Abstract
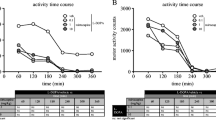
Monoamine oxidase (MAO) type B (MAO-B) inhibition was shown to confer anti-parkinsonian benefit as monotherapy and adjunct to l-3,4-dihydroxyphenylalanine (l-DOPA) in clinical trials. Here, we explore the anti-parkinsonian effect of MAO type A (MAO-A) inhibition as monotherapy, as the enzyme MAO-A is also encountered within the primate and human basal ganglia, where it metabolises dopamine, albeit to a lesser extent than MAO-B. In six 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-lesioned marmosets, we assessed the anti-parkinsonian effect of the reversible MAO-A inhibitor moclobemide (0.1 and 1 mg/kg) as monotherapy and compared it to that of l-DOPA and vehicle treatments. Moclobemide significantly reversed parkinsonism (by 39%, P < 0.01), while eliciting only mild dyskinesia and psychosis-like behaviours (PLBs). In contrast, l-DOPA anti-parkinsonian effect was accompanied by marked dyskinesia and PLBs. MAO-A inhibition with moclobemide may provide anti-parkinsonian benefit when administered without l-DOPA and might perhaps be considered as monotherapy for the treatment of Parkinson’s disease in the early stages of the condition.

Similar content being viewed by others
References
Apaydin S, Goldeli E, Uyar M, Erhan E, Yegul I, Tuglular I (2001) The antinociceptive effect of moclobemide on the vocalization threshold to paw pressure in a rat model of unilateral mononeuropathy. Pharmacol Res 44(6):503–507. https://doi.org/10.1006/phrs.2001.0895
Bonnet U (2002) Moclobemide: evolution, pharmacodynamic, and pharmacokinetic properties. CNS Drug Rev 8(3):283–308. https://doi.org/10.1111/j.1527-3458.2002.tb00229.x
Connolly BS, Lang AE (2014) Pharmacological treatment of Parkinson disease: a review. JAMA 311(16):1670–1683. https://doi.org/10.1001/jama.2014.3654
Dietrichs E, Odin P (2017) Algorithms for the treatment of motor problems in Parkinson’s disease. Acta Neurol Scand 136(5):378–385. https://doi.org/10.1111/ane.12733
Fox SH, Visanji N, Reyes G, Huot P, Gomez-Ramirez J, Johnston T, Brotchie JM (2010) Neuropsychiatric behaviors in the MPTP marmoset model of Parkinson’s disease. Can J Neurol Sci 37(1):86–95
Frouni I, Hamadjida A, Kwan C, Bedard D, Nafade V, Gaudette F, Nuara SG, Gourdon JC, Beaudry F, Huot P (2019) Activation of mGlu2/3 receptors, a novel therapeutic approach to alleviate dyskinesia and psychosis in experimental parkinsonism. Neuropharmacology 158:107725. https://doi.org/10.1016/j.neuropharm.2019.107725
Gaweska H, Fitzpatrick PF (2011) Structures and mechanism of the monoamine oxidase family. Biomol Concepts 2(5):365–377. https://doi.org/10.1515/BMC.2011.030
Gimenez-Roldan S, Dobato JL, Mateo D (1997) Treatment of depression in Parkinson’s disease with moclobemide: a pilot open-label study. Parkinsonism Relat Disord 3(4):219–225. https://doi.org/10.1016/s1353-8020(97)00028-x
Haefely W, Burkard WP, Cesura AM, Kettler R, Lorez HP, Martin JR, Richards JG, Scherschlicht R, Da Prada M (1992) Biochemistry and pharmacology of moclobemide, a prototype RIMA. Psychopharmacology (Berl) 106(Suppl):S6–S14. https://doi.org/10.1007/bf02246225
Hamadjida A, Nuara SG, Veyres N, Frouni I, Kwan C, Sid-Otmane L, Harraka MJ, Gourdon JC, Huot P (2017) The effect of mirtazapine on dopaminergic psychosis and dyskinesia in the parkinsonian marmoset. Psychopharmacology 234(6):905–911. https://doi.org/10.1007/s00213-017-4530-z
Hamadjida A, Nuara SG, Bedard D, Frouni I, Kwan C, Gourdon JC, Huot P (2018a) Nefazodone reduces dyskinesia, but not psychosis-like behaviours, in the parkinsonian marmoset. Naunyn Schmiedeberg’s Arch Pharmacol 391(12):1339–1345. https://doi.org/10.1007/s00210-018-1549-6
Hamadjida A, Nuara SG, Bedard D, Gaudette F, Beaudry F, Gourdon JC, Huot P (2018b) The highly selective 5-HT2A antagonist EMD-281,014 reduces dyskinesia and psychosis in the l-DOPA-treated parkinsonian marmoset. Neuropharmacology 139:61–67. https://doi.org/10.1016/j.neuropharm.2018.06.038
Hamadjida A, Nuara SG, Gourdon JC, Huot P (2018c) The effect of mianserin on the severity of psychosis and dyskinesia in the parkinsonian marmoset. Prog Neuro-Psychopharmacol Biol Psychiatry 81:367–371. https://doi.org/10.1016/j.pnpbp.2017.09.001
Hamadjida A, Nuara SG, Gourdon JC, Huot P (2018d) Trazodone alleviates both dyskinesia and psychosis in the parkinsonian marmoset model of Parkinson’s disease. J Neural Transm (Vienna) 125(9):1355–1360. https://doi.org/10.1007/s00702-017-1830-8
Holford NH, Guentert TW, Dingemanse J, Banken L (1994) Monoamine oxidase-A: pharmacodynamics in humans of moclobemide, a reversible and selective inhibitor. Br J Clin Pharmacol 37(5):433–439. https://doi.org/10.1111/j.1365-2125.1994.tb05710.x
Huot P, Johnston TH, Lewis KD, Koprich JB, Reyes MG, Fox SH, Piggott MJ, Brotchie JM (2011) Characterization of 3,4-methylenedioxymethamphetamine (MDMA) enantiomers in vitro and in the MPTP-lesioned primate: R-MDMA reduces severity of dyskinesia, whereas S-MDMA extends duration of ON-time. J Neurosci 31(19):7190–7198. https://doi.org/10.1523/JNEUROSCI.1171-11.2011
Huot P, Johnston TH, Koprich JB, Fox SH, Brotchie JM (2012) L-DOPA pharmacokinetics in the MPTP-lesioned macaque model of Parkinson’s disease. Neuropharmacology 63(5):829–836. https://doi.org/10.1016/j.neuropharm.2012.06.012
Iravani MM, Jackson MJ, Kuoppamaki M, Smith LA, Jenner P (2003) 3,4-methylenedioxymethamphetamine (ecstasy) inhibits dyskinesia expression and normalizes motor activity in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated primates. J Neurosci 23(27):9107–9115
Jansen Steur EN, Ballering LA (1999) Combined and selective monoamine oxidase inhibition in the treatment of depression in Parkinson’s disease. Adv Neurol 80:505–508
Kulisevsky J, Oliveira L, Fox SH (2018) Update in therapeutic strategies for Parkinson’s disease. Curr Opin Neurol 31(4):439–447. https://doi.org/10.1097/WCO.0000000000000579
Kwan C, Frouni I, Bedard D, Nuara SG, Gourdon JC, Hamadjida A, Huot P (2019) 5-HT2A blockade for dyskinesia and psychosis in Parkinson’s disease: is there a limit to the efficacy of this approach? A study in the MPTP-lesioned marmoset and a literature mini-review. Exp Brain Res 237(2):435–442. https://doi.org/10.1007/s00221-018-5434-9
Lakshmana MK, Rao BS, Dhingra NK, Ravikumar R, Govindaiah SS, Meti BL, Raju TR (1998) Role of monoamine oxidase type A and B on the dopamine metabolism in discrete regions of the primate brain. Neurochem Res 23(8):1031–1037. https://doi.org/10.1023/a:1020799700885
May T, Pawlik M, Rommelspacher H (1991) [3H]harman binding experiments. II: regional and subcellular distribution of specific [3H]harman binding and monoamine oxidase subtypes A and B activity in marmoset and rat. J Neurochem 56(2):500–508. https://doi.org/10.1111/j.1471-4159.1991.tb08178.x
Nair NP, Ahmed SK, Kin NM (1993) Biochemistry and pharmacology of reversible inhibitors of MAO-A agents: focus on moclobemide. J Psychiatry Neurosci 18(5):214–225
O'Carroll AM, Fowler CJ, Phillips JP, Tobbia I, Tipton KF (1983) The deamination of dopamine by human brain monoamine oxidase. Specificity for the two enzyme forms in seven brain regions. Naunyn Schmiedeberg’s Arch Pharmacol 322(3):198–202. https://doi.org/10.1007/bf00500765
PD Med Collaborative Group, Gray R, Ives N, Rick C, Patel S, Gray A, Jenkinson C, McIntosh E, Wheatley K, Williams A, Clarke CE (2014) Long-term effectiveness of dopamine agonists and monoamine oxidase B inhibitors compared with levodopa as initial treatment for Parkinson’s disease (PD MED): a large, open-label, pragmatic randomised trial. Lancet 384(9949):1196–1205. https://doi.org/10.1016/S0140-6736(14)60683-8
Seppi K, Ray Chaudhuri K, Coelho M, Fox SH, Katzenschlager R, Perez Lloret S, Weintraub D, Sampaio C, the collaborators of the Parkinson’s Disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine C (2019) Update on treatments for nonmotor symptoms of Parkinson’s disease-an evidence-based medicine review. Mov Disord 34(2):180–198. https://doi.org/10.1002/mds.27602
Sternic N, Kacar A, Filipovic S, Svetel M, Kostic VS (1998) The therapeutic effect of moclobemide, a reversible selective monoamine oxidase A inhibitor, in Parkinson’s disease. Clin Neuropharmacol 21(2):93–96
Steur EN, Ballering LA (1997) Moclobemide and selegeline in the treatment of depression in Parkinson’s disease. J Neurol Neurosurg Psychiatry 63(4):547. https://doi.org/10.1136/jnnp.63.4.547
Takats A, Tarczy M, Simo M, Szombathelyi E, Bodrogi A, Karpati R (1994) Moclobemide/aurorix/treatment in Parkinson’s disease with depression [Abstract]. 11th International Symposium on Parkinson’s Disease:260
Tong J, Meyer JH, Furukawa Y, Boileau I, Chang LJ, Wilson AA, Houle S, Kish SJ (2013) Distribution of monoamine oxidase proteins in human brain: implications for brain imaging studies. J Cereb Blood Flow Metab 33(6):863–871. https://doi.org/10.1038/jcbfm.2013.19
Youdim MB, Edmondson D, Tipton KF (2006) The therapeutic potential of monoamine oxidase inhibitors. Nat Rev Neurosci 7(4):295–309. https://doi.org/10.1038/nrn1883
Zhukovsky P, Alsio J, Jupp B, Xia J, Giuliano C, Jenner L, Griffiths J, Riley E, Ali S, Roberts AC, Robbins TW, Dalley JW (2017) Perseveration in a spatial-discrimination serial reversal learning task is differentially affected by MAO-A and MAO-B inhibition and associated with reduced anxiety and peripheral serotonin levels. Psychopharmacology 234(9–10):1557–1571. https://doi.org/10.1007/s00213-017-4569-x
Funding
PH has research support from Parkinson Canada, Fonds de Recherche Québec – Santé, the Weston Brain Institute, the Michael J Fox Foundation for Parkinson’s Research, the Natural Sciences and Engineering Research Council of Canada and Healthy Brains for Healthy Lives.
Author information
Authors and Affiliations
Contributions
AH, SN, JC and PH conceived and designed research. AH, SN, CK, IF, DB and JC conducted experiments. PH analysed data. PH wrote the manuscript. All authors read and approved the manuscript.
The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author institutional email address: philippe.huot@mcgill.ca
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Experiments were approved by McGill University and the Montreal Neurological Institute Animal Care Committees, which are in accordance with the regulations defined by the Canadian Council on Animal Care.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PZFX 137 kb)
Rights and permissions
About this article
Cite this article
Hamadjida, A., Nuara, S.G., Frouni, I. et al. Monoamine oxidase A inhibition as monotherapy reverses parkinsonism in the MPTP-lesioned marmoset. Naunyn-Schmiedeberg's Arch Pharmacol 393, 2139–2144 (2020). https://doi.org/10.1007/s00210-020-01927-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01927-w