Abstract
Rationale
Attention is compromised in many psychiatric disorders, including attention-deficit/hyperactivity disorder (ADHD). While dopamine and noradrenaline systems have been implicated in ADHD, their exact role in attentional processing is yet unknown.
Objectives
We applied the theory of visual attention (TVA) model, adapted from human research, to the rat 5-choice serial reaction time task (5CSRTT) to investigate catecholaminergic modulation of visual attentional processing in healthy subjects of high- and low-attention phenotypes.
Methods
Rats trained on the standard 5CSRTT and tested with variable stimulus durations were treated systemically with noradrenergic and/or dopaminergic agents (atomoxetine, methylphenidate, amphetamine, phenylephrine and atipamezole). TVA modelling was applied to estimate visual processing speed for correct and incorrect visual perceptual categorisations, independent of motor reaction times, as measures of attentional capacity.
Results
Atomoxetine and phenylephrine decreased response frequencies, including premature responses, increased omissions and slowed responding. In contrast, methylphenidate, amphetamine and atipamezole sped up responding and increased premature responses. Visual processing speed was also affected differentially. Atomoxetine and phenylephrine slowed, whereas methylphenidate and atipamezole sped up, visual processing, both for correct and incorrect categorisations. Amphetamine selectively improved visual processing for correct, though not incorrect, responses in high-attention rats only, possibly reflecting improved attention.
Conclusions
These data indicate that the application of TVA to the 5CSRTT provides an enhanced sensitivity to capturing attentional effects. Unexpectedly, we found overall slowing effects, including impaired visual processing, following drugs either increasing extracellular noradrenaline (atomoxetine) or activating the α1-adrenoceptor (phenylephrine), while also ameliorating premature responses (impulsivity). In contrast, amphetamine had potential pro-attentional effects by enhancing visual processing, probably due to central dopamine upregulation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Attentional capacity is essential in the interaction with our surroundings, where we need to swiftly select the most relevant sensory information for conscious perception, while discounting irrelevant stimuli (Cohen 2014). Compromised attention is a hallmark of a range of psychiatric disorders, including attention-deficit/hyperactivity disorder (ADHD) (Arnsten 2006), but the neurobiological foundation of attention and how current pharmacological treatments, targeting dopaminergic and noradrenergic neural systems, act on this psychological construct still remains to be understood.
In humans, the theory of visual attention (TVA) model (Bundesen 1990; Bundesen et al. 2005) has been used to study attentional capacity in pharmacological studies and different clinical conditions, including ADHD (Habekost 2015). In this mathematical model, a set of simple equations describes visual attention mechanisms, including processing speed of visual perceptual categorisations, which is a measure of attentional processing independent of motor reaction time (Bundesen 1990; Habekost and Starrfelt 2009; Bundesen et al. 2015). To enable translational studies on attention, the TVA model was recently adapted to model effects of the anti-cholinergic drug scopolamine in mice in a 5-choice serial reaction time task (5CSRTT) (Fitzpatrick et al. 2017)—a well-established task used to assess attention and impulsivity in rodents (Robbins 2002). We now adapt the TVA model to the rat 5CSRTT to study dopaminergic and noradrenergic modulation of attentional processing capacity in rats. We increased attentional load by making target stimuli temporally, as well as spatially, unpredictable using a variable stimulus duration (vSD) regimen. This vSD-5CSRTT combined with the TVA model (TVA-5CSRTT) is equivalent to previous human TVA paradigms with single-letter exposures (Bundesen and Harms 1999) and provides an exact and reliable prediction of attentional capacity with high translational value (Habekost and Starrfelt 2009; Habekost et al. 2014) that has never previously been used to study catecholaminergic modulation of visual attentional processing in rats.
Methylphenidate (MPH), amphetamine (AMPH) and atomoxetine (ATO) are commonly used to treat ADHD (Bidwell et al. 2011). However, the specific, and potentially dissociable, effects of these compounds on attention remain unclear. Furthermore, inconsistencies in their effects exist between preclinical and clinical studies, supporting the need for improved translational investigation. In healthy humans, the stimulant drugs, MPH and AMPH, apparently improve ‘processing speed accuracy’ (Marraccini et al. 2016) and TVA modelling has shown that MPH improves visual processing speed in participants with low baseline attentional performance (Finke et al. 2010). Despite clinical studies indicating cognitive enhancing effects of stimulants, studies using the standard 5CSRTT in intact rodents generally fail to find consistent pro-attentional effects on the accuracy variable after clinically relevant (low to moderate) doses of MPH (Navarra et al. 2008; Milstein et al. 2010) and AMPH (Cole and Robbins 1987; Harrison et al. 1999; Van Gaalen et al. 2006; Loos et al. 2010; Balachandran et al. 2018). However, some have reported stimulants to improve attention in low-attention (Robinson 2012; Caballero-Puntiverio et al. 2017) and high-impulsive (Caprioli et al. 2015) rodents as well as with a variable ITI challenge (Toschi et al. 2021), while impairing attention in rats with profound forebrain NA depletion (Cole and Robbins 1987).
The NA reuptake inhibitor, ATO, consistently improves attention in clinical ADHD studies (Wilens et al. 2006; Faraone and Glatt 2010; Hazell et al. 2011), but only a few studies have assessed attentional effects of acute ATO in healthy humans. These have shown that ATO improves rapid visual information processing (Crockett et al. 2010), but has no effect on attentional performance in the stop-signal reaction time task (Chamberlain et al. 2007) or in a recent human CombiTVA study (Lansner 2022). Likewise, results in rodents have been diverse depending on task and attentional phenotype. While 5CSRTT studies with ATO have shown no effects on accuracy in rodents (Robinson et al. 2008; Fernando et al. 2012; Sun et al. 2012; Pillidge et al. 2014), including an ADHD-like mouse model (Pillidge et al. 2014), some studies have shown improved accuracy in rodents with low attentional baselines (Robinson 2012) or if challenged with vSD (Callahan et al. 2019).
Previous work in rats and monkeys suggest prefrontal cortical noradrenergic α2-adrenoceptor involvement in beneficial effects of MPH and ATO in attention (Arnsten and Dudley 2005; Gamo et al. 2010). Guanifacine, an α2-adrenoceptor agonist, has been approved as an ADHD medication (Bidwell et al. 2011), and blocking the α2-adrenoceptor has produced phenotypes similar to ADHD in monkeys (Arnsten and Li 2005). However, studies are inconsistent in regard to its beneficial effects on attention, as some studies have shown impaired attentional performance after α2-adrenoceptor agonist administration in humans (Smith and Nutt 1996; Coull et al. 2004) and rats (Sirviö et al. 1994; Ruotsalainen et al. 1997; Brown et al. 2012; Fernando et al. 2012), while blocking the α2-adrenoceptor has been reported to improve attention in humans (Mervaala et al. 1993) and rats performing different attention tasks (Sirvio et al. 1993; Koskinen et al. 2003; Lapiz and Morilak 2006; Brown et al. 2012; Bari and Robbins 2013). Previous studies have also indicated a role for the noradrenergic α1-adrenoceptor in attention, for instance by increasing vigilance (Sirviö and MacDonald 1999). Only a few 5CSRTT studies have investigated the role of α1-adrenoceptors in attention, with some indicating α1-adrenoceptor activation to improve attention (Puumala and Sirviö 1997), while others reported no attentional effects (Pattij et al. 2012). In the present study, we used atipamezole (ATI), an α2-adrenoceptor antagonist and phenylephrine (PHEN), an α1-adrenoceptor agonist, to further investigate the role of α2- and α1-adrenoceptors in visual attentional processing.
Despite increasing insights into the role of DA and NA in attention, it is still unknown how these functions translate into quantitative measures of visual attention. In the current study, the TVA model adapted from human studies in attention was applied to the well-established 5CSRTT paradigm in rats to assess the effects of pharmacological challenges of the DA and NE systems on visual attentional processing in healthy rats. We hypothesised that the quantitative parameters assessed in this paradigm reflect mental psychological processes that are differentially modified by pharmacological challenges targeting the DA and NA transmitter systems.
Methods and materials
Animals
Outbred male Lister Hooded rats (N = 24; Charles River, Margate, UK) weighing 280–300 g at the beginning of the experiments were used. Animals were allowed to acclimatise to the animal facility under a 12-h:12-h light cycle (lights off at 7 AM) for a minimum of 7 days before any procedures began. When rats reached a body weight of approximately 300 g, they were food restricted to maintain approximately 90% of their free-feeding weight trajectory (19 g of Purina rodent chow per animal and day; adjusted for reward pellet consumption during testing). Water was available ad libitum and food was given at the end of each day’s testing. This research has been regulated under the Animals (Scientific Procedures) Act 1986 Amendment Regulations 2012 (Project licence PA9FBFA9F held by Dr AL MIlton) following ethical review by the University of Cambridge Animal Welfare and Ethical Review Body.
Drugs
Methylphenidate hydrochloride (1 mg/kg; Johnson Matthey, Edinburgh, UK), atomoxetine hydrochloride (1 mg/kg; Sigma-Aldrich, Dorset, UK), d-amphetamine humisulfate salt (0.2 mg/kg; Sigma-Aldrich, Dorset, UK), atipamezole hydrochloride (0.3 mg/kg; Abcam, Cambridge, UK) and phenylephrine hydrochloride (1 mg/kg; Sigma-Aldrich, Dorset, UK) were dissolved in 0.9% saline to 1 ml/kg fresh on the day of testing.
Doses were determined based on an extensive previous literature employing dose–response studies on the 5CSRTT and relevant behavioural tasks as well as further piloting. In general, we were interested in behaviourally relevant doses and attempted to choose as low doses as possible to avoid disruptive effects and to simulate likely clinical dosage. It is a limitation of the design that we were unable to obtain detailed dose–response data for all compounds; however, this constraint was pragmatically necessary to obtain data on the range of drugs investigated. The 1 mg/kg MPH dose was chosen based on previous dose–response studies using doses ranging around 0.3–3 mg/kg showing moderate doses around 1 mg/kg to improve attention under increased task demand (e.g. Koffarnus and Katz 2011; Berridge et al. 2006; Tomlinson et al. 2014; Navarra et al. 2017), while higher doses potentially would impair performance by inducing disruptive impulsivity (e.g. Milstein et al. 2010). Furthermore, we conducted a dose–response study on the effects of 1 and 3 mg/kg MPH on a vITI-5CSRTT paradigm showing that 1 mg/kg (as opposed to 3 mg/kg) MPH improved performance (more rewards earned) on the 5 s ITI (the ITI used in the present study), while not increasing premature responding to the same extend as 3 mg/kg MPH (Toschi et al. 2021). The 0.2 mg/kg AMPH dose was chosen based on a previous dose–response study showing improved attention in a signal detection task after an equivalent low-dose AMPH, as opposed to higher doses (1.25 mg/kg), which impaired attention (Turner and Burne 2016). We further tested the 0.2 mg/kg AMPH dose in our recent publication on vITI-5CSRTT showing improved attention selectively at short (3 s) ITIs (Toschi et al. 2021). The 1 mg/kg ATO dose was chosen based on dose–response studies using doses ranging around 0.3–3 mg/kg (e.g. Benn and Robinson 2017; Ding et al. 2018; Baarendse and Vanderschuren 2012; Callahan et al. 2019; Fernando et al. 2012; Robinson et al. 2008; Koffarnus and Katz 2011, Tomlinson et al. 2014). We chose the moderate dose of 1 mg/kg ATO, which consistently improves impulsivity (e.g. Toschi et al. 2021; Navarra et al. 2008; Higgins et al. 2020), with some potential to affect accuracy (e.g. Navarra et al. 2008; Tomlinson et al. 2014) without extensively disrupting performance which 3 mg/kg ATO would potentially do (e.g. increased omissions in Koffarnus and Katz 2011). For ATI, we based our dose on previous dose–response studies employing doses in the range of around 0.03–1 mg/kg ATI showing 0.3 mg/kg ATI to improve attention (Sirviö et al. 1993) and stop-signal reaction time (Bari and Robbins et al. 2013). The PHEN dose was based on a previous 5CSRTT paper with extensive data on dose–response (Pattij et al. 2012) and further piloting, where we first tested the 3 mg/kg as it indicated some improvement in Pattij et al., but as this was sedative in our pilot rats, we chose the 1 mg/kg dose, where the rats could perform the task.
5-choice serial reaction time task (5CSRTT)
Apparatus
Details of the behavioural apparatus have been provided previously (Bari et al. 2008). In brief, we used twelve five-choice operant chambers (Med Associates Inc., St. Albans, USA) each contained within a ventilated and sound-attenuated chamber. Each chamber comprised five evenly and distinctly spaced apertures containing an LED light set into a curved wall at the rear of the chamber. On the opposite wall of the chamber, a central food magazine was located, into which 45-mg reward pellets could be delivered (TestDiet 5UTL, Sandown Scientific, Middlesex, UK). Infrared beams located at the entrance of each aperture and the food magazine allowed the detection of nose pokes. The chambers were controlled by computers using WhiskerServer and FiveChoice client software (Cardinal and Aitken, 2010).
Pretraining: 5-choice serial reaction time task (5CSRTT)
All rats were trained in the 5CSRTT as described in detail previously (Bari et al. 2008). In short, animals were trained through progressing training stages (as described in Bari et al. 2008) to detect a brief visual cue appearing pseudorandomnly in one of five apertures of the rear wall of the operant chamber. Each trial was initiated by the rat nose poking into the food magazine and the visual cue is presented after an intertrial interval (ITI) of 5 s. A response was deemed ‘correct’ if the animal nose-poked into the hole with the visual stimulus. A nose-poke response occurring before the appearance of the visual cue was considered ‘premature’, while one occurring in any of the other apertures without the visual cue was considered ‘incorrect’. A failure to respond within 5 s (limited hold) of target presentation was recorded as an ‘omission’. Correct responses were rewarded with one food pellet, while incorrect, premature and omission responses were punished with a time-out (TO) period of 5 s, following which another trial could be initiated. Nose pokes in any of the apertures made after a correct or incorrect response, but prior to reward collection, were deemed ‘perseverative’ but were not signalled by punishment (i.e. TO). Each training and baseline session lasted maximum 100 trials or 30 min, whichever was reached first. In this cohort of rats, a stable baseline performance on the 5CSRTT was reached at training stage 11 (Bari et al. 2008), i.e. stimulus duration of 600 ms and an ITI of 5 s, where > 80% accuracy and < 20% omissions was reached.
Variable stimulus duration challenge (vSD-5CSRTT)
For testing drug effects on behaviour, the rats were challenged with a variable stimulus duration (vSD) schedule with fixed 5 s ITI schedule in a prolonged session of 60 min, or 200 trials, to allow for sufficient trials at each SD. In this schedule, the SDs (75, 150, 300, 600 and 1200 ms) were presented pseudorandomly in blocks of 50, offering both increased and decreased task difficulty within-session compared with the baseline conditions (Fig. S1). Both the variable nature of the schedule and the inclusion of short SD challenges the attention of the rats sufficiently to allow for the detection of potential attention-enhancing drug effects, and the vSD schedule also allows for TVA modelling to be applied. A pilot study was performed to select these SDs that increased the attentional load while still maintaining high motivation levels throughout the session.
Behavioural testing and drug administration
Drugs were administered sub-cutaneously 40 min prior to testing the animals on a vSD session of the 5CSRTT. The experiment consisted of two separate within-subject cross-over Latin-square designs, to control for training and cross-over effects. These two Latin-square designs, and each of the testing days, were separated by at least 3 days of washout and re-baseline sessions. In Latin-square 1: vehicle, AMPH (0.2 mg/kg), MPH (1 mg/kg) and ATO (1 mg/kg) were administered. In Latin-square 2: vehicle, ATI (0.3 mg/kg) and PHEN (1 mg/kg) were administered. Out of the initial 24 rats, 23 rats took part in the Latin-square 1 design, while 22 rats took part in the following Latin-square 2 design. This was due to two rats experiencing spontaneuous seizures: one rat being euthanised during pretraining, and another rat being euthanised between the two experiments.
TVA modelling of 5-CSRTT data (TVA-5CSRTT)
Based on the framework of human TVA (Bundesen 1998; Bundesen and Harms 1999; Bundesen et al. 2005; Habekost 2015) and the recently developed TVA model for mouse 5CSRTT (Fitzpatrick et al. 2017), we developed a four-parameter TVA model adapted to the rat 5CSRTT. This makes it possible, for the first time, to estimate visual perceptual processing speed independent of motor reaction times from rat 5CSRTT data. TVA describes visual attention as a parallel processing race where different visual perceptual categorisations of a stimulus compete for entrance into visual short-term memory. For the rat 5CSRTT, we assume that one correct categorisation races against four incorrect categorisations. This is an extension of the TVA model for mouse 5CSRTT where only correct categorisations were modelled as only very few incorrect responses were observed for the mice. The rat TVA model thus focuses in greater detail on the perceptual processes compared with the mouse TVA model.
We assume that the rat makes a correct motor response if a correct visual perceptual categorisation finishes first and before time \(\tau {,}\) where \(\tau\) is the stimulus duration. In contrast, the rat makes an incorrect motor response if an incorrect visual perceptual categorisation finishes first and before time \(\tau\). The sampling time for both correct and incorrect categorisations are assumed exponentially distributed with rate parameter \({\nu }_{\mathrm{c}}\) and \({\nu }_{\mathrm{i}}\), respectively, but delayed by a constant \({t}_{0}\), which is the time it takes the rat to orient toward the stimulus and initiate the race. If no correct or incorrect categorisation is made before time \(\tau {,}\) we assume that with a certain probability (\({p}_{\mathrm{g}}\)) the rat guesses randomly among the five possible responses. An omission occurs if no categorisation is made before time \({\tau}\) and the rat does not choose to make a random response. In total, the TVA model for rat 5CSRTT has four free parameters: \({\nu }_{\mathrm{c}}\), \({\nu }_{\mathrm{i}}\), \({t}_{0}\) and \({p}_{\mathrm{g}}\). The probabilities of making a correct response, \({p}_{\mathrm{c}}\), an incorrect response, \({p}_{\mathrm{i}}\), or an omission, \({p}_{\mathrm{o}}\), are calculated as follows:
If \(\begin{aligned}p_{\mathrm c}=\int_0^{\tau-t_0}\nu_{\mathrm c}e^{-\nu_{\mathrm c}t}\cdot e^{-4\nu_{\mathrm i}t}dt+e^{-\nu_{\mathrm c}(\tau-t_0)}\cdot e^{-4\nu_{\mathrm i}\left(\tau-t_0\right)}\cdot p_{\mathrm g}\cdot\frac{1}{5}=\frac{\nu_{\mathrm c}}{\nu_{\mathrm c}+4\nu_{\mathrm i}}\left(1-e^{-\left(\nu_{\mathrm c}+4\nu_{\mathrm i}\right)\cdot\left(\tau-t_0\right)}\right)+e^{-\left(\nu_{\mathrm c}+4\nu_{\mathrm i}\right)\cdot\left(\tau-t_0\right)}\cdot p_{\mathrm g}\cdot\frac{1}{5}\\ p_{\mathrm i}=\int_0^{\tau-t_0}\nu_{\mathrm i}e^{-4\nu_{\mathrm i}t}\cdot e^{-\nu_{\mathrm c}t}dt+e^{-\nu_{\mathrm c}(\tau-t_0)}\cdot e^{-4\nu_{\mathrm i}\left(\tau-t_0\right)}\cdot p_{\mathrm g}\cdot\frac{1}{5}=\frac{\nu_{\mathrm i}}{\nu_{\mathrm c}+4\nu_{\mathrm i}}\left(1-e^{-\left(\nu_{\mathrm c}+4\nu_{\mathrm i}\right)\cdot\left(\tau-t_0\right)}\right)+e^{-\left(\nu_{\mathrm c}+4\nu_{\mathrm i}\right)\cdot\left(\tau-t_0\right)}\cdot p_{\mathrm g}\cdot\frac{1}{5}\\p_{\mathrm o}=e^{-\left(\nu_{\mathrm c}+4\nu_{\mathrm i}\right)\cdot\left(\tau-t_0\right)}\cdot\left(1-p_{\mathrm g}\right)\end{aligned}\)
If \(\tau \leq {t}_{0}\), then
TVA parameters were estimated by performing a maximum-likelihood fitting procedure using the Nelder-Mead simplex optimisation algorithm in Matlab 2017. Figure 1A shows the TVA model fitted to representative data from a rat performing a vSD-5CSRTT challenge session. We assessed the goodness-of-fit based on a pseudo-R2 (Nagelkerke 1991), which was calculated as
in which LL0 is the log-likelihood of the restricted model, LL1 is the log-likelihood of a less restricted model, and n is the number of trials. We set the LL1 as the log-likelihood of the TVA model and LL0 as the likelihood of a null model with only \({p}_{\mathrm{g}}\) as parameter. Thus, this pseudo-R2 reflects the proportion of variation explained by the TVA model relative to a null model with constant propabilities of making a correct response, an incorrect response and an omission across all stimulus durations. For Latin-square 1, the average \({R}^{2}\) s were 0.69, 0.69, 0.69 and 0.57 for vehicle, AMPH, MPH and ATO, respectively. For the Latin-square 2, the average \({R}^{2}\) s were 0.71, 0.72 and 0.66 for vehicle, ATI and PHEN, respectively. Altogether, this shows that the TVA model explains a large proportion of the variation in the data.
Effects of stimulant and non-stimulant drugs on modelled TVA-5CSRTT parameters. A The TVA model fitted to representative data from a rat performing a vSD challenge session. B–D presents results for Latin-square 1 (LS1, top panels) and Latin-square 2 (LS2, bottom panels) with TVA-modelled parameters willingness to guess (pg) (B), visual processing speed for correct responses (νc) (C) and visual processing speed for incorrect responses (νi) (D). LA, low-attention rats; HA, high-attention rats; ATO, atomoxetine; MPH, methylphenidate; AMPH, amphetamine; PHEN, phenylephrine; ATI, atipamezole. Results are represented as mean ± SEM; ***p < 0.001; **p < 0.01; *p < 0.05
Data analysis
Main 5CSRTT parameters of interest were correct responses, choice accuracy (% correct/(correct + incorrect)), omissions, premature responses, response latency to make a correct response after the onset of the target stimulus (ms) and latency to collect food from the magazine after a correct trial (ms). We also analysed incorrect responses, perseverative responses and response latency to make an incorrect response (as shown in supplementary Figs. S2-S3). TVA parametres of interest were visual processing speed for correct (\({\nu }_{\mathrm{c}}\)) and incorrect (\({\nu }_{\mathrm{i}}\)) responses and guessing probability (\({p}_{\mathrm{g}}\)). Based on mean accuracy score averaged across 13 separate days of baseline training, we classified low attention (LA) and high attention (HA) subgroups as the lower and upper 30th percentiles, respectively (n = 7 per subgroup).
Visualisation and statistical tests were performed with RStudio, version 1.2.1335 (RStudio, Inc.). Response frequencies (correct and incorrect responses, omissions and premature responses) were square-root transformed, latencies were log transformed and probabilities (accuracy and pg) were arcsin transformed to ensure normality, as confirmed with a quantile–quantile plot of residuals. Within each Latin-square design, differences in drug effects on the above parameters were analysed using linear mixed-effects model analysis with the lmer package in R. The model contained either one fixed factor (dose) or two fixed factors (drug and phenotype) and one random factor (subject; to account for individual differences between rats). When relevant, further analyses were performed by conducting separate multilevel models on ‘drug’ for each phenotype. These analyses were followed by post hoc Dunnet’s corrected pairwise comparisons with vehicle. We also ran models with two fixed factors being drug and SD, and three fixed factors being drug, phenotype and SD, but without finding significant interactions. Because drug effects were not dependent on SD, it is not included here. Linear correlations between TVA parameters and standard parameters were performed using Pearson’s coefficient r. Significance was set at α = 0.05.
Results
No drugs affected accuracy, but ATO and PHEN decreased correct and incorrect responses, increased omissions and slowed responding. In contrast, ATI, MPH and AMPH did not affect correct or incorrect response frequencies, but speed up responding and increased premature responding. TVA-modelled visual processing speed was also affected differentially. While ATO and PHEN slowed, ATI, MPH and AMPH speed up, visual processing, both for correct and incorrect categorisations. AMPH selectively improved visual processing for correct, not incorrect, responses in high-attention rats only, reflecting improved attention. For readability, statistical details on pairwise comparisons are presented in Tables 2 and 3.
Performance with variable stimulus duration (vSD) challenge
Behavioural performance in vehicle-treated rats during the vSD challenge was examined (Fig. S1) (see supplementary material for statistical details), but in short, performance improved with increasing SDs, i.e. increasing accuracy and correct responses as well as decreasing incorrect responses, omissions and response latencies, while leaving reward collection latency unaffected. Thus, attentional performance overall improved with increasing SDs and was dependent on SD, as expected (Fitzpatrick et al. 2017).
Correlation between standard and TVA-modelled parameters in the 5CSRTT
To investigate how TVA-modelled parameters relate to standard 5CSRTT parameters, we analysed correlations in VEH groups averaged across Latin-square experiments (mean VEH; Tables 1 and S1) (for statistical details, see Table 1). The TVA-modelled νc parameter correlated positively with correct responses and negatively with omissions, while it did not significantly correlate with any other parameters such as, e.g. latencies, accuracy and premature responses. This indicated higher νc to be associated with enhanced correct responding and task engagement independent of errors and motor reaction times. TVA-modelled νi correlated negatively with omissions and positively with perseverative nosepokes. Thus, higher νi was associated with higher task engagement and increased (unrewarded) perseverative responding. pg correlated positively with incorrect responses and negatively with accuracy and omissions. Thus, higher willingness to ‘guess’ was associated with more errors, lower accuracy and higher task engagement. Furthermore, νc and νi correlated positively, and pg correlated positively with both νc and νi, reflecting that higher νc is associated both with higher νi and willingness to guess (Table S1). Drug treatments affected these associations differentially, as described to some detail below and in supplementary material (Table S1).
Effects of atomoxetine, methylphenidate and amphetamine on modelled TVA parameters
For νc (Fig. 1C and Table 2), we found a significant effect of drugs overall (F3, 618 = 226.09, p < 0.0001); νc was significantly decreased by ATO and increased by both MPH and AMPH. We found a significant drug × phenotype interaction (F3, 372 = 8.77, p < 0.0001), and significant main effect of drugs (F3, 372 = 139.77, p < 0.0001), but not of phenotype. Significant drug effects were present both in LA (F3, 186 = 130.42, p < 0.0001) and HA (F3, 186 = 46.06, p < 0.0001) rats. In both LA and HA rats, νc was significantly decreased by ATO and increased by MPH, while AMPH significantly increased νc only in HA rats, not in LA rats.
For νi (Fig. 1D and Table 2), we found a significant effect of drugs overall (F3, 618 = 90.02, p < 0.0001); ATO significantly decreased νi, while both MPH and AMPH significantly increased νi. We found a significant drug × phenotype interaction (F3, 372 = 23.23, p < 0.0001) and significant main effect of drug (F3, 372 = 66.07, p < 0.0001), but not of phenotype. Significant drug effects were present in LA (F3, 186 = 58.05, p < 0.0001) and HA (F3, 186 = 10.37, p < 0.0001) rats. In both LA and HA rats, νi was significantly decreased by ATO and increased by MPH. AMPH did not affect νi in neither LA nor HA rats.
For pg (Fig. 1B and Table 2), we found a significant effect of drugs overall (F3, 618 = 877.31, p < 0.0001); ATO significantly decreased, while MPH and AMPH significantly increased, the probability of guessing. We found a significant drug × phenotype interaction (F3, 372 = 9.48, p < 0.0001), and a significant main effect of drug (F3, 372 = 617.51, p < 0.0001), but not of phenotype. Significant drug effects were present both in LA (F3, 186 = 351.76, p < 0.0001) and HA (F3, 186 = 274.35, p < 0.0001) rats. In both LA and HA rats, guessing probability was decreased by ATO and increased by MPH and AMPH.
Some drugs affected the associations between TVA-modelled and standard parameters (Figs. S6-S7 and Table S1). In short, ATO induced a positive correlation between νc and errors committed and removed correlation with omissions, while producing an association between higher νi and reduced inhibitory control. AMPH treatment produced an association of higher νc with higher accuracy and fewer errors. MPH did not affect correlations.
Effects of atipamezole and phenyphrine on modelled TVA parameters
For νc (Fig. 1C and Table 3), we found a significant effect of drugs overall (F2, 431.36 = 65.80, p < 0.0001); νc was significantly decreased by PHEN and increased by ATI. We found no drug × phenotype interaction or main effect of phenotype, but there was a significant main effect of drugs (F2, 249.37 = 57.61, p < 0.0001).
For νi (Fig. 1D and Table 3), we found a significant effect of drugs overall (F2, 431.68 = 80.68, p < 0.0001); νi was significantly decreased by PHEN and increased by ATI. We found a significant drug × phenotype interaction (F2, 249.55 = 24.75, p < 0.0001) and significant main effect of drug (F2, 249.55 = 125.25, p < 0.0001), but not of phenotype. Significant drug effects were present in LA (F2, 111.27 = 126.56, p < 0.0001) and HA (F2, 138 = 20.90, p < 0.0001) rats. Both in LA and HA rats, νi was significantly decreased by PHEN and increased by ATI.
For guessing probability (Fig. 1B and Table 3), we found a significant effect of drugs overall (F2, 431.21 = 109.13, p < 0.0001); pg was significantly decreased by PHEN and increased by ATI. We found a significant main effect of drug (F2, 249.57 = 142.26, p < 0.0001), but no drug × phenotype interaction or main effect of phenotype.
Some drugs affected the associations between visual processing speed and other parameters (Figs. S6-S7 and Table S1). Like AMPH, ATI induced a positive correlation between νc and accuracy. ATI also produced a positive correlation between νc (and νi) and reward magazine perseveration. PHEN did not significantly change correlations.
Effects of atomoxetine, methylphenidate and amphetamine on standard vSD-5CSRTT parameters
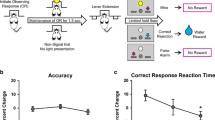
No drugs affected accuracy (Fig. 2A and Table 2), but there was a main effect of phenotype (F1, 12 = 7.52, p = 0.018). This confirms that LA rats had significantly lower accuracy than HA rats, irrespective of treatment.
Effects of stimulant and non-stimulant drugs on standard 5CSRTT parameters. A–D presents results for Latin-square 1 (LS1, top panels) and Latin-square 2 (LS2, bottom panels) with standard 5CSRTT parameters accuracy (A), correct responses (B), omissions (C), latency to respond correctly (D), reward collection latency (E) and premature responses (F). LA, low-attention rats; HA, high-attention rats; ATO, atomoxetine; MPH, methylphenidate; AMPH, amphetamine; PHEN, phenylephrine; ATI, atimpamezole. Results are represented as mean ± SEM; ***p < 0.001; **p < 0.01; *p < 0.05; #p < 0.1
Drug treatment significantly affected correct responses (Fig. 2B and Table 2) (F3, 434 = 36.08, p < 0.0001); ATO significantly reduced correct responses, while MPH and AMPH had no effects. We found a significant drug × phenotype interaction (F3, 260 = 3.79, p = 0.011) as well as a main effect of drug (F3, 260 = 20.58, p < 0.001) and a trending effect of phenotype (F1, 12 = 4.56, p = 0.054). Significant drug effects were present in LA (F3, 130 = 18.71, p < 0.0001) and HA (F3, 130 = 4.05, p = 0.0086) rats; ATO decreased number of correct responses in LA rats, not HA, rats.
For omissions (Fig. 2C and Table 2), we found a significant effect of drugs overall (F3, 434 = 161.34, p < 0.0001), where ATO significantly increased number of omissions, while MPH and AMPH significantly decreased it. We found a trending drug × phenotype interaction (F3, 260 = 2.36, p = 0.072) and a significant main effect of drugs (F3, 260 = 92.06, p < 0.0001), but not of phenotype. Significant drug effects were present in LA (F3, 130 = 51.21, p < 0.0001) and HA (F3, 130 = 44.71, p < 0.0001) rats; ATO increased omissions in LA and HA rats, while MPH decreased omissions in HA rats and trended towards doing so in LA rats. On the other hand, AMPH only decreased omissions in HA rats, without affecting LA rats.
For latency to respond correctly (Fig. 2D and Table 2), we found a significant effect of drugs overall (F3, 433.01 = 69.12, p < 0.0001); ATO significantly prolonged correct latency, while MPH and AMPH significantly speeded it. When investigating for phenotype dependency, we found a significant main effect of drug (F3, 259.02 = 30.84, p < 0.0001), but no drug × phenotype interaction and no main effect of phenotype.
For reward collection latency (Fig. 2E and Table 2), we found a significant effect of drugs overall (F3, 433 = 104.18, p < 0.0001); collection latency was significantly lengthened by ATO and shortened by AMPH, while MPH had no effect. When investigating for phenotype dependency, we found a significant main effect of drug (F3, 259 = 47.20, p < 0.0001), but no drug × phenotype interaction and no main effect of phenotype.
For premature reponses (Fig. 2F and Table 2), we found a significant effect of drugs overall (F3, 618 = 430, p < 0.0001); ATO significantly decreased premature responses, while MPH and AMPH significantly increased prematures. We found a significant drug × phenotype interaction (F3, 372 = 14.91, p < 0.0001) and significant main effect of drug (F3, 372 = 188.00, p < 0.0001), but not of phenotype. Significant drug effects were present in LA (F3, 186 = 176.5, p < 0.0001) and HA (F3, 186 = 43.37, p < 0.0001) rats. In both LA and HA rats, premature responses were significantly decreased by ATO and increased by MPH and AMPH.
Effects of atipamezole and phenylephrine on standard vSD-5CSRTT parameters
No drugs affected accuracy (Fig. 2A and Table 3), but there was a main effect of phenotype (F1, 10.5 = 12.97, p = 0.0045), confirming that LA rats had significantly lower accuracy than HA rats irrespective of treatment.
For correct responses (Fig. 2B and Table 3), we found a significant effect of drugs overall (F2, 302.7 = 9.42, p < 0.0001); PHEN significantly reduced number of correct responses, while ATI had no effect. We found a significant drug × phenotype interaction (F2, 175.01 = 3.48, p = 0.033) as well as a main effect of drug (F2, 175.01 = 12.55, p < 0.001) and phenotype (F1, 11.18 = 6.03, p = 0.032). Significant drug effects were present in LA rats (F2, 78.15 = 11.00, p < 0.0001), but not HA rats (F2, 96 = 2.02, p = 0.14); PHEN decreased the number of correct responses in LA rats, without affecting HA rats.
For omissions (Fig. 2C and Table 3), we found a significant effect of drugs overall (F2, 301.67 = 32.25, p < 0.0001). PHEN significantly increased number of omissions, while ATI significantly decreased it. We found a trending drug × phenotype interaction (F2, 173.75 = 2.90, p = 0.058) and a significant main effect of drugs (F2, 173.75 = 36.84, p < 0.0001), but not of phenotype. Significant drug effects were present in LA rats (F2, 77.48 = 18.22, p < 0.0001) and HA rats (F2, 96 = 24.14, p < 0.0001); PHEN increased omissions in HA rats, not LA rats, while ATI decreased omissions both in LA and HA rats.
For correct response latency (Fig. 2D and Table 3), we found a significant effect of drugs overall (F2, 300.54 = 19.53, p < 0.0001); ATI significantly decreased response latency, while PHEN increased it. When investigating phenotype dependency, we found a significant main effect of drug (F2, 173.51 = 12.39, p < 0.0001), a trending main effect of phenotype (F1, 10.23 = 3.63, p = 0.085), but no drug × phenotype interaction.
For reward collection latency (Fig. 2E and Table 3), we found a significant effect of drugs overall (F2, 301.1 = 19.50, p < 0.0001); PHEN significantly slowed collection latency, while ATI had no effect. When investigating for phenotype dependency, we found a significant main effect of drug (F2, 173.14 = 17.04, p < 0.0001), but no drug × phenotype interaction and no main effect of phenotype.
For premature reponses (Fig. 2F and Table 3), we found a significant effect of drugs overall (F2, 431.49 = 93.27, p < 0.0001), with premature responses being significantly decreased by PHEN and increased by ATI. We found a significant drug × phenotype interaction (F2, 249.57 = 6.80, p = 0.0013) and a significant main effect of drug (F2, 249.57 = 110.00, p < 0.0001), but not of phenotype. Significant drug effects were present in LA (F2, 111.33 = 42.7, p < 0.0001) and HA (F2, 138 = 80.66, p < 0.0001) rats. Premature responses were decreased by PHEN in HA, not in LA, and were increased by ATI in both LA and HA rats.
Results summary
Main results are summarised in Table 4.
Discussion
To understand whether stimulant and non-stimulant drugs specifically affect visual attentional processing, we adapted the human TVA model (Bundesen 1990; Bundesen and Harms 1999; Bundesen et al. 2005) to the rat 5CSRTT. We administered drugs relevant for ADHD pharmacological therapies, i.e. AMPH, MPH and ATO, as well as the relatively selective noradrenergic agents, ATI and PHEN targeting the α2- and α1-adrenoceptors, respectively. While no drugs affected accuracy, dissociable effects were observed on TVA-modelled visual processing speed. ATO and PHEN surprisingly slowed, whereas ATI and MPH speeded up visual processing, both for correct and incorrect decisions. Thus, in the present study, ATO produced attentional deficits possibly due to slowed visual processing. In contrast, AMPH selectively improved visual processing for correct, not incorrect, decisions in HA rats, reflecting improved attention in high performers—surprisingly, without affecting low performers. Overall, this suggests catecholaminergic modulation to be involved in visual attentional processing.
Effects of stimulant drugs on attention; MPH versus AMPH
That AMPH and MPH did not affect accuracy is generally consistent with previous 5CSRTT studies in healthy rodents treated with comparable (low to moderate) doses of MPH (Navarra et al. 2008; Milstein et al. 2010; Fernando et al. 2012; Pattij et al. 2012; Hauser et al. 2017) and AMPH (Cole and Robbins 1987; Harrison et al. 1999; Van Gaalen et al. 2006; Loos et al. 2010; Balachandran et al. 2018; Higgins et al. 2020). Although a few studies have reported these drugs to improve accuracy during increased task demand and/or in low-attention rats (Koffarnus and Katz 2011; Robinson 2012; Caballero-Puntiverio et al. 2017; Toschi et al. 2021) as well as in high-impulsive rats (Caprioli et al. 2015), we did not observe any effects on accuracy in LA rats. Studies using other rodent attentional tasks have reported beneficial attentional effects of comparable doses of AMPH or MPH (Berridge et al. 2006, 2012; Tomlinson et al. 2014; Turner and Burne 2016; Navarra et al. 2017; MacQueen et al. 2018; Caballero-Puntiverio et al. 2019; Young et al. 2020) as well as in a genetic ADHD-like mouse model (Nilsson et al. 2018) and prefrontal cortex lesioned animals (Chudasama et al. 2005), although not in all studies (Ding et al. 2018; Caballero-Puntiverio et al. 2020). Thus, while results have been inconsistent, low doses of stimulant drugs potentially facilitate certain attentional processes, which may not be fully captured by standard 5CSRTT attentional parameters such as accuracy. Supporting this, AMPH and MPH did induce fewer omissions and faster responding, which may reflect improved attention (Lezak et al. 2012), general arousal (e.g. Rapoport et al. 1980; Berridge 2006) or reduced fatigue (Choi and Raymer 2019).
To understand whether MPH and AMPH specifically affect visual attentional processing, we applied the TVA model. Human TVA modelling has shown MPH to improve visual processing speed in participants with poor baseline attention (Finke et al. 2010). In our study, MPH increased visual processing speed both for correct and incorrect responses in LA and HA rats, indicating a more general arousal effect rather than a specific attentional effect. On the other hand, AMPH did not affect visual processing in LA rats, but selectively improved attention in HA rats, as visual processing speed was enhanced for correct responses without affecting it for incorrect responses. Correspondingly, omissions were also reduced by AMPH only in HA rats. Further supporting an AMPH-induced change in attentional capacity is that AMPH produced a positive correlation between νc and accuracy; thus, higher accuracy was associated with higher νc—this was not the case for MPH (or VEH) treated rats. Thus, in our study, AMPH improved attentional capacity in high performers specifically indicating that AMPH treatment can improve visual attentional processing, although not necessarily in low-attention individuals as might have been expected given its efficacy in treating ADHD.
While attentional effects of stimulants have been variable in previous studies, it is a consistent finding in rodent 5CSRTT studies that both AMPH and MPH induce impulsivity, as well as hyperactivity (e.g. Cole and Robbins 1987; Harrison et al. 1999; Pattij et al. 2007; Navarra et al. 2008; Baarendse and Vanderschuren 2012; Higgins et al. 2020; Toschi et al. 2021), consistent with the present study.
Altogether, the stimulant-induced effects indicate heightened arousal or overall behavioural activation, possibly due to enhanced motivation; which is also supported by an increased willingness to ‘guess’ under uncertainty, when no information is available. Worth noting, in contrast to AMPH-induced improving effects on omissions and visual attentional processing, the speeding effect on reaction times was evident only in LA rats, indicating differential underlying neural mechanisms. Thus, AMPH-induced improved attentional processing, and decreased omissions are not directly associated with faster responding, indicating that decreased omissions may reflect attentional engagement and task motivation, while faster responding may be more associated with locomotor activation, i.e. hyperactivity. This is further supported by the fact that νc did not correlate with latencies, but rather it correlated negatively with omissions and positively with correct responses.
Our observation that AMPH facilitates attentional processing adds to previous studies reporting pro-attentional effects of psychostimulants in healthy humans (e.g. metanalyses (Marraccini et al. 2016)) and ADHD patients (e.g. Losier et al. 1996; Faraone and Biederman 2002; Faraone and Buitelaar 2010). That AMPH has a higher degree of pro-attentional effects than MPH supports a meta-analysis of human studies arriving at the same conclusion (Faraone and Buitelaar 2010). Furthermore, it is in line with a recent study showing AMPH, not MPH or ATO, to improve visual processing speed in a human continuous performance task (CPT) with TVA modelling—a novel CombiTVA paradigm (Lansner 2022). These results highlight the importance of refining the study of stimulant drug effects on attention with tools, such as the TVA model, that can measure attentional effects previously reported in humans, but not directly captured by standard 5CSRTT parameters.
Effects of non-stimulant versus stimulant drugs on attention: ATO versus MPH/AMPH
For nearly all parameters, ATO-induced behavioural effects contrasted with MPH- and AMPH-induced effects. ATO slowed visual processing, as modelled by TVA, both for correct and incorrect responses, indicating diminished attentional capacity. This was not reflected in accuracy though, which was, like MPH and AMPH, not affected by ATO, in line with previous rodent attention studies using similar doses of ATO (Blondeau and Dellu-Hagedorn 2007; Robinson et al. 2008; Tsutsui-Kimura et al. 2009; Fernando et al. 2012; Sun et al. 2012; Pillidge et al. 2014; Ding et al. 2018; Higgins et al. 2020). However, some studies have shown ATO-induced attentional improvement during vSD attentional challenge in rats and mice (Caballero-Puntiverio et al. 2019, 2020; Callahan et al. 2019) or in poorly performing rats (Robinson 2012; Tomlinson et al. 2014) and, on the other hand, attentional impairment in highly performing rats (Tomlinson et al. 2014) and under a variable ITI challenge (Higgins et al. 2020; Toschi et al. 2021).
In humans, only a few studies have assessed attentional effects of acute ATO. ATO improves rapid visual information processing (Crockett et al. 2010), but has no effect on attentional performance in a stop-signal reaction time task (Chamberlain et al. 2007) or in a recent human CombiTVA study, which also showed reduced short-term memory capacity after ATO (Lansner 2022). However, a positive association has been found between the dopamine beta-hydroxylase genotype (responsible for NA synthesis) and sustained attention in human TVA-modelled CPT (Shalev et al. 2019). Thus, acute ATO treatment has shown inconsistent effects on attentional parameters depending on attentional load, task, baseline performance and presumably also dose.
The present study shows detrimental effects of ATO on visual attentional processing. However, the observed ATO-induced slowed visual processing may not specifically indicate poor attention, but could be secondary to a general behavioural slowing as νc and νi are both slowed and accompanied by slowed motor responding and reward collection. Accordingly, ATO also generally reduced correct, incorrect and premature reponses as well as increased omissions and, consequently, a reduced willingness to ‘guess’ (i.e. random responses). Thus, in addition to possibly reflecting inattentiveness, general behavioural slowing may also reflect lack of motivation and general hypoactivity (i.e. mild sedation). ATO-induced slowed reaction times has been reported in previous rodent 5CSRTT studies (Blondeau and Dellu-Hagedorn 2007; Bari et al. 2009; Jentsch et al. 2009; Baarendse and Vanderschuren 2012; Fernando et al. 2012; Robinson 2012; Sun et al. 2012; Benn and Robinson 2017; Ding et al. 2018; Caballero-Puntiverio et al. 2019), although not in others (Robinson et al. 2008; Tsutsui-Kimura et al. 2009; Koffarnus and Katz 2011; Paterson et al. 2011, 2012; Pillidge et al. 2014; Liu et al. 2015). In humans, ATO generally does not affect reaction times (Shang and Gau 2012; Ni et al. 2013, 2016; Bédard et al. 2015). In fact, some studies in humans contrast rodent studies, reporting ATO to actually shorten reaction times (Gau and Shang 2010; Wehmeier et al. 2011, 2012; Kratz et al. 2012; Fan et al. 2017). That ATO may decrease motivation is supported by the slowed reward collection by ATO in the present study and previous 5CSRTT studies in rodents (Navarra et al. 2008; Pillidge et al. 2014), as well as ATO-induced dimished motivation for effort-demanding reward collection in a progressive ratio test independent of locomotor activity (Higgins et al. 2020). Consistently, NA is an appetite-suppressant in rodents (Rinaman 2011; Roman et al. 2016) and humans with ADHD (Hah and Chang 2005; Kratochvil et al. 2011; Walker et al. 2015). Taken together, slowing effects of ATO in the present study may be due to hypoactivity (i.e. mild sedation) in conjunction with decreased motivation for reward.
The opposing effects of MPH/AMPH and ATO are consistent with dissociable behavioural effects on the 5CSRTT of ATO and MPH in striatal regions (Economidou et al. 2012), and also of increased DA stimulating motivation (Achterberg et al. 2016; Yohn et al. 2016a) and increased NA diminishing motivation, possibly, to some degree, through ATO-induced serotonergic modulation (Gallezot et al. 2011; Mathes et al. 2013; Rosenberg et al. 2013; Ding et al. 2014; Yohn et al. 2016b, a)—although presumably not in the PFC (Bymaster et al. 2002). Furthermore, similar to MPH and AMPH, ATO increases extracellular NA and DA levels in the prefrontal cortex (Bymaster et al. 2002). However, in contrast to stimulants, ATO does not affect (Bymaster et al. 2002; Heal et al. 2009) or may even decrease (Yohn et al. 2016a), DA release in striatal regions, which may explain why ATO reduces response rate and speed, as opposed to stimulants. Additonally, via its actions on NA mediated by alpha-1 receptors, ATO can also indirectly increase prefrontal cortical acetylcholine at 1 mg/kg, which may thus contribute to ATO’s working memory enhancing effects (Tzavara et al. 2006). Therefore, although ATO is a highly specific NET inhibitor, some of its effects may ultimately be mediated by its indirect actions on other neurotransmitters, although it seems unlikely that any pro-cholinergic actions of ATO would produce slowed visual processing as seen here. Moreover, Bari et al. (2011) provided pharmacological evidence that the ameliorative effects on impulsive responding produced by intra-PFC ATO were mediated by noradrenergic rather than dopaminergic mechanisms.
Our results suggest catecholaminergic modulation to be implicated in visual attentional processing, and that it may play a complementary role to the cholinergic system in attention, as we previously showed anti-cholinergic treatment to reduce TVA-modelled visual processing speed (Fitzpatrick et al. 2017). The slowing effects of ATO actually are similar to some anti-cholinergic effects seen in the mouse TVA-5CSRTT, where scopolamine slowed visual processing as well as reaction times and reward collection, while also increasing omissions. However, in contrast to ATO, scopolamine increased premature responses, indicating separate underlying actions.
Involvement of α1- and α2-adrenoceptors in attention
As ATO increases extracellular NA globally, it was relevant to investigate the role of specific adrenoceptor subtypes. Previous studies have indicated an attentional role for the high-affinity and abundant α1-adrenoceptors (Berridge 2006; Spencer et al. 2012), which, in the present study, was activated by PHEN. That PHEN had similar behavioural effects to ATO across nearly all parameters, including slowed visual processing, suggests that, at least partly, ATO’s effects are mediated via α1-adrenoceptor activation, but this was not directly tested in this study. The lack of PHEN-induced effects on accuracy, and its slowing effect on visual processing, somewhat contradicts previous studies claiming that improvements on attention (in rats) following dopamine D3 agonist- (Marshall et al. 2019) or low-dose MPH administration (Berridge et al. 2006, 2012; Navarra et al. 2017) were dependent on activation of the α1-adrenoceptor (Berridge et al. 2012). Similar claims of a pro-cognitive effect of activating the α1-adrenoceptor were brought forward by studies showing that the putative α1-adrenoceptor agonist, St-587, improves accuracy in the 5CSRTT with shortened SD (Puumala et al. 1997) and that α1-adrenoceptor antagonism impairs 5CSRTT accuracy (Puumala et al. 1997) and go accuracy in a rat stop-signal reaction time task (Bari and Robbins 2013) while slowing responding (Hahn and Stolerman 2005; Bari and Robbins 2013). Thus, our data does not provide evidence for the claim that α1-adrenoceptor activation increases vigilance as previously suggested (Sirviö and MacDonald 1999); instead, it induced a general behavioural hypoactivity, similar to ATO. Although we had chosen a dose of PHEN previously shown not to slow rats (Pattij et al. 2012), we cannot rule out that we would have seen potential beneficial effects had we tested lower doses of PHEN.
Cortical NA depletion does not affect choice accuracy in rat 5CSRTT (Ruotsalainen et al. 1997), but impairs performance when attentional demand is increased (Carli et al. 1983; Milstein et al. 2007; Cole and Robbins 1992). This impairment is exacerbated by α2 agonism (Milstein et al. 2007), indicating potentially benefical effects of blocking the α2-adrenoceptor. A few previous studies have shown ATI to improve 5CSRTT accuracy (Sirvio et al. 1993; Koskinen et al. 2003) or to have no effect on it (Sirviö et al. 1994). In other rodent attentional tasks, α2-adrenoceptor antagonism improves sustained attention in a stop-signal reaction time task (Bari and Robbins 2013), auditory cue detection (Brown et al. 2012) and attentional set shifting (Devauges and Sara 1990; Lapiz and Morilak 2006). However, our data do not fully support a selective pro-attentive effect of α2-adrenoceptor antagonism, rather ATI prompts a general behavioural activation similar to that of MPH, both speeding up visual processing and reaction times, while decreasing omissions and diminishing inhibitory control. This is in line with previous studies showing α2-adrenoceptor antagonism to increase locomotor activity (Niittykoski et al. 1998) and impair inhibitory response control (Sirviö et al. 1994; Ruotsalainen et al. 1997; Koskinen et al. 2003; Sun et al. 2010) in rats and healthy human subjects (Swann et al. 2005, 2013; Sun et al. 2010); effects possibly mediated by the PFC as shown in monkeys (Ma et al. 2005).
Conclusions
Unexpectedly, we captured overall slowing effects, including impaired visual processing, of drugs increasing extracellular noradrenaline (ATO) or activating the α1-adrenoceptor (PHEN). In contrast, we found overall speeding effects of drugs enhancing both dopaminergic and noradrenergic transmission (MPH, AMPH and ATI). We conclude that, while ATO decreases impulsivity, which is presumably a significant part of its therapeutic effect in ADHD, it may also produce detrimental effects such as general behavioural slowing and diminished visual processing, at least after acute dosing. In contrast, a single low dose of amphetamine had potential pro-attentional effects by enhancing visual processing, probably due to central dopamine upregulation.
That no drugs affected accuracy, but had differential effects on visual perceptual processing speed, suggests that more temporally dynamic and detailed attentional measures, like TVA-modelled parameters, are needed to fully capture attentional effects as an addition to standard parameters, such as accuracy. Thus, these data indicate that applying TVA to 5CSRTT performance provides enhanced sensitivity to capturing attentional effects compared with standard 5CSRTT variables, both via increased attentional load and TVA modelling. This application of the TVA model to rodents further enables future translational investigations of neural mechanisms underlying visual attentional processing. The potential cross-species translational value of applying TVA modelling to the rodent 5CSRTT is exemplified by recent studies of healthy humans performing TVA-CPT with acute MPH, AMPH or ATO treatment, where AMPH was the only drug specifically improving TVA-modelled visual processing speed, and neither MPH or ATO improved visual processing speed (Lansner 2022). Nevertheless, we should also acknowledge limitations of the present findings that should be remediated by future studies: more extensive dose–response determinations are required to confirm whether the present effects observed at single doses hold over a wider range, and it will be necessary to compare the present acute actions with effects of chronic dosing, as occurs clinically in the treatment of ADHD.
References
Achterberg EJM, Van Kerkhof LWM, Servadio M et al (2016) Contrasting roles of dopamine and noradrenaline in the motivational properties of social play behavior in rats. Neuropsychopharmacology 41:858–868. https://doi.org/10.1038/npp.2015.212
Arnsten A, Li B (2005) Neurobiology of executive functions: catecholamine influences on prefrontal cortical functions. Biol Psychiatry 57:1377–1384. https://doi.org/10.1016/j.bps.2004.08.019
Arnsten AFT (2006) Fundamentals of attention-deficit/hyperactivity disorder: Circuits and Pathways. J Clin Psychiatry 67:7–12
Arnsten AFT, Dudley AG (2005) Methylphenidate improves prefrontal cortical cognitive function through α2 adrenoceptor and dopamine D1 receptor actions: relevance to therapeutic effects in Attention Deficit Hyperactivity Disorder. Behav Brain Funct 1:1–9. https://doi.org/10.1186/1744-9081-1-2
Baarendse PJJ, Vanderschuren LJMJ (2012) Dissociable effects of monoamine reuptake inhibitors on distinct forms of impulsive behavior in rats. Psychopharmacology 219:313–326. https://doi.org/10.1007/s00213-011-2576-x
Balachandran RC, Sieg ML, Tran CTQ et al (2018) Cholinergic and dopaminergic interactions alter attention and response inhibition in Long-Evans rats performing the 5-choice serial reaction time task. Pharmacol Biochem Behav 175:160–173. https://doi.org/10.1016/j.pbb.2018.10.006
Bari A, Dalley JW, Robbins TW (2008) The application of the 5-choice serial reaction time task for the assessment of visual attentional processes and impulse control in rats. Nat Protoc 3:759–767. https://doi.org/10.1038/nprot.2008.41
Bari A, Eagle DM, Mar AC et al (2009) Dissociable effects of noradrenaline, dopamine, and serotonin uptake blockade on stop task performance in rats. Psychopharmacology 205:273–283. https://doi.org/10.1007/s00213-009-1537-0
Bari A, Mar AC, Theobald DEH, Elands SA, Oganya KC, Eagle DM, Robbins TW (2011) Prefrontal and monoaminergic contributions to stop-signal performance in rats. J Neurosci 31:9254–9263. https://doi.org/10.1523/JNEUROSCI.1543-11.2011
Bari A, Robbins TW (2013) Noradrenergic versus dopaminergic modulation of impulsivity, attention and monitoring behaviour in rats performing the stop-signal task: possible relevance to ADHD. Psychopharmacology 230:89–111. https://doi.org/10.1007/s00213-013-3141-6
Bédard ACV, Stein MA, Halperin JM et al (2015) Differential impact of methylphenidate and atomoxetine on sustained attention in youth with attention-deficit/hyperactivity disorder. J Child Psychol Psychiatry Allied Discip 56:40–48. https://doi.org/10.1111/jcpp.12272
Benn A, Robinson ESJ (2017) Differential roles for cortical versus sub-cortical noradrenaline and modulation of impulsivity in the rat. Psychopharmacology 234:255–266. https://doi.org/10.1007/s00213-016-4458-8
Berridge CW (2006) Neural substrates of psychostimulant-induced arousal. Neuropsychopharmacology 31:2332–2340. https://doi.org/10.1038/sj.npp.1301159
Berridge CW, Devilbiss DM, Andrzejewski ME et al (2006) Methylphenidate preferentially increases catecholamine neurotransmission within the prefrontal cortex at low doses that enhance cognitive function. Biol Psychiatry 60:1111–1120. https://doi.org/10.1016/j.biopsych.2006.04.022
Berridge CW, Shumsky JS, Andrzejewski ME et al (2012) Differential sensitivity to psychostimulants across prefrontal cognitive tasks: differential involvement of noradrenergic α 1- and α 2-receptors. Biol Psychiatry 71:467–473. https://doi.org/10.1016/j.biopsych.2011.07.022
Bidwell LC, McClernon FJ, Kollins SH (2011) Cognitive enhancers for the treatment of ADHD. Pharmacol Biochem Behav 99:262–274. https://doi.org/10.1016/j.pbb.2011.05.002
Blondeau C, Dellu-Hagedorn F (2007) Dimensional analysis of ADHD subtypes in rats. Biol Psychiatry 61:1340–1350. https://doi.org/10.1016/j.biopsych.2006.06.030
Brown DC, Co MS, Wolff RC, Atzori M (2012) alpha-Adrenergic receptors in auditory cue detection : a 2 receptor blockade suppresses false alarm responding in the rat. Neuropharmacology 62:2178–2183. https://doi.org/10.1016/j.neuropharm.2011.12.024
Bundesen C (1998) A computational theory of visual attention. Philos Trans R Soc Lond B Biol Sci 353:1271–1281. https://doi.org/10.1098/rstb.1998.0282
Bundesen C (1990) A theory of visual attention. Psychol Rev 97:523–547. https://doi.org/10.1037/0033-295X.97.4.523
Bundesen C, Habekost T, Kyllingsbæk S (2005) A neural theory of visual attention: Bridging cognition and neurophysiology. Psychol Rev 112:291–328. https://doi.org/10.1037/0033-295X.112.2.291
Bundesen C, Harms L (1999) Single-letter recognition as a function of exposure duration. Psychol Res 62:275–279. https://doi.org/10.1007/s004260050057
Bundesen C, Vangkilde S, Petersen A (2015) Recent developments in a computational theory of visual attention (TVA). Vision Res 116:210–218. https://doi.org/10.1016/j.visres.2014.11.005
Bymaster FP, Katner JS, Nelson DL et al (2002) Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology 27:699–711. https://doi.org/10.1016/S0893-133X(02)00346-9
Caballero-Puntiverio M, Fitzpatrick CM, Woldbye DPD, Andreasen JT (2017) Effects of amphetamine and methylphenidate on attentional performance and impulsivity in the mouse 5-Choice Serial Reaction Time Task. J Psychopharmacol 31:272–283. https://doi.org/10.1177/0269881116684339
Caballero-Puntiverio M, Lerdrup LS, Arvastson L et al (2020) ADHD medication and the inverted U-shaped curve: a pharmacological study in female mice performing the rodent continuous performance test (rCPT). Prog Neuro-Psychopharmacol Biol Psychiatry 99:109823. https://doi.org/10.1016/j.pnpbp.2019.109823
Caballero-Puntiverio M, Lerdrup LS, Grupe M et al (2019) Effect of ADHD medication in male C57BL/6J mice performing the rodent continuous performance test. Psychopharmacology 236:1839–1851. https://doi.org/10.1007/s00213-019-5167-x
Callahan PM, Plagenhoef MR, Blake DT, Terry AV (2019) Atomoxetine improves memory and other components of executive function in young-adult rats and aged rhesus monkeys. Neuropharmacology 155:65–75. https://doi.org/10.1016/j.neuropharm.2019.05.016
Caprioli D, Jupp B, Hong YT et al (2015) Dissociable rate-dependent effects of oral methylphenidate on impulsivity and D2/3 receptor availability in the striatum. J Neurosci 35:3747–3755. https://doi.org/10.1523/JNEUROSCI.3890-14.2015
Carli M, Robbins TW, Evenden JL, Everitt BJ (1983) Effects of lesions to ascending noradrenergic neurones on performance of a 5-choice serial reaction task in rats; implications for theories of dorsal noradrenergic bundle function based on selective attention and arousal. Behav Brain Res 9:361–380. https://doi.org/10.1016/0166-4328(83)90138-9
Chamberlain SR, del Campo N, Dowson J et al (2007) Atomoxetine improved response inhibition in adults with attention deficit/hyperactivity disorder. Biol Psychiatry 62:977–984. https://doi.org/10.1016/j.biopsych.2007.03.003
Choi Y, Raymer BK (2019) Sleep modulating agents. Bioorganic Med Chem Lett 29:2025–2033. https://doi.org/10.1016/j.bmcl.2019.06.043
Chudasama Y, Nathwani F, Robbins TW (2005) D-Amphetamine remediates attentional performance in rats with dorsal prefrontal lesions. Behav Brain Res 158:97–107. https://doi.org/10.1016/j.bbr.2004.08.011
Cohen RA (2014) The neuropsychology of attention, 2nd edn. Springer, New York, NY. https://doi.org/10.1007/978-0-387-72639-7
Cole BJ, Robbins TW (1987) Amphetamine impairs the discriminative performance of rats with dorsal noradrenergic bundle lesions on a 5-choice serial reaction time task: New evidence for central dopaminergic-noradrenergic interactions. Psychopharmacology (Berl) 458–466
Cole BJ, Robbins TW (1992) Forebrain norepinephrine: role in controlled information processing in the rat. Neuropsychopharmacology 7(2):129–42
Coull JT, Jones MEP, Egan TD et al (2004) Attentional effects of noradrenaline vary with arousal level: selective activation of thalamic pulvinar in humans. Neuroimage 22:315–322. https://doi.org/10.1016/j.neuroimage.2003.12.022
Crockett MJ, Clark L, Hauser MD, Robbins TW (2010) Serotonin selectively influences moral judgment and behavior through effects on harm aversion. Proc Natl Acad Sci U S A 107:17433–17438. https://doi.org/10.1073/pnas.1009396107
Devauges V, Sara SJ (1990) Activation of the Noradrenergic System Facilitates an Attentional Shift in the Rat. Behav Brain Res 39:19–23
Ding YS, Naganawa M, Gallezot JD et al (2014) Clinical doses of atomoxetine significantly occupy both norepinephrine and serotonin transports: implications on treatment of depression and ADHD. Neuroimage 86:164–171. https://doi.org/10.1016/j.neuroimage.2013.08.001
Ding Z, Brown JW, Rueter LE, Mohler EG (2018) Profiling attention and cognition enhancing drugs in a rat touchscreen-based continuous performance test. Psychopharmacology 235:1093–1105. https://doi.org/10.1007/s00213-017-4827-y
Economidou D, Theobald DEH, Robbins TW et al (2012) Norepinephrine and dopamine modulate impulsivity on the five-choice serial reaction time task through opponent actions in the shell and core sub-regions of the nucleus accumbens. Neuropsychopharmacology 37:2057–2066. https://doi.org/10.1038/npp.2012.53
Fan LY, Chou TL, Gau SSF (2017) Neural correlates of atomoxetine improving inhibitory control and visual processing in drug-naïve adults with attention-deficit/hyperactivity disorder. Hum Brain Mapp 38:4850–4864. https://doi.org/10.1002/hbm.23683
Faraone SV, Buitelaar J (2010) Comparing the efficacy of stimulants for ADHD in children and adolescents using meta-analysis. Eur Child Adolesc Psychiatry 19:353–364. https://doi.org/10.1007/s00787-009-0054-3
Faraone SV, Glatt SJ (2010) A comparison of the efficacy of medications for adult attention-deficit/ hyperactivity disorder using meta-analysis of effect sizes. J Clin Psychiatry 71:754–763. https://doi.org/10.4088/JCP.08m04902pur
Faraone SV, Biederman J (2002) Efficacy of Adderall for attention-deficit/hyperactivity disorder: a meta-analysis. J Atten Disord 6:69–75
Fernando ABP, Economidou D, Theobald DE et al (2012) Modulation of high impulsivity and attentional performance in rats by selective direct and indirect dopaminergic and noradrenergic receptor agonists. Psychopharmacology 341–352. https://doi.org/10.1007/s00213-011-2408-z
Finke K, Dodds CM, Bublak P et al (2010) Effects of modafinil and methylphenidate on visual attention capacity: a tva-based study. Psychopharmacology 210:317–329. https://doi.org/10.1007/s00213-010-1823-x
Fitzpatrick CM, Caballero-Puntiverio M, Gether U et al (2017) Theory of visual attention (TVA) applied to mice in the 5-choice serial reaction time task. Psychopharmacology (Berl) 845–855. https://doi.org/10.1007/s00213-016-4520-6
Gallezot JD, Weinzimmer D, Nabulsi N et al (2011) Evaluation of [11C]MRB for assessment of occupancy of norepinephrine transporters: studies with atomoxetine in non-human primates. Neuroimage 56:268–279. https://doi.org/10.1016/j.neuroimage.2010.09.040
Gamo N, Wang M, Arnsten AFT (2010) Methylphenidate and atomoxetine enhance prefrontal function through α2-adrenergic and dopamine D1 receptors. JAAC 49:1011–1023. https://doi.org/10.1016/j.jaac.2010.06.015
Gau SSF, Shang CY (2010) Improvement of executive functions in boys with attention deficit hyperactivity disorder: an open-label follow-up study with once-daily atomoxetine. Int J Neuropsychopharmacol 13:243–256. https://doi.org/10.1017/S1461145709990836
Habekost T (2015) Clinical TVA-based studies: a general review. Front Psychol 6:1–18. https://doi.org/10.3389/fpsyg.2015.00290
Habekost T, Petersen A, Vangkilde S (2014) Testing attention: comparing the ANT with TVA-based assessment. Behav Res Methods 46:81–94. https://doi.org/10.3758/s13428-013-0341-2
Habekost T, Starrfelt R (2009) Visual attention capacity: a review of TVA-based patient studies: cognition and neurosciences. Scand J Psychol 50:23–32. https://doi.org/10.1111/j.1467-9450.2008.00681.x
Hah M, Chang K (2005) Adolescents with bipolar disorders 15:996–1004
Hahn B, Stolerman IP (2005) Modulation of nicotine-induced attentional enhancement in rats by adrenoceptor antagonists. Psychopharmacology 438–447. https://doi.org/10.1007/s00213-004-1969-5
Harrison AA, Everitt BJ, Robbins TW (1999) Central serotonin depletion impairs both the acquisition and performance of a symmetrically reinforced go/no-go conditional visual discrimination. Behav Brain Res 100:99–112. https://doi.org/10.1016/S0166-4328(98)00117-X
Hauser J, Reissmann A, Oliver TS (2017) Effects of methylphenidate on attention in Wistar rats treated with the neurotoxin N - (2-chloroethyl ) - N -ethyl-2-. J Neural Transm 124:643–654. https://doi.org/10.1007/s00702-017-1679-x
Hazell PL, Kohn MR, Dickson R et al (2011) Core ADHD symptom improvement with atomoxetine versus methylphenidate: a direct comparison meta-analysis. J Atten Disord 15:674–683. https://doi.org/10.1177/1087054710379737
Heal DJ, Cheetham SC, Smith SL (2009) The neuropharmacology of ADHD drugs in vivo: Insights on efficacy and safety. Neuropharmacology 57:608–618. https://doi.org/10.1016/j.neuropharm.2009.08.020
Higgins GA, Silenieks LB, MacMillan C et al (2020) Characterization of amphetamine, methylphenidate, nicotine, and atomoxetine on measures of attention, impulsive action, and motivation in the rat: implications for translational research. Front Pharmacol 11. https://doi.org/10.3389/fphar.2020.00427
Jentsch JD, Aarde SM, Seu E (2009) Effects of atomoxetine and methylphenidate on performance of a lateralized reaction time task in rats. Psychopharmacology 202:497–504. https://doi.org/10.1007/s00213-008-1181-0
Koffarnus MN, Katz JL (2011) Response requirement and increases in accuracy produced by stimulant drugs in a 5-choice serial reaction-time task in rats. Psychopharmacology 213:723–733. https://doi.org/10.1007/s00213-010-2027-0
Koskinen T, Haapalinna A, Sirviö J (2003) a -Adrenoceptor-mediated modulation of 5-HT 2 receptor agonist induced impulsive responding in a 5-choice serial. Pharmacol Toxicol 92:214–225
Kratochvil CJ, Vaughan BS, Stoner JA et al (2011) A double-blind, placebo-controlled study of atomoxetine in young children with ADHD. Pediatrics 127. https://doi.org/10.1542/peds.2010-0825
Kratz O, Studer P, Baack J et al (2012) Differential effects of methylphenidate and atomoxetine on attentional processes in children with ADHD: an event-related potential study using the Attention Network Test. Prog Neuro-Psychopharmacol Biol Psychiatry 37:81–89. https://doi.org/10.1016/j.pnpbp.2011.12.008
Lansner J (2022) The aim of your attention - investigating the influence of monoamine reuptake inhibition on visual attention – cognitive, pharmacological, and methodological perspectives [doctoral thesis]. Department of Psychology, University of Copenhagen
Lapiz MDS, Morilak DA (2006) Noradrenergic modulation of cognitive function in rat medial prefrontal cortex as measured by attentional set shifting capability. Neuroscience 137:1039–1049. https://doi.org/10.1016/j.neuroscience.2005.09.031
Lezak MD, Howieson DB, Bigler ED, Tranel D (2012) Neuropsychological assessment, 5th edn. Oxford University Press, New York
Liu Y, Huang T, Tung C, Lin C (2015) Effects of atomoxetine on attention and impulsivity in the five-choice serial reaction time task in rats with lesions of dorsal noradrenergic ascending bundle. Prog Neuropsychopharmacol Biol Psychiatry 56:81–90. https://doi.org/10.1016/j.pnpbp.2014.08.007
Loos M, Staal J, Schoffelmeer ANM et al (2010) Inhibitory control and response latency differences between C57BL / 6J and DBA / 2J mice in a Go / No-Go and 5-choice serial reaction time task and strain-specific responsivity to amphetamine. Behav Brain Res 214:216–224. https://doi.org/10.1016/j.bbr.2010.05.027
Losier BJ, McGrath PJ, Klein RM (1996) Error patterns on the continuous performance test in non-medicated and medicated samples of children with and without ADHD: A meta-analytic review. J Child Psychol Psychiatry Allied Discip 37:971–987. https://doi.org/10.1111/j.1469-7610.1996.tb01494.x
Ma CL, Arnsten AFT, Li BM (2005) Locomotor hyperactivity induced by blockade of prefrontal cortical α 2-adrenoceptors in monkeys. Biol Psychiatry 57:192–195. https://doi.org/10.1016/j.biopsych.2004.11.004
MacQueen DA, Minassian A, Kenton JA et al (2018) Amphetamine improves mouse and human attention in the 5-choice continuous performance test. Neuropharmacology 138:87–96. https://doi.org/10.1016/j.neuropharm.2018.05.034
Marraccini ME, Weyandt LL, Rossi JS, Gudmundsdottir BG (2016) Neurocognitive enhancement or impairment? A systematic meta-analysis of prescription stimulant effects on processing speed, decision-making, planning, and cognitive perseveration. Exp Clin Psychopharmacol 24:269–284. https://doi.org/10.1037/pha0000079
Marshall CA, Brodnik ZD, Mortensen OV et al (2019) Neuropharmacology selective activation of dopamine D3 receptors and norepinephrine transporter blockade enhances sustained attention. Neuropharmacology 148:178–188. https://doi.org/10.1016/j.neuropharm.2019.01.003
Mathes CM, Gregson JR, Spector AC (2013) The selective serotonin reuptake inhibitor paroxetine decreases breakpoint of rats engaging in a progressive ratio licking task for sucrose and quinine solutions. Chem Senses 38:211–220. https://doi.org/10.1093/chemse/bjs096
Mervaala E, Alhainen K, Helkala E-L et al (1993) Electrophysiological and neuropsychological effects of a central alpha2-antagonist atipamezole in healthy volunteers. Behav Brain Res 55:85–91
Milstein JA, Dalley JW, Robbins TW (2010) Methylphenidate-induced impulsivity : pharmacological antagonism by β -adrenoreceptor blockade. J Psychopharmacol 24:309–321
Milstein JA, Lehmann O, Tehobald DEH et al (2007) Selective depletion of cortical noradrenaline by anti- dopamine beta-hydroxylase – saporin impairs attentional function and enhances the effects of guanfacine in the rat. Psychopharmacology (Berl) 51–63. https://doi.org/10.1007/s00213-006-0594-x
Nagelkerke NJ (1991) A note on a general definition of the coefficient of determination. Biometrika 78(3):691–692
Navarra R, Graf R, Huang Y et al (2008) Effects of atomoxetine and methylphenidate on attention and impulsivity in the 5-choice serial reaction time test. Prog Neuropsychopharmacol Biol Psychiatry 32:34–41. https://doi.org/10.1016/j.pnpbp.2007.06.017
Navarra RL, Clark BD, Gargiulo AT, Waterhouse BD (2017) Methylphenidate enhances early-stage sensory processing and rodent performance of a visual signal detection task. Nat Publ Gr 42:1326–1337. https://doi.org/10.1038/npp.2016.267
Ni HC, Hwang Gu SL, Lin HY et al (2016) Atomoxetine could improve intra-individual variability in drug-naïve adults with attention-deficit/hyperactivity disorder comparably with methylphenidate: a head-to-head randomized clinical trial. J Psychopharmacol 30:459–467. https://doi.org/10.1177/0269881116632377
Ni HC, Shang CY, Gau SSF et al (2013) A head-To-head randomized clinical trial of methylphenidate and atomoxetine treatment for executive function in adults with attention-deficit hyperactivity disorder. Int J Neuropsychopharmacol 16:1959–1973. https://doi.org/10.1017/S1461145713000357
Niittykoski M, Lappalainen R, Jolkkonen J et al (1998) Systemic administration of atipamezole, a selective antagonist of alpha- 2 adrenoceptors, facilitates behavioural activity but does not influence short-term or long-term memory in trimethyltin-intoxicated and control rats. Neurosci Biobehav Rev 22:735–750. https://doi.org/10.1016/S0149-7634(98)00002-5
Nilsson SRO, Heath CJ, Takillah S et al (2018) Continuous performance test impairment in a 22q11.2 microdeletion mouse model: improvement by amphetamine. Transl Psychiatry 8:247. https://doi.org/10.1038/s41398-018-0295-3
Paterson NE, Ricciardi J, Wetzler C, Hanania T (2011) Sub-optimal performance in the 5-choice serial reaction time task in rats was sensitive to methylphenidate, atomoxetine and d -amphetamine, but unaffected by the COMT inhibitor tolcapone. Neurosci Res 69:41–50. https://doi.org/10.1016/j.neures.2010.10.001
Paterson NE, Wetzler C, Hackett A, Hanania T (2012) Impulsive action and impulsive choice are mediated by distinct neuropharmacological substrates in rat. Int J Neuropsychopharmacol 15:1473–1487. https://doi.org/10.1017/S1461145711001635
Pattij T, Janssen MCW, Vanderschuren LJMJ et al (2007) Involvement of dopamine D1 and D2 receptors in the nucleus accumbens core and shell in inhibitory response control. Psychopharmacology 191:587–598. https://doi.org/10.1007/s00213-006-0533-x
Pattij T, Schetters D, Schoffelmeer ANM, Van Gaalen MM (2012) On the improvement of inhibitory response control and visuospatial attention by indirect and direct adrenoceptor agonists. Psychopharmacology 219:327–340. https://doi.org/10.1007/s00213-011-2405-2
Pillidge K, Porter AJ, Vasili T et al (2014) Atomoxetine reduces hyperactive / impulsive behaviours in neurokinin-1 receptor ‘ knockout ’ mice. Pharmacol Biochem Behav 127:56–61. https://doi.org/10.1016/j.pbb.2014.10.008
Puumala T, Riekkinen P, Sirvio J (1997) Modulation of vigilance and behavioral activation by alpha-1 adrenoceptors in the rat. Pharmacol Biochem Behav 56:705–712
Puumala T, Sirviö J (1997) Stimulation and blockade of alpha1 adrenoceptors affect behavioural activity, but not spatial working memory assessed by delayed non-matching to position task in rats. J Psychopharmacol 11:45–51
Rapoport JL, Buchsbaum MS, Weingartner H et al (1980) Dextroamphetamine: its cognitive and behavioral effects in normal and hyperactive boys and normal men. Arch Gen Psychiatry 37:933–943. https://doi.org/10.1001/archpsyc.1980.01780210091010
Rinaman L (2011) Hindbrain noradrenergic A2 neurons: diverse roles in autonomic, endocrine, cognitive, and behavioral functions. Am J Physiol - Regul Integr Comp Physiol 300:222–235. https://doi.org/10.1152/ajpregu.00556.2010
Robbins TW (2002) The 5-choice serial reaction time task: behavioural pharmacology and functional neurochemistry. Psychopharmacology 163:362–380. https://doi.org/10.1007/s00213-002-1154-7
Robinson ESJ (2012) Blockade of noradrenaline re-uptake sites improves accuracy and impulse control in rats performing a five-choice serial reaction time tasks. Psychopharmacology 219:303–312. https://doi.org/10.1007/s00213-011-2420-3
Robinson ESJ, Eagle DM, Mar AC et al (2008) Similar effects of the selective noradrenaline reuptake inhibitor atomoxetine on three distinct forms of impulsivity in the rat. Neuropsychopharmacology 33:1028–1037. https://doi.org/10.1038/sj.npp.1301487
Roman CW, Derkach VA, Palmiter RD (2016) Genetically and functionally defined NTS to PBN brain circuits mediating anorexia. Nat Commun 7:11905. https://doi.org/10.1038/ncomms11905
Rosenberg MB, Carroll FI, Negus SS (2013) Effects of monoamine reuptake inhibitors in assays of acute pain-stimulated and pain-depressed behavior in rats. J Pain 14:246–259. https://doi.org/10.1016/j.jpain.2012.11.006
Ruotsalainen S, Haapalinna A, Riekkinen PJ, Sirviö J (1997) Dexmedetomidine reduces response tendency, but not accuracy of rats in attention and short-term memory tasks. Pharmacol Biochem Behav 56:31–40
Shalev N, Vangkilde S, Neville MJ et al (2019) Dissociable catecholaminergic modulation of visual attention: differential effects of catechol-O-methyltransferase and dopamine beta-hydroxylase genes on visual attention. Neuroscience 412:175–189. https://doi.org/10.1016/j.neuroscience.2019.05.068
Shang CY, Gau SSF (2012) Improving visual memory, attention, and school function with atomoxetine in boys with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol 22:353–363. https://doi.org/10.1089/cap.2011.0149
Sirvio J, Jäkälä P, Mazurkiewicz M et al (1993) Dose- and parameter-dependent effects of atipamezole, an a2-antagonist, on the performance of rats in a five-choice serial reaction time task. Pharmacol Biochem Behav 45:123–129
Sirviö J, MacDonald E (1999) Central α1-adrenoceptors: their role in the modulation of attention and memory formation. Pharmacol Ther 83:49–65. https://doi.org/10.1016/S0163-7258(99)00017-0
Sirviö J, Mazurkiewicz M, Haapalinna A et al (1994) The effects of selective alpha-2 adrenergic agents on the performance of rats in a 5-choice serial reaction time task. Brain Res Bull 35:451–455
Smith A, Nutt D (1996) Noradrenaline and attention lapses. Nature 380:1996
Spencer RC, Klein RM, Berridge CW (2012) Psychostimulants act within the prefrontal cortex to improve cognitive function. Biol Psychiatry 72:221–227. https://doi.org/10.1016/j.biopsych.2011.12.002
Sun H, Cocker PJ, Zeeb FD, Winstanley CA (2012) Chronic atomoxetine treatment during adolescence decreases impulsive choice, but not impulsive action, in adult rats and alters markers of synaptic plasticity in the orbitofrontal cortex. Psychopharmacology 219:285–301. https://doi.org/10.1007/s00213-011-2419-9
Sun H, Green TA, Theobald DEH et al (2010) Yohimbine increases impulsivity through activation of cAMP response element binding in the Orbitofrontal Cortex. Biol Psychiatry 67:649–656. https://doi.org/10.1016/j.biopsych.2009.11.030
Swann AC, Birnbaum D, Jagar AA et al (2005) Acute yohimbine increases laboratory-measured impulsivity in normal subjects. Biol Psychiatry 57:1209–1211. https://doi.org/10.1016/j.biopsych.2005.02.007
Swann AC, Lijffijt M, Lane SD et al (2013) Norepinephrine and impulsivity: effects of acute yohimbine. Psychopharmacology 83–94. https://doi.org/10.1007/s00213-013-3088-7
Tomlinson A, Grayson B, Marsh S et al (2014) Pay attention to impulsivity : modelling low attentive and high impulsive subtypes of adult ADHD in the 5-choice continuous performance task (5C-CPT ) in female rats. Eur Neuropsychopharmacol 24:1371–1380. https://doi.org/10.1016/j.euroneuro.2014.04.008
Toschi C, Hervig ME, Moazen P et al (2021) Adaptive aspects of impulsivity and interactions with effects of catecholaminergic agents in the 5-choice serial reaction time task : implications for ADHD. Psychopharmacology 238:2601–2615. https://doi.org/10.1007/s00213-021-05883-y
Tsutsui-Kimura I, Ohmura Y, Izumi T et al (2009) The effects of serotonin and/or noradrenaline reuptake inhibitors on impulsive-like action assessed by the three-choice serial reaction time task: a simple and valid model of impulsive action using rats. Behav Pharmacol 20:474–483. https://doi.org/10.1097/FBP.0b013e3283305e65
Turner KM, Burne THJ (2016) Improvement of attention with amphetamine in low- and high-performing rats. Psychopharmacology 233:3383–3394. https://doi.org/10.1007/s00213-016-4376-9
Tzavara ET, Bymaster FP, Overshiner CD et al (2006) Procholinergic and memory enhancing properties of the selective norepinephrine uptake inhibitor atomoxetine. Mol Psychiatry 11(2):187–195. https://doi.org/10.1038/sj.mp.4001763
Van Gaalen MM, Brueggeman RJ, Bronius PFC et al (2006) Behavioral disinhibition requires dopamine receptor activation. Psychopharmacology 187:73–85. https://doi.org/10.1007/s00213-006-0396-1
Walker DJ, Mason O, Clemow DB, Day KA (2015) Atomoxetine treatment in adults with attention-deficit/hyperactivity disorder. Postgrad Med 127:686–701. https://doi.org/10.1080/00325481.2015.1081046
Wehmeier PM, Schacht A, Ulberstad F et al (2012) Does atomoxetine improve executive function, inhibitory control, and hyperactivity? Results from a placebo-controlled trial using quantitative measurement technology. J Clin Psychopharmacol 32:653–660. https://doi.org/10.1097/JCP.0b013e318267c304
Wehmeier PM, Schacht A, Wolff C et al (2011) Neuropsychological outcomes across the day in children with attention-deficit/hyperactivity disorder treated with atomoxetine: results from a placebo-controlled study using a computer-based continuous performance test combined with an infra-red motion-tra. J Child Adolesc Psychopharmacol 21:433–444. https://doi.org/10.1089/cap.2010.0142
Wilens TE, Newcorn JH, Kratochvil CJ et al (2006) Long-term atomoxetine treatment in adolescents with attention-deficit/hyperactivity disorder. J Pediatr 149:112–119. https://doi.org/10.1016/j.jpeds.2006.01.052
Yohn SE, Errante EE, Rosenbloom-Snow A et al (2016a) Blockade of uptake for dopamine, but not norepinephrine or 5-HT, increases selection of high effort instrumental activity: Implications for treatment of effort-related motivational symptoms in psychopathology. Neuropharmacology 109:270–280. https://doi.org/10.1016/j.neuropharm.2016.06.018
Yohn SE, Lopez-Cruz L, Hutson PH et al (2016b) Effects of lisdexamfetamine and s-citalopram, alone and in combination, on effort-related choice behavior in the rat. Psychopharmacology 233:949–960. https://doi.org/10.1007/s00213-015-4176-7
Young JW, Roberts BZ, Breier M, Swerdlow NR (2020) Amphetamine improves rat 5-choice continuous performance test (5C-CPT ) irrespective of concurrent low-dose haloperidol treatment. Psychopharmacology 237:1959–1972. https://doi.org/10.1007/s00213-020-05511-1
Funding
This work was funded by the Lundbeck Foundation (held by UG; R223-2016–261). CT is the recipient of a Medical Research Council doctoral training award at Cambridge University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T. W. R. discloses consultancy with Cambridge Cognition, Arcadia, Takeda, Greenfield Bioventures, Cassava Inc.; he holds research grants with Shionogi and GlaxoSmithKline. M. E. H., C. T., A. P., S. V. and U. G. declare no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hervig, M.ES., Toschi, C., Petersen, A. et al. Theory of visual attention (TVA) applied to rats performing the 5-choice serial reaction time task: differential effects of dopaminergic and noradrenergic manipulations. Psychopharmacology 240, 41–58 (2023). https://doi.org/10.1007/s00213-022-06269-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-022-06269-4