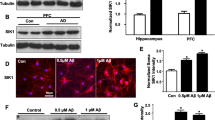
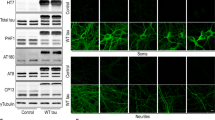
Abstract
Cadmium is a neurotoxic compound which induces cognitive alterations similar to those produced by Alzheimer’s disease (AD). However, the mechanism through which cadmium induces this effect remains unknown. In this regard, we described in a previous work that cadmium blocks cholinergic transmission and induces a more pronounced cell death on cholinergic neurons from basal forebrain which is partially mediated by AChE overexpression. Degeneration of basal forebrain cholinergic neurons, as happens in AD, results in memory deficits attributable to the loss of cholinergic modulation of hippocampal synaptic circuits. Moreover, cadmium has been described to activate GSK-3β, induce Aβ protein production and tau filament formation, which have been related to a selective loss of basal forebrain cholinergic neurons and development of AD. The present study is aimed at researching the mechanisms of cell death induced by cadmium on basal forebrain cholinergic neurons. For this purpose, we evaluated, in SN56 cholinergic mourine septal cell line from basal forebrain region, the cadmium toxic effects on neuronal viability through muscarinic M1 receptor, AChE splice variants, GSK-3β enzyme, Aβ and tau proteins. This study proves that cadmium induces cell death on cholinergic neurons through blockade of M1 receptor, overexpression of AChE-S and GSK-3β, down-regulation of AChE-R and increase in Aβ and total and phosphorylated tau protein levels. Our present results provide new understanding of the mechanisms contributing to the harmful effects of cadmium on cholinergic neurons and suggest that cadmium could mediate these mechanisms by M1R blockade through AChE splices altered expression.






Similar content being viewed by others
References
Anagnostaras SG, Murphy GG, Hamilton SE, Mitchell SL, Rahnama NP, Nathanson NM, Silva AJ (2003) Selective cognitive dysfunction in acetylcholine M1 muscarinic receptor mutant mice. Nat Neurosci 6(1):51–58
Andersson H, Petersson-Grawe K, Lindqvist E, Luthman J, Oskarsson A, Olson L (1997) Low-level cadmium exposure of lactating rats causes alterations in brain serotonin levels in the offspring. Neurotoxicol Teratol 19(2):105–115
Atri A, Sherman S, Norman KA, Kirchhoff BA, Nicolas MM, Greicius MD, Cramer SC, Breiter HC, Hasselmo ME, Stern CE (2004) Blockade of central cholinergic receptors impairs new learning and increases proactive interference in a word paired-associate memory task. Behav Neurosci 118(1):223–236
ATSDR. Agency for Toxic Substances and Disease Registry (2012) Toxicological profile for cadmium. Agency for Toxic Substances and Disease Registry, Atlanta
Auermann E, Dassler HG, Cumbrowski J, Kneuer M, Jacobi J, Kuhn H (1979) Cadmium content of vegetable foods in the effective range of a lead smelting plant. Nahrung 23(9–10):875–890
Avila J, Lucas JJ, Perez M, Hernandez F (2004) Role of tau protein in both physiological and pathological conditions. Physiol Rev 84(2):361–384
Beharry C, Cohen LS, Di J, Ibrahim K, Briffa-Mirabella SA, Adel C (2014) Tau-induced neurodegeneration: mechanisms and targets. Neurosci Bull 30(2):346–358
Berson A, Soreq H (2010) It all starts at the ends: multifaceted involvement of C- and N-terminally modified cholinesterases in Alzheimer’s disease. Rambam Maimonides Med J 1(2):e0014
Berson A, Knobloch M, Hanan M, Kirchhoff BA, Nicolas MM, Greicius MD, Cramer SC, Breiter HC, Hasselmo ME, Stern CE (2008) Changes in readthrough acetylcholinesterase expression modulate amyloid-beta pathology. Brain 131(Pt 1):109–119
Bierer LM, Haroutunian V, Gabriel S, Knott PJ, Carlin LS, Purohit DP, Perl DP, Schmeidler J, Kanof P, Davis KL (1995) Neurochemical correlates of dementia severity in Alzheimer’s disease: relative importance of the cholinergic deficits. J Neurochem 64(2):749–760
Bilgen I, Oner G, Edremitlioglu M, Alkan Z, Cirrik S (2003) Involvement of cholinoceptors in cadmium-induced endothelial dysfunction. J Basic Clin Physiol Pharmacol 14(1):55–76
Birikh KR, Sklan EH, Shoham S, Soreq H (2003) Interaction of “readthrough” acetylcholinesterase with RACK1 and PKCbeta II correlates with intensified fear-induced conflict behavior. Proc Natl Acad Sci USA 100(1):283–288
Bond CE, Patel P, Crouch L, Tetlow N, Day T, Abu-Hayyeh S, Williamson C, Greenfield SA (2006) Astroglia up-regulate transcription and secretion of ‘readthrough’ acetylcholinesterase following oxidative stress. Eur J Neurosci 24(2):381–386
Budd DC, McDonald J, Emsley N, Cain K, Tobin AB (2003) The C-terminal tail of the M3-muscarinic receptor possesses anti-apoptotic properties. J Biol Chem 278(21):19565–19573
Budd DC, Spragg EJ, Ridd K, Tobin AB (2004) Signalling of the M3-muscarinic receptor to the anti-apoptotic pathway. Biochem J 381(Pt 1):43–49
Campanari ML, Garcia-Ayllon MS, Belbin O, Galceran J, Lleo A, Saez-Valero J (2014) Acetylcholinesterase modulates presenilin-1 levels and gamma-secretase activity. J Alzheimer’s Dis 41(3):911–924
Davis AA, Fritz JJ, Wess J, Lah JJ, Levey AI (2010) Deletion of M1 muscarinic acetylcholine receptors increases amyloid pathology in vitro and in vivo. J Neurosci 30(12):4190–4196
De Sarno P, Shestopal SA, King TD, Zmijewska A, Song L, Jope RS (2003) Muscarinic receptor activation protects cells from apoptotic effects of DNA damage, oxidative stress, and mitochondrial inhibition. J Biol Chem 278(13):11086–11093
Del Pino J, Zeballos G, Anadon MJ, Capo MA, Diaz MJ, Garcia J, Frejo MT (2014) Higher sensitivity to cadmium induced cell death of basal forebrain cholinergic neurons: a cholinesterase dependent mechanism. Toxicology 325:151–159
Dori A, Cohen J, Silverman WF, Pollack Y, Soreq H (2005) Functional manipulations of acetylcholinesterase splice variants highlight alternative splicing contributions to murine neocortical development. Cereb Cortex 15(4):419–430
Everitt BJ, Robbins TW (1997) Central cholinergic systems and cognition. Annu Rev Psychol 48:649–684
Evron T, Greenberg D, Mor TS, Soreq H (2007) Adaptive changes in acetylcholinesterase gene expression as mediators of recovery from chemical and biological insults. Toxicology 233(1–3):97–107
Falnoga I, Tusek-Znidaric M, Horvat M, Stegnar P (2000) Mercury, selenium, and cadmium in human autopsy samples from Idrija residents and mercury mine workers. Environ Res 84(3):211–218
Fisher A (2012) Cholinergic modulation of amyloid precursor protein processing with emphasis on M1 muscarinic receptor: perspectives and challenges in treatment of Alzheimer’s disease. J Neurochem 120(Suppl 1):22–33
Ghelardini C, Galeotti N, Matucci R, Bellucci C, Gualtieri F, Capaccioli S, Quattrone A, Bartolini A (1999) Antisense ‘knockdowns’ of M1 receptors induces transient anterograde amnesia in mice. Neuropharmacology 38(3):339–348
Gill TM, Gallagher M (1998) Evaluation of muscarinic M2 receptor sites in basal forebrain and brainstem cholinergic systems of behaviorally characterized young and aged long-evans rats. Neurobiol Aging 19(3):217–225
Giraldo E, Lloret A, Fuchsberger T, Vina J (2014) Abeta and tau toxicities in Alzheimer’s are linked via oxidative stress-induced p38 activation: protective role of vitamin E. Redox Biol 2:873–877
Gkanti V, Stolakis V, Kalafatakis K, Liapi C, Zissis KM, Zarros A, Tsakiris S (2014) Postnuclear supernatants of rat brain regions as substrates for the in vitro assessment of cadmium-induced neurotoxicity on acetylcholinesterase activity. Biol Trace Elem Res 158(1):87–89
Goncalves JF, Fiorenza AM, Spanevello RM, Mazzanti CM, Bochi GV, Antes FG, Stefanello N, Rubin MA, Dressler VL, Morsch VM, Schetinger MR (2010) N-acetylcysteine prevents memory deficits, the decrease in acetylcholinesterase activity and oxidative stress in rats exposed to cadmium. Chem Biol Interact 186(1):53–60
Graham ES, Woo KK, Aalderink M, Fry S, Greenwood JM, Glass M, Dragunow M (2013) M1 muscarinic receptor activation mediates cell death in M1-HEK293 cells. PLoS One 8(9):e72011
Greenberg DS, Toiber D, Berson A, Soreq H (2010) Acetylcholinesterase variants in Alzheimer’s disease: from neuroprotection to programmed cell death. Neurodegener Dis 7(1–3):60–63
Grisaru D, Pick M, Perry C, Sklan EH, Almog R, Goldberg I, Naparstek E, Lessing JB, Soreq H, Deutsch V (2006) Hydrolytic and nonenzymatic functions of acetylcholinesterase comodulate hemopoietic stress responses. J Immunol 176(1):27–35
Gutierrez-Reyes EY, Albores A, Rios C (1998) Increase of striatal dopamine release by cadmium in nursing rats and its prevention by dexamethasone-induced metallothionein. Toxicology 131(2–3):145–154
Hammond DN, Lee HJ, Tonsgard JH, Wainer BH (1990) Development and characterization of clonal cell lines derived from septal cholinergic neurons. Brain Res 512(2):190–200
Hawkes C, Jhamandas JH, Kar S (2005) Selective loss of basal forebrain cholinergic neurons by 192 IgG-saporin is associated with decreased phosphorylation of Ser glycogen synthase kinase-3beta. J Neurochem 95(1):263–272
Hedlund B, Bartfai T (1979) The importance of thiol- and disulfide groups in agonist and antagonist binding to the muscarinic receptor. Mol Pharmacol 15(3):531–544
Hedlund B, Gamarra M, Bartfai T (1979) Inhibition of striatal muscarinic receptors in vivo by cadmium. Brain Res 168(1):216–218
Hossain S, Liu HN, Nguyen M, Shore G, Almazan G (2009) Cadmium exposure induces mitochondria-dependent apoptosis in oligodendrocytes. Neurotoxicology 30(4):544–554
Hudgens ED, Ji L, Carpenter CD, Petersen SL (2009) The gad2 promoter is a transcriptional target of estrogen receptor (ER)alpha and ER beta: a unifying hypothesis to explain diverse effects of estradiol. J Neurosci 29(27):8790–8797
IPCS. International program on chemical safety (1992) Environmental health criteria 134. Cadmium. WHO, Geneva
Jiang LF, Yao TM, Zhu ZL, Wang C, Ji LN (2007) Impacts of Cd(II) on the conformation and self-aggregation of Alzheimer’s tau fragment corresponding to the third repeat of microtubule-binding domain. Biochim Biophys Acta 11(21):24
Jin T, Lu J, Nordberg M (1998) Toxicokinetics and biochemistry of cadmium with special emphasis on the role of metallothionein. Neurotoxicology 19(4–5):529–535
Kar S, Slowikowski SP, Westaway D, Mount HT (2004) Interactions between beta-amyloid and central cholinergic neurons: implications for Alzheimer’s disease. J Psychiatry Neurosci 29(6):427–441
Klein AM, Kowall NW, Ferrante R (1999) Neurotoxicity and oxidative damage of beta amyloid 1–42 versus beta amyloid 1–40 in the mouse cerebral cortex. Ann NY Acad Sci 893:314–320
Lahmy V, Meunier J, Malmstrom S, Naert G, Givalois L, Kim SH, Villard V, Vamvakides A, Maurice T (2013) Blockade of Tau hyperphosphorylation and Abeta(1)(−)(4)(2) generation by the aminotetrahydrofuran derivative ANAVEX2-73, a mixed muscarinic and sigma(1) receptor agonist, in a nontransgenic mouse model of Alzheimer’s disease. Neuropsychopharmacology 38(9):1706–1723
Li X, Lv Y, Yu S, Zhao H, Yao L (2012) The effect of cadmium on Abeta levels in APP/PS1 transgenic mice. Exp Ther Med 4(1):125–130
Lindenboim L, Pinkas-Kramarski R, Sokolovsky M, Stein R (1995) Activation of muscarinic receptors inhibits apoptosis in PC12M1 cells. J Neurochem 64(6):2491–2499
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25(4):402–408
Luchese C, Brandao R, de Oliveira R, Nogueira CW, Santos FW (2007) Efficacy of diphenyl diselenide against cerebral and pulmonary damage induced by cadmium in mice. Toxicol Lett 173(3):181–190
Nitsch RM, Slack BE, Wurtman RJ, Growdon JH (1992) Release of Alzheimer amyloid precursor derivatives stimulated by activation of muscarinic acetylcholine receptors. Science 258(5080):304–307
Panayi AE, Spyrou NM, Iversen BS, White MA, Part P (2002) Determination of cadmium and zinc in Alzheimer’s brain tissue using inductively coupled plasma mass spectrometry. J Neurol Sci 195(1):1–10
Pari L, Murugavel P (2007) Diallyl tetrasulfide improves cadmium induced alterations of acetylcholinesterase, ATPases and oxidative stress in brain of rats. Toxicology 234(1–2):44–50
Pavia J, de Ceballos ML, Sanchez de la Cuesta F (1998) Alzheimer’s disease: relationship between muscarinic cholinergic receptors, beta-amyloid and tau proteins. Fundam Clin Pharmacol 12(5):473–481
Rojo AI, Sagarra MR, Cuadrado A (2008) GSK-3beta down-regulates the transcription factor Nrf2 after oxidant damage: relevance to exposure of neuronal cells to oxidative stress. J Neurochem 105(1):192–202
Salmon A, Erb C, Meshorer E, Ginzberg D, Adani Y, Rabinovitz I, Amitai G, Soreq H (2005) Muscarinic modulations of neuronal anticholinesterase responses. Chem Biol Interact 157–158:105–113
Sarchielli E, Pacini S, Morucci G, Punzi T, Marini M, Vannelli GB, Gulisano M (2012) Cadmium induces alterations in the human spinal cord morphogenesis. Biometals 25(1):63–74
Scheiderer CL, McCutchen E, Thacker EE, Kolasa K, Ward MK, Parsons D, Harrell LE, Dobrunz LE, McMahon LL (2006) Sympathetic sprouting drives hippocampal cholinergic reinnervation that prevents loss of a muscarinic receptor-dependent long-term depression at CA3-CA1 synapses. J Neurosci 26(14):3745–3756
Shaltiel G, Hanan M, Wolf Y, Barbash S, Kovalev E, Shoham S, Soreq H (2013) Hippocampal microRNA-132 mediates stress-inducible cognitive deficits through its acetylcholinesterase target. Brain Struct Funct 218(1):59–72
Shukla A, Shukla GS, Srimal RC (1996) Cadmium-induced alterations in blood-brain barrier permeability and its possible correlation with decreased microvessel antioxidant potential in rat. Hum Exp Toxicol 15(5):400–405
Silman I, Sussman JL (2005) Acetylcholinesterase: ‘classical’ and ‘non-classical’ functions and pharmacology. Curr Opin Pharmacol 5(3):293–302
Smedman M, Potempska A, Rubenstein R, Ju W, Ramakrishna N, Denman RB (1997) Effects of cadmium, copper, and zinc and beta APP processing and turnover in COS-7 and PC12 cells. Relationship to Alzheimer disease pathology. Mol Chem Neuropathol 31(1):13–28
Soreq H, Seidman S (2001) Acetylcholinesterase—new roles for an old actor. Nat Rev Neurosci 2(4):294–302
Tamagno E, Parola M, Bardini P, Piccini A, Borghi R, Guglielmotto M, Santoro G, Davit A, Danni O, Smith MA, Perry G, Tabaton M (2005) Beta-site APP cleaving enzyme up-regulation induced by 4-hydroxynonenal is mediated by stress-activated protein kinases pathways. J Neurochem 92(3):628–636
Toiber D, Berson A, Greenberg D, Melamed-Book N, Diamant S, Soreq H (2008) N-acetylcholinesterase-induced apoptosis in Alzheimer’s disease. PLoS One 3(9):e3108
Toiber D, Greenberg DS, Soreq H (2009) Pro-apoptotic protein-protein interactions of the extended N-AChE terminus. J Neural Transm 116(11):1435–1442
Voytko ML (1996) Cognitive functions of the basal forebrain cholinergic system in monkeys: memory or attention? Behav Brain Res 75(1–2):13–25
Wang B, Du Y (2013) Cadmium and its neurotoxic effects. Oxid Med Cell Longev 2013:898034
Wang SH, Shih YL, Kuo TC, Ko WC, Shih CM (2009) Cadmium toxicity toward autophagy through ROS-activated GSK-3beta in mesangial cells. Toxicol Sci 108(1):124–131
Ward NL, Hagg T (2000) BDNF is needed for postnatal maturation of basal forebrain and neostriatum cholinergic neurons in vivo. Exp Neurol 162(2):297–310
Zheng WH, Bastianetto S, Mennicken F, Ma W, Kar S (2002) Amyloid beta peptide induces tau phosphorylation and loss of cholinergic neurons in rat primary septal cultures. Neuroscience 115(1):201–211
Zimmermann M (2013) Neuronal AChE splice variants and their non-hydrolytic functions: redefining a target of AChE inhibitors? Br J Pharmacol 170(5):953–967
Acknowledgments
The authors thank Miguel Capo and Margarita Lobo, Professors of Toxicology from Universidad Complutense de Madrid, for their counseling during the preparation of the present work.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Javier del Pino and Gabriela Zeballos have contributed equally to this work.
Rights and permissions
About this article
Cite this article
del Pino, J., Zeballos, G., Anadón, M.J. et al. Cadmium-induced cell death of basal forebrain cholinergic neurons mediated by muscarinic M1 receptor blockade, increase in GSK-3β enzyme, β-amyloid and tau protein levels. Arch Toxicol 90, 1081–1092 (2016). https://doi.org/10.1007/s00204-015-1540-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-015-1540-7