Abstract
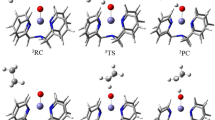
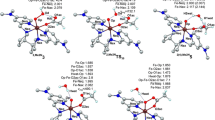
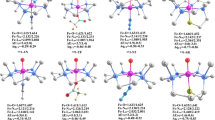
The C–H bond activation by non-heme iron-oxo species is one of the most interesting fundamental steps in organic oxidation reactions. This work focuses on the C–H bond activation by an external electric field (EEF) in a non-heme complex. The hydrogen atom abstraction from 2,5-(MeO)2C6H3CH2OH by [(N4Py)FeIV(O)]2+ utilizing an EEF was studied using density functional theory. Our analysis suggests that the mechanism of this process can be characterized as a mixture of hydrogen atom transfer and concerted-asynchronous electron–proton transfer that can be modulated by the EEF. Our study demonstrates an EEF that decreases the reaction barrier by increasing the electron donor orbital energy, which facilitates electron transfer. The reaction finally becomes barrierless when the EEF is greater than 0.0045au. The finding from this work demonstrates the exciting possibility of tuning the reaction mechanism using the EEF.







Similar content being viewed by others
References
He C, Mishina Y (2004) Modeling non-heme iron proteins. Curr Opin Chem Biol 8(2):201–208
Cook GK, Mayer JM (1995) C–H bond activation by metal oxo species: chromyl chloride oxidations of cyclooctane, isobutane, and toluene. J Am Chem Soc 117(27):7139–7156
Kumar D, Hirao H, Que L, Shaik S (2005) Theoretical investigation of C–H hydroxylation by (N4Py) FeIV O2+: an oxidant more powerful than P450? J Am Chem Soc 127(22):8026–8027
Balcells D, Clot Eric, Eisenstein Odile (2010) C–H bond activation in transition metal species from a computational perspective. Chem Rev 110(2):749–823
Yang T, Quesne MG, Neu HM, Cantu Reinhard FG, Goldberg DP, de Visser SP (2016) Singlet versus triplet reactivity in an Mn(V)-oxo species: testing theoretical predictions against experimental evidence. J Am Chem Soc 138(38):12375–12386
Cantu Reinhard FG, Faponle AS, de Visser SP (2016) Substrate sulfoxidation by an iron(IV)-oxo complex: benchmarking computationally calculated barrier heights to experiment. The journal of physical chemistry A 120(49):9805–9814
Hirao H, Que L Jr, Nam W, Shaik S (2008) A two-state reactivity rationale for counterintuitive axial ligand effects on the C–H activation reactivity of nonheme FeIV = O oxidants. Chem Eur J 14(6):1740–1756
Janardanan D, Wang Y, Schyman P, Que L Jr, Shaik S (2010) The fundamental role of exchange-enhanced reactivity in C–H activation by S = 2 oxo iron (IV) complexes. Angew Chem Int Ed 49(19):3342–3345
Nam W (2007) High-valent iron (IV)–oxo complexes of heme and non-heme ligands in oxygenation reactions. Acc Chem Res 40(7):522–531
Chen H, Lai W, Shaik S (2010) Exchange-enhanced H-abstraction reactivity of high-valent nonheme iron (IV)-oxo from coupled cluster and density functional theories. J Phys Chem Lett 1(10):1533–1540
Fukuzumi S, Kotani H, Suenobu T, Hong S, Lee YM, Nam W (2010) Contrasting effects of axial ligands on electron-transfer versus proton-coupled electron-transfer reactions of nonheme oxoiron (IV) complexes. Chem Eur J 16(1):354–361
Comba P, Fukuzumi S, Kotani H, Wunderlich S (2010) Electron-transfer properties of an efficient nonheme iron oxidation catalyst with a tetradentate bispidine ligand. Angew Chem 49(14):2622–2625
Que L Jr, Ho RY (1996) Dioxygen activation by enzymes with mononuclear non-heme iron active sites. Chem Rev 96(7):2607–2624
Gardner KA, Mayer JM (1995) Understanding C–H bond oxidations: H and H-transfer in the oxidation of toluene by permanganate. Science 269(5232):1849–1851
de Visser SP, Oh K, Han A-R, Nam W (2007) Combined experimental and theoretical study on aromatic hydroxylation by mononuclear nonheme iron (IV)-oxo complexes. Inorg Chem 46(11):4632–4641
Hirao H, Kumar D, Que L Jr, Shaik S (2006) Two-state reactivity in alkane hydroxylation by non-heme iron-oxo complexes. J Am Chem Soc 128(26):8590–8606
de Visser SP (2009) Trends in substrate hydroxylation reactions by heme and nonheme iron (IV)-oxo oxidants give correlations between intrinsic properties of the oxidant with barrier height. J Am Chem Soc 132(3):1087–1097
Sastri CV, Lee J, Oh K, Lee YJ, Lee J, Jackson TA, Ray K, Hirao H, Shin W, Halfen JA (2007) Axial ligand tuning of a nonheme iron (IV)-oxo unit for hydrogen atom abstraction. Proc Natl Acad Sci 104(49):19181–19186
Klinker EJ, Shaik S, Hirao H, Que L Jr (2009) A two-state reactivity model explains unusual kinetic isotope effect patterns in C–H bond cleavage by nonheme oxo iron (IV) complexes. Angew Chem Int Ed 48(7):1291–1295
Janardanan D, Usharani D, Chen H, Shaik S (2011) Modeling C–H abstraction reactivity of nonheme Fe(IV) O oxidants with alkanes: what role do counter ions play? J Phys Chem Lett 2(20):2610–2617
Shaik S, Hirao H, Kumar D (2007) Reactivity of high-valent iron-oxo species in enzymes and synthetic reagents: a tale of many states. Acc Chem Res 40(7):532–542
Morimoto Y, Kotani H, Park J, Lee YM, Nam W, Fukuzumi S (2010) Metal ion-coupled electron transfer of a nonheme oxoiron (IV) complex: Remarkable enhancement of electron-transfer rates by Sc3+. J Am Chem Soc 133(3):403–405
Lee YM, Kotani H, Suenobu T, Nam W, Fukuzumi S (2008) Fundamental electron-transfer properties of non-heme oxoiron(IV) complexes. J Am Chem Soc 130(2):434–435
Latifi R, Bagherzadeh M, de Visser SP (2009) Origin of the correlation of the rate constant of substrate hydroxylation by nonheme iron (IV)–oxo complexes with the bond-dissociation energy of the C–H bond of the substrate. Chem Eur J 15(27):6651–6662
Fukuzumi S (2009) Roles of metal ions in controlling bioinspired electron-transfer systems. metal ion-coupled electron transfer. Prog Inorg Chem 56:49–154
Morimoto Y, Park J, Suenobu T, Lee YM, Nam W, Fukuzumi S (2012) Mechanistic borderline of one-step hydrogen atom transfer versus stepwise Sc3+-coupled electron transfer from benzyl alcohol derivatives to a non-heme iron(IV)-oxo complex. Inorg Chem 51(18):10025–10036
Wimalasena K, May SW (1987) Mechanistic studies on dopamine beta-monooxygenase catalysis: N-dealkylation and mechanism-based inhibition by benzylic-nitrogen-containing compounds. Evidence for a single-electron-transfer mechanism. J Am Chem Soc 109(13):4036–4046
Baciocchi E, Bietti M, Lanzalunga O, Lapi A, Raponi D (2010) N-Demethylation of N, N-Dimethylanilines by the Benzotriazole N-Oxyl radical: evidence for a two-step electron transfer–proton transfer mechanism. J Organ Chem 75(5):1378–1385
Barbieri A, De Gennaro M, Di Stefano S, Lanzalunga O, Lapi A, Mazzonna M, Olivo G, Ticconi B (2015) Isotope effect profiles in the N-demethylation of N, N-dimethylanilines: a key to determine the p K a of nonheme Fe (iii)–OH complexes. Chem Commun 51(24):5032–5035
Li C, Wu W, Cho KB, Shaik S (2009) Oxidation of tertiary amines by cytochrome P450—kinetic isotope effect as a spin-state reactivity probe. Chem Eur J 15(34):8492–8503
Roberts KM, Jones JP (2010) Anilinic N-oxides support cytochrome P450-mediated N-dealkylation through hydrogen-atom transfer. Chem Eur Jl 16(27):8096–8107
Usharani D, Lacy DC, Borovik AS, Shaik S (2013) Dichotomous hydrogen atom transfer vs proton-coupled electron transfer during activation of X–H bonds (X = C, N, O) by nonheme iron-oxo complexes of variable basicity. J Am Chem Soc 135(45):17090–17104
Warren JJTTA, Mayer JM (2010) Thermochemistry of proton-coupled electron transfer reagents and its implications. Chem Rev 110(12):6961–7001
Sirjoosingh A, Hammes-Schiffer S (2011) Proton-coupled electron transfer versus hydrogen atom transfer: generation of charge-localized diabatic states. J Phys Chem A 115(11):2367–2377
Mayer JM, Hrovat DA, Thomas JL, Borden WT (2002) Proton-coupled electron transfer versus hydrogen atom transfer in benzyl/toluene, methoxyl/methanol, and phenoxyl/phenol self-exchange reactions. J Am Chem Soc 124(37):11142–11147
Jeong YJ, Kang Y, Han AR, Lee YM, Kotani H, Fukuzumi S, Nam W (2008) Hydrogen atom abstraction and hydride transfer reactions by iron (IV)-oxo porphyrins. Angew Chem Int Ed 47(38):7321–7324
Tishchenko O, Truhlar DG, Ceulemans A, Nguyen MT (2008) A unified perspective on the hydrogen atom transfer and proton-coupled electron transfer mechanisms in terms of topographic features of the ground and excited potential energy surfaces as exemplified by the reaction between phenol and radicals. J Am Chem Soc 130(22):7000–7010
Mayer JM (2010) Understanding hydrogen atom transfer: from bond strengths to Marcus theory. Acc Chem Res 44(1):36–46
Lai W, Li C, Chen H, Shaik S (2012) Hydrogen-abstraction reactivity patterns from A to Y: the valence bond way. Angew Chem Int Ed 51(23):5556–5578
Saouma CT, Mayer JM (2014) Do spin state and spin density affect hydrogen atom transfer reactivity? Chem Sci 5(1):21–31
Hammes-Schiffer S (2010) Introduction: proton-coupled electron transfer. Chem Rev 110:6937–6938
Yang L, Chen X, Qu Z, Gao J (2018) Combined multistate and Kohn–Sham density functional theory studies of the elusive mechanism of N-dealkylation of N, N-dimethylanilines mediated by the biomimetic nonheme oxidant FeIV (O)(N4Py)(ClO4)2. Front Chem 6:406
Lao K, Franzen S, Steffen M, Lambright D, Stanley R, Boxer SG (1995) Effects of applied electric fields on the quantum yields for the initial electron transfer steps in bacterial photosynthesis II. Dynamic Stark effect. Chem Phys 197(3):259–275
Seki K, Traytak S, Tachiya M (2003) Rigorous calculation of electric field effects on the free energy change of the electron transfer reaction. J Chem Phys 118(2):669–679
Shaik S, de Visser SP, Kumar D (2004) External electric field will control the selectivity of enzymatic-like bond activations. J Am Chem Soc 126(37):11746–11749
De Biase PM, Doctorovich F, Murgida DH, Estrin DA (2007) Electric field effects on the reactivity of heme model systems. Chem Phys Lett 434(1–3):121–126
Hirao H, Chen H, Carvajal MA, Wang Y, Shaik S (2008) Effect of external electric fields on the C–H Bond activation reactivity of nonheme iron-oxo reagents. J Am Chem Soc 130(11):3319–3327
Frisch MJ, Trucks G, Schlegel H, Scuseria G, Robb M, Cheeseman J, Scalmani G, Barone V, Mennucci B, Petersson G (2009) Gaussian 09, Revision D. 01. Gaussian Inc, Wallingford
Becke AD (1993) Becke’s three parameter hybrid method using the LYP correlation functional. J Chem Phys 98:5648–5652
Rauhut G, Pulay P (1995) Transferable scaling factors for density functional derived vibrational force fields. J Phys Chem 99(10):3093–3100
Becke AD (1992) Density-functional thermochemistry. I. The effect of the exchange-only gradient correction. J Chem Phys 96:2155–2160
Wang Y, Yang C, Wang H, Han K, Shaik S (2007) A new mechanism for ethanol oxidation mediated by cytochrome P450 2E1: bulk polarity of the active site makes a difference. Chembiochem Eur J Chem Biol 8(3):277–281
Schröder D, Shaik S, Schwarz H (2000) Two-state reactivity as a new concept in organometallic chemistry. Acc Chem Res 33(3):139–145
Shaik S, Kumar D, de Visser SP, Altun A, Thiel W (2005) Theoretical perspective on the structure and mechanism of cytochrome P450 enzymes. Chem Rev 105(6):2279–2328
Fried SD, Boxer SG (2017) Electric fields and enzyme catalysis. Annu Rev Biochem 86:387–415
Acknowledgements
This work was supported by the National Science Foundation of China (Nos. 21503089) and China Postdoctoral Science Foundation (2015M581393). Z.X.Q. thanks Adam Grofe for constructive suggestions as well as the improvement in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, M., Qu, Z. The C–H bond activation by non-heme oxidant [(N4Py)FeIV(O)]2+ with external electric field. Theor Chem Acc 139, 64 (2020). https://doi.org/10.1007/s00214-020-2581-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-020-2581-4