Abstract
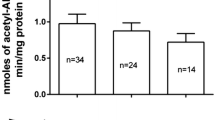
We used cryopreserved human hepatocytes that express rapid, intermediate, and slow acetylator N-acetyltransferase 2 (NAT2) genotypes to measure the N-acetylation of β-naphthylamine (BNA) which is one of the aromatic amines found in cigarette smoke including E-cigarettes. We investigated the role of NAT2 genetic polymorphism in genotoxicity and oxidative stress induced by BNA. In vitro BNA NAT2 activities in rapid acetylators was 1.6 and 3.5-fold higher than intermediate (p < 0.01) and slow acetylators (p < 0.0001). BNA N-acetylation in situ was 3 to 4- fold higher in rapid acetylators than slow acetylators, following incubation with 10 and 100 µM BNA (p < 0.01). DNA damage was two to threefold higher in the rapid versus slow acetylators (p < 0.0001) and 2.5-fold higher in intermediate versus slow acetylators following BNA treatment at 100 and 1000 μM, ROS/RNS level was the highest in rapid acetylators followed by intermediate and then slow acetylators (p < 0.0001). Our findings show that the N-acetylation of BNA is NAT2 genotype dependent in cryopreserved human hepatocytes and our data further document an important role for NAT2 genetic polymorphism in modifying BNA-induced genotoxicity and oxidative damage.





Similar content being viewed by others
References
Adams SP, Laws GM, Storer RD, DeLuca JG, Nichols WW (1996) Detection of DNA damage induced by human carcinogens in acellular assays: potential application for determining genotoxic mechanisms. Mutat Res/genet Toxicol 368(3):235–248. https://doi.org/10.1016/S0165-1218(96)90065-8
Allen CE, Doll MA, Hein DW (2017) N-Acetyltransferase 2 genotype-dependent N-acetylation of hydralazine in human hepatocytes. Drug Metab Dispos 45(12):1276–1281. https://doi.org/10.1124/dmd.117.078543
Baumgartner KB, Schlierf TJ, Yang D, Doll MA, Hein DW (2009) N-acetyltransferase 2 genotype modification of active cigarette smoking on breast cancer risk among hispanic and non-hispanic white women. Toxicol Sci 112(1):211–220. https://doi.org/10.1093/toxsci/kfp199
Bellamri M, Le Hegarat L, Turesky RJ, Langouet S (2017) Metabolism of the tobacco carcinogen 2-amino-9H-pyrido[2,3-b]indole (AalphaC) in primary human hepatocytes. Chem Res Toxicol 30(2):657–668. https://doi.org/10.1021/acs.chemrestox.6b00394
Bellamri M, Walmsley SJ, Brown C et al (2022) DNA damage and oxidative stress of tobacco smoke condensate in human bladder epithelial cells. Chem Res Toxicol. https://doi.org/10.1021/acs.chemrestox.2c00153
Besaratinia A, Tommasi S (2013) Genotoxicity of tobacco smoke-derived aromatic amines and bladder cancer: current state of knowledge and future research directions. FASEB J 27(6):2090–2100. https://doi.org/10.1096/fj.12-227074
Blackadar CB (2016) Historical review of the causes of cancer. World J Clin Oncol 7(1):54–86. https://doi.org/10.5306/wjco.v7.i1.54
Boffetta P (2008) Tobacco smoking and risk of bladder cancer. Scand J Urol Nephrol 42(sup218):45–54
Brambilla G, Martelli A (1990) Human hepatocytes in genotoxicity assays. Pharmacol Res 22(4):381–392. https://doi.org/10.1016/1043-6618(90)90746-Z
Chun YJ, Kim D (2016) Cancer activation and polymorphisms of human cytochrome P450 1B1. Toxicol Res 32(2):89–93. https://doi.org/10.5487/TR.2016.32.2.089
Chung KT, Chen SC, Claxton LD (2006) Review of the Salmonella typhimurium mutagenicity of benzidine, benzidine analogues, and benzidine-based dyes. Mutat Res 612(1):58–76. https://doi.org/10.1016/j.mrrev.2005.08.001
Corvi R, Madia F (2017) In vitro genotoxicity testing-Can the performance be enhanced? Food Chem Toxicol 106(Pt B):600–608. https://doi.org/10.1016/j.fct.2016.08.024
Czubacka E, Czerczak S (2020) 2-naphthylamine toxicity. Med Pr 71(2):205–220. https://doi.org/10.13075/mp.5893.00921
Doll MA, Hein DW (2001) Comprehensive human NAT2 genotype method using single nucleotide polymorphism-specific polymerase chain reaction primers and fluorogenic probes. Anal Biochem 288(1):106–108. https://doi.org/10.1006/abio.2000.4892
Doll MA, Salazar-Gonzalez RA, Bodduluri S, Hein DW (2017) Arylamine N-acetyltransferase 2 genotype-dependent N-acetylation of isoniazid in cryopreserved human hepatocytes. Acta Pharm Sin B 7(4):517–522. https://doi.org/10.1016/j.apsb.2017.05.003
Doll MA, Zang Y, Moeller T, Hein DW (2010) Codominant expression of N-acetylation and O-acetylation activities catalyzed by N-acetyltransferase 2 in human hepatocytes. J Pharmacol Exp Ther 334(2):540–544. https://doi.org/10.1124/jpet.110.168567
Fontana L, Delort L, Joumard L et al (2009) Genetic polymorphisms in CYP1A1, CYP1B1, COMT, GSTP1 and NAT2 genes and association with bladder cancer risk in a French cohort. Anticancer Res 29(5):1631–1635
Fretland AJ, Leff MA, Doll MA, Hein DW (2001) Functional characterization of human N-acetyltransferase 2 (NAT2) single nucleotide polymorphisms. Pharmacogenet Genomics 11(3):207–215
Fuller TW, Acharya AP, Meyyappan T et al (2018) Comparison of bladder carcinogens in the urine of E-cigarette users versus non E-cigarette using controls. Sci Rep 8(1):507. https://doi.org/10.1038/s41598-017-19030-1
Grant DM (2021) Carcinogenesis. In: Harrington LA, Tannock IF, Hill RP, Cescon DW (eds) The basic science of oncology, vol 6e. McGraw-Hill Education, New York
Groff K, Evans SJ, Doak SH et al (2021) In vitro and integrated in vivo strategies to reduce animal use in genotoxicity testing. Mutagenesis 36(6):389–400. https://doi.org/10.1093/mutage/geab035
Guengerich FP, Shimada T (1998) Activation of procarcinogens by human cytochrome P450 enzymes. Mutat Res 400(1–2):201–213. https://doi.org/10.1016/s0027-5107(98)00037-2
Guo L, Dial S, Shi L et al (2011) Similarities and differences in the expression of drug-metabolizing enzymes between human hepatic cell lines and primary human hepatocytes. Drug Metab Dispos 39(3):528–538. https://doi.org/10.1124/dmd.110.035873
Habil MR, Doll MA, Hein DW (2020) N-acetyltransferase 2 acetylator genotype-dependent N-acetylation of 4-aminobiphenyl in cryopreserved human hepatocytes. Pharmacogenet Genomics 30(3):61–65. https://doi.org/10.1097/FPC.0000000000000394
Habil MR, Salazar-González RA, Doll MA, Hein DW (2022) Differences in β-naphthylamine metabolism and toxicity in Chinese hamster ovary cell lines transfected with human CYP1A2 and NAT2*4, NAT2*5B or NAT2*7B N-acetyltransferase 2 haplotypes. Arch Toxicol. https://doi.org/10.1007/s00204-022-03367-2
Hakura A, Shimada H, Nakajima M et al (2005) Salmonella/human S9 mutagenicity test: a collaborative study with 58 compounds. Mutagenesis 20(3):217–228. https://doi.org/10.1093/mutage/gei029
Hammons GJ, Milton D, Stepps K, Guengerich FP, Tukey RH, Kadlubar FF (1997) Metabolism of carcinogenic heterocyclic and aromatic amines by recombinant human cytochrome P450 enzymes. Carcinogenesis 18(4):851–854. https://doi.org/10.1093/carcin/18.4.851
Hein DW (2006) N-acetyltransferase 2 genetic polymorphism: effects of carcinogen and haplotype on urinary bladder cancer risk. Oncogene 25(11):1649–1658. https://doi.org/10.1038/sj.onc.1209374
Hein DW, Doll MA (2017) Role of the N-acetylation polymorphism in solithromycin metabolism. Pharmacogenomics 18(8):765–772. https://doi.org/10.2217/pgs-2017-0045
Hein DW, Doll MA, Rustan TD et al. (1993) Metabolic activation and deactivation of arylamine carcinogens by recombinant human NAT1 and polymorphic NAT2 acetyltransferases. Carcinogenesis 14(8):1633–1638. https://doi.org/10.1093/carcin/14.8.1633
Hu H, Gehart H, Artegiani B et al (2018) Long-term expansion of functional mouse and human hepatocytes as 3D organoids. Cell 175(6):1591–1606. https://doi.org/10.1016/j.cell.2018.11.013
Kemp CJ (2015) Animal models of chemical carcinogenesis: driving breakthroughs in cancer research for 100 years. Cold Spring Harb Protoc 2015(10):865–874. https://doi.org/10.1101/pdb.top069906
Kirkland D, Pfuhler S, Tweats D et al (2007) How to reduce false positive results when undertaking in vitro genotoxicity testing and thus avoid unnecessary follow-up animal tests: report of an ECVAM workshop. Mutat Res 628(1):31–55. https://doi.org/10.1016/j.mrgentox.2006.11.008
Kobets T, Duan JD, Brunnemann KD, Vock E, Deschl U, Williams GM (2019) DNA-damaging activities of twenty-four structurally diverse unsubstituted and substituted cyclic compounds in embryo-fetal chicken livers. Mutat Res Genet Toxicol Environ Mutagen 844:10–24. https://doi.org/10.1016/j.mrgentox.2019.06.004
Krech E, Selinski S, Blaszkewicz M et al (2017) Urinary bladder cancer risk factors in an area of former coal, iron, and steel industries in Germany. J Toxicol Environ Health A 80(7–8):430–438. https://doi.org/10.1080/10937404.2017.1304719
Lin HD, Tseng YK, Yuh CH, Chen SC (2022) Low concentrations of 4-ABP promote liver carcinogenesis in human liver cells and a zebrafish model. J Hazard Mater 423(Pt A):126954. https://doi.org/10.1016/j.jhazmat.2021.126954
Lin HD, Wang FZ, Lee CY et al (2020) 4-Aminobiphenyl inhibits the DNA homologous recombination repair in human liver cells: The role of miR-630 in downregulating RAD18 and MCM8. Toxicology 440:152441. https://doi.org/10.1016/j.tox.2020.152441
McQueen CA, Chau B, Erickson RP, Tjalkens RB, Philbert MA (2003) The effects of genetic variation in N-acetyltransferases on 4-aminobiphenyl genotoxicity in mouse liver. Chem Biol Interact 146(1):51–60. https://doi.org/10.1016/S0009-2797(03)00073-5
McQueen CA, Maslansky CJ, Williams GM (1983) Role of the acetylation polymorphism in determining susceptibility of cultured rabbit hepatocytes to DNA damage by aromatic amines. Cancer Res 43(7):3120–3123
Mittal RD, Srivastava DS, Mandhani A (2004) NAT2 gene polymorphism in bladder cancer: a study from North India. Int Braz J Urol 30(4):279–285. https://doi.org/10.1590/s1677-55382004000400003
Müller K, Kasper P, Müller L (1993) An assessment of the in vitro hepatocyte micronucleus assay. Mutat Res Environ Mutagen Relat Subj 292(3):213–224. https://doi.org/10.1016/0165-1161(93)90024-T
Nakano M, Shinagawa T, Eitaki Y et al (2021) Risk of bladder cancer in male Japanese workers exposed to ortho-toluidine and other aromatic amines. Int Arch Occup Environ Health 94(6):1427–1439
Nauwelaers G, Bessette EE, Gu D et al (2011) DNA adduct formation of 4-aminobiphenyl and heterocyclic aromatic amines in human hepatocytes. Chem Res Toxicol 24(6):913–925. https://doi.org/10.1021/tx200091y
Neis JM, van Gemert PJL, Roelofs HMJ, Bos RP, Henderson PT (1985) Metabolic activation of N-acetylbenzidine and N, N′-diacetylbenzidine to mutagens, using isolated hepatocytes and the 9000×g liver supernatant from rat, hamster and guinea pig. Mutat Res Fundam Mol Mech Mutagen 151(2):195–200. https://doi.org/10.1016/0027-5107(85)90071-5
O’Brien PJ, Chan K, Silber PM (2004) Human and animal hepatocytes in vitro with extrapolation in vivo. Chem Biol Interact 150(1):97–114. https://doi.org/10.1016/j.cbi.2004.09.003
Ohnishi S, Murata M, Kawanishi S (2002) Oxidative DNA damage induced by a metabolite of 2-naphthylamine, a smoking-related bladder carcinogen. Jpn J Cancer Res 93(7):736–743. https://doi.org/10.1111/j.1349-7006.2002.tb01314.x
Orzechowski A, Schrenk D, Bock KW (1992) Metabolism of 1- and 2-naphthylamine in isolated rat hepatocytes. Carcinogenesis 13(12):2227–2232. https://doi.org/10.1093/carcin/13.12.2227
Pathak KV, Bellamri M, Wang Y, Langouet S, Turesky RJ (2015) 2-Amino-9H-pyrido[2,3-b]indole (AalphaC) adducts and thiol oxidation of serum albumin as potential biomarkers of tobacco smoke. J Biol Chem 290(26):16304–16318. https://doi.org/10.1074/jbc.M115.646539
Salazar-Gonzalez RA, Zhang X, Doll MA, Lykoudi A, Hein DW (2019) Role of the human N-acetyltransferase 2 genetic polymorphism in metabolism and genotoxicity of 4,4′-methylenedianiline. Arch Toxicol 93(8):2237–2246. https://doi.org/10.1007/s00204-019-02516-4
Saletta F, Matullo G, Manuguerra M, Arena S, Bardelli A, Vineis P (2007) Exposure to the tobacco smoke constituent 4-aminobiphenyl induces chromosomal instability in human cancer cells. Cancer Res 67(15):7088–7094. https://doi.org/10.1158/0008-5472.CAN-06-4420
Sanada H, Koyama N, Wako Y, Kawasako K, Hamada S (2015) Repeated-dose liver micronucleus test of 4,4’-methylenedianiline using young adult rats. Mutat Res Genet Toxicol Environ Mutagen 780–781:31–35. https://doi.org/10.1016/j.mrgentox.2014.06.001
Skare JA, Hewitt NJ, Doyle E, Powrie R, Elcombe C (2009) Metabolite screening of aromatic amine hair dyes using in vitro hepatic models. Xenobiotica 39(11):811–825. https://doi.org/10.3109/00498250903134443
Sugamori KS, Brenneman D, Grant DM (2006) In vivo and in vitro metabolism of arylamine procarcinogens in acetyltransferase-deficient mice. Drug Metab Dispos 34(10):1697–1702. https://doi.org/10.1124/dmd.106.010819
Tomioka K, Saeki K, Obayashi K, Kurumatani N (2016) Risk of lung cancer in workers exposed to benzidine and/or beta-naphthylamine: a systematic review and meta-analysis. J Epidemiol 26(9):447–458. https://doi.org/10.2188/jea.JE20150233
Vineis P, Marinelli D, Autrup H et al (2001) Current smoking, occupation, N-acetyltransferase-2 and bladder cancer: a pooled analysis of genotype-based studies. Cancer Epidemiol Biomark Prev 10(12):1249–1252
Wahyuni EA, Chen CY, Wu HN, Chien CC, Chen SC (2021) Propolis alleviates 4-aminobiphenyl-induced oxidative DNA damage by inhibition of CYP2E1 expression in human liver cells. Environ Toxicol 36(8):1504–1513. https://doi.org/10.1002/tox.23147
Wang H, Iwasaki M, Haiman CA et al (2015) Interaction between red meat intake and NAT2 genotype in increasing the risk of colorectal cancer in Japanese and African Americans. PLoS ONE 10(12):e0144955. https://doi.org/10.1371/journal.pone.0144955
You Z, Brezzell MD, Das SK, Hooberman BH, Sinsheimer JE (1994) Substituent effects on the in vitro and in vivo genotoxicity of 4-aminobiphenyl and 4-aminostilbene derivatives. Mutat Res Genet Toxicol 320(1):45–58. https://doi.org/10.1016/0165-1218(94)90058-2
Zhang XF, Bian JC, Zhang XY et al (2005) Are polymorphisms of N-acetyltransferase genes susceptible to primary liver cancer in Luoyang, China? World J Gastroenterol 11(10):1457–1462. https://doi.org/10.3748/wjg.v11.i10.1457
Zhu Z, Zhang J, Jiang W, Zhang X, Li Y, Xu X (2015) Risks on N-acetyltransferase 2 and bladder cancer: a meta-analysis. Onco Targets Ther 8:3715–3720. https://doi.org/10.2147/OTT.S82927
Acknowledgements
This work was partially supported by United States Public Health Service Grants P20-GM113226, P30-ES030283 and P42-ES023716.
Author information
Authors and Affiliations
Contributions
MRH: writing the original draft, doing experiments, data visualization, formal analysis, review and editing. RAS-G: doing experiments, data visualization, formal analysis, investigation, writing, review and editing. MAD: doing experiments, data visualization, formal analysis, investigation, writing, review and editing. DWH: conceptualization, methodology, validation, formal analysis, resources, writing, review and editing, visualization, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Habil, M.R., Salazar-González, R.A., Doll, M.A. et al. N-acetyltransferase 2 acetylator genotype-dependent N-acetylation and toxicity of the arylamine carcinogen β-naphthylamine in cryopreserved human hepatocytes. Arch Toxicol 96, 3257–3263 (2022). https://doi.org/10.1007/s00204-022-03381-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-022-03381-4