dependence of optokinetic after-nystagmus on gravity. J Neurophysiol 66:422–1439
Erickson RG, Barmack NH (1980) A comparison of the horizontal and vertical optokinetic reflexes of the rabbit. Exp Brain Res 40:448–456
Fernandez C, Goldberg JM, Abend W (1972) Response to static tilts of peripheral neurons innervating otolith organs of the Squirrel Monkey. J Neurophysiol 35:978–997
Fetter M, Heimberger J, Black R, Hermann W, Sievering F, Dichgans J (1996) Otolith-semicircular canal interaction during postrotatory nystagmus in humans. Exp Brain Res 108:463–472
Gizzi M, Raphan T, Rudolph S (1994) Orientation of human optokinetic nystagmus to gravity: a model-based approach. Exp Brain Res 99:347–360
Guedry FE (1965) Orientation of the rotation axis relative to gravity: its influence on nystagmus and sense of rotation. Acta Otolaryngol 60:30–48
Harris LR (1987) Vestibular and optokinetic eye movements evoked in the cat by rotation about a tilted axis. Exp Brain Res 66:522–532
Harris LR, Barnes GR (1987) Orientation of vestibular nystagmus is modified by head tilt. In: Graham MD, Kemink JLN (eds) The vestibular system: neurophysiology and clinical research. Raven, New York, pp 539–549
Haustein W (1989) Considerations on Listing’s law and primary position by means of matrix description of eye position control. Biol Cybern 60:411–420
Holly JE, Vrubleskis A, Carlson LE (2008) Whole-motion model of perception during forward- and backward-facing centrufuge runs. J Vestib Res 18:171–186
Keller EL, Precht W (1978) Persistence of visual response in vestibular nucleus neurons in cerebellectomized cat. Exp Brain Res 32:591–594
Kitama T, Luan H, Ishida M, Satp Y (2004) Effect of side-down tilt on optokinetic nystagmus and optokinetic after-nystagmus in cats. Neurosci Res 48:269–283
Kubo T, Matsunaga T, Matano S (1977) Convergence of ampullar and macular inputs on vestibular nuclei unit of the rat. Acta Otolaryngol (Stockh) 84:166–177
Loe PR, Tomko DL, Werner G (1973) The neural signal of angular head position in primary afferent vestibualr nerve axons. J Physiol (Lond) 230:29–50
Maioli C (1988) Optokinetic nystagmus: modelling the velocity storage mechanism. J Neurosci 8:821–832
Maruta J, MacDougall HG, Simpson JI, Raphan T, Cohen B (2006) Eye velocity asymmetry, ocular orientation, and convergence induced by angular rotation in the rabbit. Vis Res 46:961–969
Maruta J, Raphan T, Simpson JI, Cohen B (2008) Vertical (Z-axis) acceleration alters the ocular response to linear acceleration in the rabbit. Exp Brain Res 185:87–99
Merfeld DM, Young LR, Paige GD, Tomko DL (1993) Three dimensional eye movements of squirrel monkeys following postrotatory tilt. J Vest Res 3:123–139
Minor LB, Goldberg JM (1990) Influence of static head position on the horizontal nystagmus evoked by caloric, rotational and optokinetic stimulation in the squirrel monkey. Exp Brain Res 82:1–13
Pettorossi VE, Errico P, Santarelli RM (1991) Contribution of the maculo-ocular reflex to gaze stability in the rabbit. Exp Brain Res 83:366–374
Pettorossi VE, Errico P, Ferraresi A (1997) Difference in quick phases induced by horizontal and vertical vestibular stimulations: role of the otolithic input. J Vest Res 7:89–99
Pettorossi VE, Errico P, Ferraresi A, Barmack NH (1999) Optokinetic and vestibular stimulation determines the spatial orientation of negative optokinetic afternystagmus in the rabbit. J Neurosci 19:1524–1531
Raphan T, Cohen B (1988) Organizational principles of velocity storage in three dimensions. The effect of gravity on cross-coupling of optokinetic after-nystagmus. Ann NY Acad Sci 545:74–92
Savitzky A, Golay MJE (1964) Smooting and differentiation of data by simplified least square procedure. Anal Chem 36:1627–1639
Schrader V, Koenig E, Dichgans J (1985) The effect of lateral head tilt on horizontal postrotatoty nystagmus I and II and the Purkinje effect. Acta Otolaryngol 100:98–105
Simpson JI, Graaf W (1981) Eye-muscle geometry and compensatory eye movements in lateral-eyed and frontal-eyed animals. Ann NY Acad Sci 374:20–30
Simpson JI, Soodak RE (1984) The accessory optic system. Ann Rev Neurosci 7:13–41
Solomon D, Cohen B (1992) Stabilization of gaze during circular locomotion in darkness. II. Contribution of velocity storage to compensatory eye and head nystagmus in the running monkey. J Neurophysiol 67:1158–1170
Soodak RE, Simpson JI (1988) The accessory optic system of rabbit. I. Basic visual response properties. J Neurophysiol 60:2037–2054
Van der Steen J, Collewijn H (1984) Ocular stability in the horizontal, frontal and sagittal planes in the rabbit. Exp Brain Res 56:263–274
Waespe W, Henn V (1978) Reciprocal changes in primary and secondary optokinetic after-nystagmus produced by repetitive optokinetic stimulation in the monkey. Arch Psychiatr Nervenkr 225:23–30
Wearne S, Raphan T, Cohen B (1998) Control of spatial orientation of the angular vestibuloocular reflex by the nodulus and uvula. J Neurophysiol 79:2690–2715
Wearne S, Raphan T, Cohen B (1999) Effects of tilt of the gravito-inertial acceleration vector on the angular vestibuloocular reflex during centrifugation. J Neurophysiol 81:2175–2190
Yakushin SB, Xiang Y, Cohen B, Raphan T (2009) Dependence of the roll angular vestibuloocular reflex (aVOR) on gravity. J Neurophysiol 102:2616–2626
Zhang X, Zakir M, Meng H, Sato H, Uchino Y (2001) Convergence of the horizontal semicircular canal and otolith afferents on cat single vestibular neurons. Exp Brain Res 140:1–11
Acknowledgments
This research was supported in part by NIH DC006668, Fondazione Cassa di Risparmio di Perugia and Prin 2007 Ministero dell’Istruzione, dell’Università e della Ricerca.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pettorossi, V.E., Ferraresi, A., Botti, F.M. et al. Head position modulates optokinetic nystagmus. Exp Brain Res 213, 141–152 (2011). https://doi.org/10.1007/s00221-011-2785-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-011-2785-x