Abstract
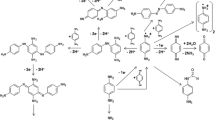
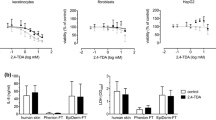
Para-phenylenediamine (PPD) is one of the most used chemicals in oxidative hair dyes. However, its use has been associated with adverse effects on health, including contact dermatitis and other systemic toxicities. Novel PPD derivatives have been proposed as a safer replacement for PPD. This can be achieved if these molecules minimally permeate the skin and/or are easily metabolised by enzymes in the skin (e.g., N-acetyltransferase-1 (NAT-1)) into innocuous compounds before gaining systemic entry. This study investigated the detoxification pathway mediated by NAT-1 enzymes on 6 synthesized PPD analogues (namely, P1–P6) with different chemical properties, to study the role of functional groups on detoxification mechanisms in HaCaT skin cells. These compounds were carefully designed with different chemical properties (whereby the ortho position of PPD was substituted by nucleophile and electrophile groups to promote N-acetylation reactions, metabolism and clearance). Compounds P2–P4 N-acetylated at 54–49 nmol/mg/min, which is 1.6 times higher than N-acetylation of PPD, upregulated NAT-1 activity from 8–7% at 50 μM to 22–11% at 100 μM and showed 4 times higher rate of elimination (k equal to 0.141 ± 0.016–0.124 ± 0.01 h−1) and 3 times faster rate of clearance (0.172 ± 0.007–0.158 ± 0.005 h−1mgprotein−1) than PPD (0.0316 ± 0.0019 h−1, 0.0576 ± 0.003 h−1mg protein−1, respectively). The data suggest that nucleophile substituted compounds detoxify at a faster rate than PPD. Our metabolic and detoxification mechanistic studies revealed significantly higher rates of N-acetylation, NAT-1 activity and higher detoxification of P2–P4 in keratinocytes, suggesting the importance of nucleophilic groups at the ortho position in PPD to reduce toxicity of aniline-based dyes on human skin cells.








Similar content being viewed by others
Abbreviations
- NAT-1:
-
N-Acetyl transferase-1
- PPD:
-
Para-phenylenediamine
- ACD:
-
Acute contact dermatitis
- HaCaT:
-
Immortalized Human Keratinocytes cells
- PABA:
-
para-Aminobenzoic acid
- CoASAc:
-
Acetyl coenzyme A
- V max :
-
The maximum rate of reaction
- K m :
-
The Michalis–Menten constant
- CLinvitro :
-
In vitro clearance
- qRT-PCR:
-
Real-time quantitative reverse transcription PCR
- MAPPD:
-
Monoacetyl para-phenylenediamine
- DAPPD:
-
Diacetyl para-phenylenediamine
- EWG:
-
Electron withdrawing groups
- EDG:
-
Electron donating groups
- Nu-:
-
Nucleophile
- E + :
-
Electrophile
References
Abd E, Yousef SA, Pastore MN, Telaprolu K et al (2016) Skin models for the testing of transdermal drugs. Clin Pharmacol 8:163–176. https://doi.org/10.2147/CPAA.S64788
Bhaiya P, Roychowdhury S, Vyas PM, Doll MA, Hein DW, Svensson CK (2006) Bioactivation, protein haptenation, and toxicity of sulfamethoxazole and dapsone in normal human dermal fibroblasts. Toxicol Appl Pharmacol 215(2):158–167. https://doi.org/10.1016/j.taap.2006.02.006
Bonifas J, Scheitza S, Clemens J, Blömeke B (2010) Characterization of N-acetyltransferase 1 activity in human keratinocytes and modulation by para-phenylenediamine. J Pharmacol Exp Ther 334(1):318–326. https://doi.org/10.1124/jpet.110.167874
Bos JD, Meinardi MM (2000) The 500 Dalton rule for the skin penetration of chemical compounds and drugs. Exp Dermatol 9(3):165–169. https://doi.org/10.1034/j.1600-0625.2000.009003165.x
Butcher NJ, Boukouvala S, Sim E, Minchin RF (2002) Pharmacogenetics of the arylamine N-acetyltransferases. Pharmacogenomics J 2(1):30–42. https://doi.org/10.1038/sj.tpj.6500053
Chaudhary SC, Sawlani KK, Singh K (2013) Paraphenylenediamine poisoning. Niger J Clin Pract 16(2):258–259. https://doi.org/10.4103/1119-3077.110138
Dancik Y, Bigliardi PL, Bigliardi MQ (2015) What happens in the skin? Integrating skin permeation kinetics into studies of developmental and reproductive toxicity following topical exposure. Reprod Toxicol 58:252–281. https://doi.org/10.1016/j.reprotox.2015.10.001
Hamann D, Yazar K, Hamann CR, Thyssen JP, Lidén C (2014) P-Phenylenediamine and other allergens in hair dye products in the United States: a consumer exposure study. Contact Dermat 70(4):213–218. https://doi.org/10.1111/cod.12164
Jacobson KB (1960) Inhibition of acetyl transfer by amethopterin. J Biol Chem 235(9):2713–2716. https://doi.org/10.1016/S0021-9258(19)76941-5
Kawakubo Y, Merk HF, Masaoudi TA, Sieben S, Blömeke B (2000) N-acetylation of paraphenylenediamine in human skin and keratinocytes. J Pharmacol Exp Ther 292(1):150–155
Krasteva M, Bons B, Ryan C, Gerberick GF (2009) Consumer allergy to oxidative hair coloring products: epidemiologic data in the literature. Dermatitis 20(3):123–141. https://doi.org/10.2310/6620.2009.08089
Moeller R, Lichter J, Blömeke B (2008) Impact of para-phenylenediamine cyclooxygenases expression and prostaglandin formation in human immortalized keratinocytes (HaCaT). Toxicology 249(2–3):167–175. https://doi.org/10.1016/j.tox.2008.05.002
Mukkanna KS, Stone NM, Ingram JR (2017) Para-phenylenediamine allergy: current perspectives on diagnosis and management. J Asthma Allergy 10:9–15. https://doi.org/10.2147/JAA.S90265
Nohynek GJ, Fautz R, Kiefferc FB, Toutaina H (2004) Toxicity and human health risk of hair dyes. Food Chem Toxicol 42(4):517–543. https://doi.org/10.1016/j.fct.2003.11.003
Nohynek GJ, Duche D, Garrigues A, Meunier PA, Toutain H, Leclaire J (2005) Under the skin: biotransformation of para-aminophenol and para-phenylenediamine in reconstructed human epidermis and human hepatocytes. Toxicol Lett 158(3):196–212. https://doi.org/10.1016/j.toxlet.2005.03.014
Pot LM, Scheitza SM, Coenraads P-J, Blomeke B (2013) Penetration and haptenation of p-phenylenediamine. Contact Dermat 68:193–207. https://doi.org/10.1111/cod.12032
Riddle B, Jencks WP (1971) Acetyl-coenzyme A : arylamine IV-acetyltransferase, role of the acetyl-enzyme intermediate and the effects of substituents on the rate. J Biol Chem 246(10):3250–3258. https://doi.org/10.1016/S0021-9258(18)62221-5
Schnuch A, Lessmann H, Frosch PJ, Uter W (2008) Para-Phenylenediamine: the profile of an important allergen. Results of the IVDK. Br J Dermatol 159(2):379–386. https://doi.org/10.1111/j.1365-2133.2008.08644.x
Sieben S, Kawakubo Y, Sachs B, Al Masaoudi T, Merk HF, Blömeke B (2001) T cell responses to paraphenylenediamine and to its metabolites mono- and diacetyl-paraphenylenediamine. Int Arch Allergy Immunol 124:356–358. https://doi.org/10.1159/000053756
Sim E, Abuhammad A, Ryan A (2014) Arylamine N-acetyltransferases: from drug metabolism and pharmacogenetics to drug discovery. Br J Pharmacol 171(11):2705–2725. https://doi.org/10.1111/bph.12598
Svensson CK (2009) Biotransformation of drugs in human skin. Drug Metab Dispos 37(2):247–253. https://doi.org/10.1124/dmd.108.024794
Thyssen JP, White JML et al (2008) Epidemiological data on consumer allergy to p-phenylenediamine. Contact Dermat 59(6):327–343. https://doi.org/10.1111/j.1600-0536.2008.01427.x
Venkatesan G, Dancik Y, Sinha A et al (2019) Facile synthesis of oligo anilines as permanent hair dyes: how chemical modifications impart colour and avoid toxicity. New J Chem 43:16188–16199. https://doi.org/10.1039/C9NJ03362A
Venkatesan G, Dancik Y, Sinha A et al (2021) Development of novel alternative hair dyes to hazardous para-phenylenediamine. J Hazard Mat 402:123712. https://doi.org/10.1016/j.jhazmat.2020.123712
Windmill KF, Gaedigk A, Hall PM et al (2000) Localization of N-acetyltransferases NAT-1 and NAT-2 in human tissues. Toxicol Sci 54(1):19–29. https://doi.org/10.1093/toxsci/54.1.19
Acknowledgements
This work was supported by the National University of Singapore (NUS), NUS Technology Acceleration Programme (TAP), Project ID TAP2002019-04-13, NanoNash Program (R-148-000-296-114 and R-148-000-284-114), the National University of Singapore Yong Loo Lin School of Medicine Nanomedicine Translational Research Program (NUHSRO/2021/034/TRP/09/Nanomedicine) and NUHSRO/2020/002/NanoNash/LOA.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical standards
The manuscript does not contain clinical studies or patient data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Venkatesan, G., Lim, Z.C., Karkhanis, A.V. et al. Investigations on detoxification mechanisms of novel para-phenylenediamine analogues through N-acetyltransferase 1 (NAT-1). Arch Toxicol 96, 153–165 (2022). https://doi.org/10.1007/s00204-021-03149-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-021-03149-2