Abstract.
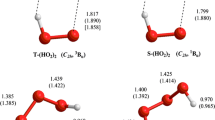
The energetics of the phenol O–H bond in methanol and the water O–H bond in liquid water were investigated by microsolvation modelling and statistical mechanics Monte Carlo simulations. The microsolvation approach was based on density functional theory calculations. Optimised structures for clusters of phenol and the phenoxy radical with one and two methanol molecules are reported. By analysing the differential solvation of phenol and the phenoxy radical in methanol, we predict that the phenol O–H homolytic bond dissociation enthalpy in solution is 24.3±11 kJ/mol above the gas-phase value. The analysis of the water O–H bond dissociation by microsolvation was based on optimised structures of OH•–(H2O)1−6 and –(H2O)1−7 clusters. Microsolvation modelling and statistical mechanics simulations predict that the HO–H bond dissociation enthalpies in the gas phase and in liquid water are very similar. Our results stress the importance of estimating the differences between the solvation enthalpies of the radical species and the parent molecule and the limitations of local models based on microsolvation.
Similar content being viewed by others
Acknowledgements.
S. G. E., R. C. G. and P. C. dC. gratefully acknowledge the support of Fundação para a Ciência e a Tecnologia. (PhD grants SFRH/BD/10200/2002, PRAXIS/XXI/BD/15920/98, and PRAXIS/XXI/BD/6503/2001). This work was partially supported by the Sapiens program of the FCT, Portugal (grant no. POCTI/43315/QUI/2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Proceedings of the 11th International Congress of Quantum Chemistry satellite meeting in honor of Jean-Louis Rivail
Rights and permissions
About this article
Cite this article
Estácio, S., Couto, P., Guedes, R. et al. Homolytic dissociation in hydrogen-bonding liquids: energetics of the phenol O–H bond in methanol and the water O–H bond in water. Theor Chem Acc 112, 282–289 (2004). https://doi.org/10.1007/s00214-004-0580-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-004-0580-5