Abstract
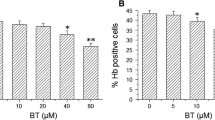
The phenolic metabolite of benzene, hydroquinone (HQ), has potential risks for hematological disorders and hematotoxicity in humans. Previous studies have revealed that reactive oxygen species, DNA methylation, and histone acetylation participate in benzene metabolites inhibiting erythroid differentiation in hemin-induced K562 cells. GATA1 and GATA2 are crucial erythroid-specific transcription factors that exhibit dynamic expression patterns during erythroid differentiation. We investigated the role of GATA factors in HQ-inhibited erythroid differentiation in K562 cells. When K562 cells were induced with 40 μM hemin for 0–120 h, the mRNA and protein levels of GATA1 and GATA2 changed dynamically. After exposure to 40 μM HQ for 72 h, K562 cells were induced with 40 μM hemin for 48 h. HQ considerably reduced the percentage of hemin-induced Hb-positive cells, decreased the GATA1 mRNA, protein, and occupancy levels at α-globin and β-globin gene clusters, and increased the GATA2 mRNA and protein levels significantly. ChIP-seq analysis revealed that HQ reduced GATA1 occupancy, and increased GATA2 occupancy at most gene loci in hemin-induced K562 cells. And GATA1 and GATA2 might play essential roles in the erythroid differentiation protein interaction network. These results elucidate that HQ decreases GATA1 occupancy and increases GATA2 occupancy at the erythroid gene loci, thereby downregulating GATA1 and upregulating GATA2 expression, which in turn modulates the expression of erythroid genes and inhibits erythroid differentiation. This partially explains the mechanism of benzene hematotoxicity.






Similar content being viewed by others
Data availability
The data are available from the corresponding author upon reasonable request.
References
Anguita E, Hughes J, Heyworth C et al (2004) Globin gene activation during haemopoiesis is driven by protein complexes nucleated by GATA-1 and GATA-2. EMBO J 23:2841–2852. https://doi.org/10.1038/sj.emboj.7600274
Bresnick EH, Katsumura KR, Lee HY et al (2012) Master regulatory GATA transcription factors: Mechanistic principles and emerging links to hematologic malignancies. Nucleic Acids Res 40:5819–5831. https://doi.org/10.1093/nar/gks281
Cheng Y, Wu W, Kumar SA et al (2009) Erythroid GATA1 function revealed by genome-wide analysis of transcription factor occupancy, histone modifications, and mRNA expression. Genome Res 19:2172–2184. https://doi.org/10.1101/gr.098921.109
Cho Y, Song SH, Lee JJ et al (2008) The role of transcriptional activator GATA-1 at human β-globin HS2. Nucleic Acids Res 36:4521–4528. https://doi.org/10.1093/nar/gkn368
Crispino JD, Horwitz MS (2017) GATA factor mutations in hematologic disease. Blood 129:2103–2110. https://doi.org/10.1182/blood-2016-09-687889
Doré LC, Chlon TM, Brown CD et al (2012) Chromatin occupancy analysis reveals genome-wide GATA factor switching during hematopoiesis. Blood 119:3724–3733. https://doi.org/10.1182/blood-2011-09-380634
Doty RT, Yan X, Lausted C et al (2019) Single-cell analyses demonstrate that a heme-GATA1 feedback loop regulates red cell differentiation. Blood 133:457–469. https://doi.org/10.1182/blood-2018-05-850412
Fu YK, Tan Y, Wu B et al (2021) Gata2-L359V impairs primitive and definitive hematopoiesis and blocks cell differentiation in murine chronic myelogenous leukemia model. Cell Death Dis 12:1–17. https://doi.org/10.1038/s41419-021-03826-1
Fujiwara T, O’Geen H, Keles S et al (2009) Discovering hematopoietic mechanisms through genome-wide analysis of GATA factor chromatin occupancy. Mol Cell 36:667–681. https://doi.org/10.1016/j.molcel.2009.11.001
Grass JA, Boyer ME, Pal S et al (2003) GATA-1-dependent transcriptional repression of GATA-2 via disruption of positive autoregulation and domain-wide chromatin remodeling. Proc Natl Acad Sci USA 100:8811–8816. https://doi.org/10.1073/pnas.1432147100
Guo Y, Fu X, Jin Y et al (2015) Histone demethylase LSD1-mediated repression of GATA-2 is critical for erythroid differentiation. Drug Des Devel Ther 9:3153–3162. https://doi.org/10.2147/DDDT.S81911
Gutiérrez L, Caballero N, Fernández-Calleja L et al (2020) Regulation of GATA1 levels in erythropoiesis. IUBMB Life 72:89–105. https://doi.org/10.1002/iub.2192
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans (2018) Benzene. International Agency for Research on Cancer, Lyon (PMID: 31769947, Bookshelf ID: NBK550157)
Ikonomi P, Noguchi CT, Miller W et al (2000) Levels of GATA-1/GATA-2 transcription factors modulate expression of embryonic and fetal hemoglobins. Gene 261:277–287. https://doi.org/10.1016/S0378-1119(00)00510-2
Im H, Grass JA, Johnson KD et al (2005) Chromatin domain activation via GATA-1 utilization of a small subset of dispersed GATA motifs within a broad chromosomal region. Proc Natl Acad Sci USA 102:17065–17070. https://doi.org/10.1073/pnas.0506164102
Kadauke S, Udugama MI, Pawlicki JM et al (2012) Tissue-specific mitotic bookmarking by hematopoietic transcription factor GATA1. Cell 150:725–737. https://doi.org/10.1016/j.cell.2012.06.038
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30. https://doi.org/10.1093/nar/28.1.27
Kaneko H, Shimizu R, Yamamoto M (2010) GATA factor switching during erythroid differentiation. Curr Opin Hematol 17:163–168. https://doi.org/10.1097/MOH.0b013e32833800b8
Lan Q, Zhang L, Li G et al (2004) Hematotoxicity in workers exposed to low levels of benzene. Science 306:1774–1776. https://doi.org/10.1126/science.1102443
Li Y, Wu XR, Li XF et al (2013) Changes in DNA methylation of erythroid-specific genes in K562 cells exposed to phenol and hydroquinone. Toxicology 312:108–114. https://doi.org/10.1016/j.tox.2013.08.007
Liang B, Chen Y, Yuan W et al (2018) Down-regulation of miRNA-451a and miRNA-486-5p involved in benzene-induced inhibition on erythroid cell differentiation in vitro and in vivo. Arch Toxicol 92:259–272. https://doi.org/10.1007/s00204-017-2033-7
Liew CK, Simpson RJY, Kwan AHY et al (2005) Zinc fingers as protein recognition motifs: Structural basis for the GATA-1/friend of GATA interaction. Proc Natl Acad Sci USA 102:583–588. https://doi.org/10.1073/pnas.0407511102
Lozzio CB, Lozzio BB (1975) Human chronic myelogenous leukemia cell-line with positive Philadelphia chromosome. Blood 45:321–334
Martowicz ML, Grass JA, Boyer ME et al (2005) Dynamic GATA factor interplay at a multicomponent regulatory region of the GATA-2 locus. J Biol Chem 280:1724–1732. https://doi.org/10.1074/jbc.M406038200
McIver SC, Kang YA, Devilbiss AW et al (2014) The exosome complex establishes a barricade to erythroid maturation. Blood 124:2285–2297. https://doi.org/10.1182/blood-2014-04-571083
Moriguchi T, Yamamoto M (2014) A regulatory network governing Gata1 and Gata2 gene transcription orchestrates erythroid lineage differentiation. Int J Hematol 100:417–424. https://doi.org/10.1007/s12185-014-1568-0
Persons DA, Allay JA, Allay ER et al (1999) Enforced expression of the GATA-2 transcription factor blocks normal hematopoiesis. Blood 93:488–499. https://doi.org/10.1182/blood.v93.2.488
Pomerantz O, Goodwin AJ, Joyce T, Lowrey CH (1998) Conserved elements containing NF-E2 and tandem GATA binding sites are required for erythroid-specific chromatin structure reorganization within the human β-globin locus control region. Nucleic Acids Res 26:5684–5691. https://doi.org/10.1093/nar/26.24.5684
Romeo P, Prandini M, Joulin V et al (1990) Megakaryocytic and erythrocytic lineages share specific transcription factors. Nature 344:447–449. https://doi.org/10.1038/344447a0
Snyder R, Hedli CC (1996) An overview of benzene metabolism. Environ Health Perspect 104:1165–1171. https://doi.org/10.1289/ehp.96104s61165
Strouboulis J, Ronchi AE (2022) GATA1: function through disorder. Blood 139:2422–2423. https://doi.org/10.1182/blood.2021015351
Stumpf M, Waskow C, Krötschel M et al (2006) The mediator complex functions as a coactivator for GATA-1 in erythropoiesis via subunit Med1/TRAP220. Proc Natl Acad Sci USA 103:18504–18509. https://doi.org/10.1073/pnas.0604494103
Tang KY, Yu CH, Jiang L et al (2016) Long-term exposure of K562 cells to benzene metabolites inhibited erythroid differentiation and elevated methylation in erythroid specific genes. Toxicol Res (Cambr) 5:1284–1297. https://doi.org/10.1039/c6tx00143b
Thoms JAI, Truong P, Subramanian S et al (2021) Disruption of a GATA2-TAL1-ERG regulatory circuit promotes erythroid transition in healthy and leukemic stem cells. Blood 138:1441–1455. https://doi.org/10.1182/blood.2020009707
Wadman IA, Osada H, Grütz GG et al (1997) The LIM-only protein Lmo2 is a bridging molecule assembling an erythroid, DNA-binding complex which includes the TAL1, E47, GATA-1 and Ldb1/NLI proteins. EMBO J 16:3145–3157. https://doi.org/10.1093/emboj/16.11.3145
Wang Z, Wang P, Zhang J et al (2023) The novel GATA1-interacting protein HES6 is an essential transcriptional cofactor for human erythropoiesis. Nucleic Acids Res. https://doi.org/10.1093/nar/gkad167
Welch JJ, Watts JA, Vakoc CR et al (2004) Global regulation of erythroid gene expression by transcription factor GATA-1. Blood 104:3136–3147. https://doi.org/10.1182/blood-2004-04-1603
Whysner J, Reddy MV, Ross PM et al (2004) Genotoxicity of benzene and its metabolites. Mutat Res 566:99–130. https://doi.org/10.1016/S1383-5742(03)00053-X
Wu XR, Xue M, Li XF et al (2011) Phenolic metabolites of benzene inhibited the erythroid differentiation of K562 cells. Toxicol Lett 203:190–199. https://doi.org/10.1016/j.toxlet.2011.03.012
Wu W, Morrissey CS, Keller CA et al (2014) Dynamic shifts in occupancy by TAL1 are guided by GATA factors and drive large-scale reprogramming of gene expression during hematopoiesis. Genome Res 24:1945–1962. https://doi.org/10.1101/gr.164830.113
Yan B, Yang J, Kim MY et al (2021) HDAC1 is required for GATA-1 transcription activity, global chromatin occupancy and hematopoiesis. Nucleic Acids Res 49:9783–9798. https://doi.org/10.1093/nar/gkab737
Yang J, Bai WL, Chen YJ, Gao A (2015) 1,4-benzoquinone-induced STAT-3 hypomethylation in AHH-1 cells: Role of oxidative stress. Toxicol Rep 2:864–869. https://doi.org/10.1016/j.toxrep.2015.05.013
Young MD, Wakefield MJ, Smyth GK et al (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:R14. https://doi.org/10.1186/gb-2010-11-2-r14
Yu M, Riva L, Xie H et al (2009) Insights into GATA-1-mediated gene activation versus repression via genome-wide chromatin occupancy analysis. Mol Cell 36:682–695. https://doi.org/10.1016/j.molcel.2009.11.002
Yu CH, Suriguga YL, Li Y et al (2014) The role of ROS in hydroquinone-induced inhibition of K562 cell erythroid differentiation. Biomed Environ Sci 27:212–214. https://doi.org/10.3967/bes2014.043
Yu CH, Jiang L, Wang Y et al (2018) Inhibition of erythroid differentiation of human leukemia K562 cells by N-acetylcysteine and ascorbic acid through downregulation of ROS. Biomed Environ Sci 31:247–251. https://doi.org/10.3967/bes2018.032
Yu CH, Li Y, Zhao X et al (2019) Benzene metabolite 1,2,4-benzenetriol changes DNA methylation and histone acetylation of erythroid-specific genes in K562 cells. Arch Toxicol 93:137–147. https://doi.org/10.1007/s00204-018-2333-6
Yu CH, Yang SQ, Li L et al (2022) Identification of potential pathways and microRNA-mRNA networks associated with benzene metabolite hydroquinone-induced hematotoxicity in human leukemia K562 cells. BMC Pharmacol Toxicol 23:1–12. https://doi.org/10.1186/s40360-022-00556-8
Zheng WW, Dong XM, Yin RH et al (2014) EDAG positively regulates erythroid differentiation and modifies GATA1 acetylation through recruiting p300. Stem Cells 32:2278–2289. https://doi.org/10.1002/stem.1723
Acknowledgements
This work was supported by grants from National Natural Science Foundation of China (Project no. 81573192).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
The manuscript does not contain clinical studies or patient data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, CH., Yang, SQ., Zhang, YJ. et al. The role of GATA switch in benzene metabolite hydroquinone inhibiting erythroid differentiation in K562 cells. Arch Toxicol 97, 2169–2181 (2023). https://doi.org/10.1007/s00204-023-03541-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03541-0