Abstract
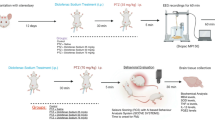
Sarin, a potent organophosphate cholinesterase inhibitor, induces an array of toxic effects including convulsions. Many antidotal treatments contain anticonvulsants to block seizure activity and the ensuing brain damage. Magnesium sulfate (MGS) is used to suppress eclamptic seizures in pregnant women with hypertension and was shown to block kainate-induced convulsions. Magnesium sulfate was evaluated herein as an anticonvulsant against sarin poisoning and its efficacy was compared with the potent anticonvulsants midazolam (MDZ) and caramiphen (CRM). Rats were exposed to a convulsant dose of sarin (96 μg/kg, im) and 1 min later treated with the oxime TMB4 and atropine to increase survival. Five minutes after initiation of convulsions, MGS, CRM, or MDZ were administered. Attenuation of tonic–clonic convulsions was observed following all these treatments. However, radio-telemetric electro-corticography (ECoG) monitoring demonstrated sustained seizure activity in MGS-injected animals while this activity was completely blocked by MDZ and CRM. This disrupted brain activity was associated with marked increase in brain translocator protein levels, a marker for brain damage, measured 1 week following exposure. Additionally, histopathological analyses of MGS-treated group showed typical sarin-induced brain injury excluding the hippocampus that was partially protected. Our results clearly show that MGS demonstrated misleading features as an anticonvulsant against sarin-induced seizures. This stems from the dissociation observed between overt convulsions and seizure activity. Thus, the presence or absence of motor convulsions may be an unreliable indicator in the assessment of clinical status and in directing adequate antidotal treatments following exposure to nerve agents in battle field or terror attacks.








Similar content being viewed by others
Abbreviations
- ACh:
-
Acetylcholine
- AChE:
-
Acetylcholinesterase
- CNS:
-
Central nervous system
- CRM:
-
Caramiphen
- ECoG:
-
Electrocorticogram
- GFAP:
-
Glial fibrillary acidic protein
- Glu:
-
Glutamate
- MDZ:
-
Midazolam
- MGS:
-
Magnesium sulfate
- MAP-2:
-
Microtubule-associated protein 2
- NeuN:
-
Neuronal nuclear antigen
- NMDA:
-
N-Methyl-D-Aspartate
- OP:
-
Organophosphorous
- TA:
-
TMB4 + Atropine
- TSPO:
-
Translocator protein
References
Baille V, Dorandeu F, Carpentier P, Bizot JC, Filliat P, Four E, Denis J, Lallement G (2001) Acute exposure to a low or mild dose of soman: biochemical, behavioral and histopathological effects. Pharmacol Biochem Behav 69:561–569
Baille-Le Crom V, Collombet JM, Carpentier P, Brochier G, Burckhart MF, Foquin A, Pernot-Marino I, Rondouin G, Lallement G (1995) Early regional changes of GFAP mRNA in rat hippocampus and dentate gyrus during soman-induced seizures. NeuroReport 7:365–369
Balali-Mood M, Balali-Mood K (2008) Neurotoxic disorders of organophosphorus compounds and their managements. Arch Iran Med 11:65–89
Ballough GP, Cann FJ, Smith CD, Forster JS, Kling CE, Filbert MG (1998) GM1 monosialoganglioside pretreatment protects against soman-induced seizure-related brain damage. Mol Chem Neuropathol 34:1–23
Benavides J, Fage D, Carter C, Scatton B (1987) Peripheral type benzodiazepine binding sites are a sensitive indirect index of neuronal damage. Brain Res 421:167–172
Benavides J, Dubois A, Scatton B (2001) Peripheral type benzodiazepine binding sites as a tool for the detection and quantification of CNS injury. Curr Protoc Neurosci Chapter 7: Unit 7 16
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chapman S, Kadar T, Gilat E (2006) Seizure duration following sarin exposure affects neuro-inflammatory markers in the rat brain. Neurotoxicology 27:277–283
Cotton DB, Hallak M, Janusz C, Irtenkauf SM, Berman RF (1993) Central anticonvulsant effects of magnesium sulphate on N-methyl-D-aspartate-induced seizures. Am J Obstet Gynecol 168:974–978
Cox JA, Lysko PG, Henneberry RC (1989) Excitatory amino acid neurotoxicity at the N-methyl-D-aspartate receptor in cultured neurons: role of the voltage-dependent magnesium block. Brain Res 499:267–272
Dingledine R, McBain CJ, McNamara JO (1990) Excitatory amino acid receptors in epilepsy. Trends Pharmacol Sci 11:334–338
Donaldson JO (1986) Does magnesium sulphate treat eclamptic convulsions? Clin Neuropharmacol 9:37–45
Euser AG, Cipolla MJ (2009) Magnesium sulphate for the treatment of eclampsia: a brief review. Stroke 40:1169–1175
Filbert MG, Forster JS, Smith CD, Ballough GP (1999) Neuroprotective effects of HU-211 on brain damage resulting from soman-induced seizures. Ann N Y Acad Sci 890:505–514
Finkelstein A, Kunis G, Berkutzki T, Ronen A, Krivoy A, Yoles E, Last D, Mardor Y, Van Shura K, McFarland E, Capacio BA, Eisner C, Gonzales M, Gregorowicz D, Eisenkraft A, McDonough JH, Schwartz M (2012) Immunomodulation by poly-YE reduces organophosphate-induced brain damage. Brain Behav Immun 26(1):159–169
Gilat E, Kadar T, Levy A, Rabinovitz I, Cohen G, Kapon Y, Sahar R, Brandeis R (2005) Anticonvulsant treatment of sarin-induced seizures with nasal midazolam: an electrographic, behavioral, and histological study in freely moving rats. Toxicol Appl Pharmacol 209:74–85
Hallak M (1998) Effect of parenteral magnesium sulphate administration on excitatory amino acid receptors in the rat brain. Magnes Res 11:117–131
Hallak M, Berman RF, Irtenkauf SM, Evans MI, Cotton DB (1992) Peripheral magnesium sulphate enters the brain and increases the threshold for hippocampal seizures in rats. Am J Obstet Gynecol 167:1605–1610
Hallak M, Berman RF, Irtenkauf SM, Janusz CA, Cotton DB (1994) Magnesium sulphate treatment decreases N-methyl-D-aspartate receptor binding in the rat brain: an autoradiographic study. J Soc Gynecol Investig 1:25–30
Jutkiewicz EM, Baladi MG, Folk JE, Rice KC, Woods JH (2006) The convulsive and electroencephalographic changes produced by nonpeptidic delta-opioid agonists in rats: comparison with pentylenetetrazol. J Pharmacol Exp Ther 317:1337–1348
Kadar T, Cohen G, Sahar R, Alkalai D, Shapira S (1992) Long-term study of brain lesions following soman, in comparison to DFP and metrazol poisoning. Hum Exp Toxicol 11:517–523
Kadar T, Shapira S, Cohen G, Sahar R, Alkalay D, Raveh L (1995) Sarin-induced neuropathology in rats. Hum Exp Toxicol 14:252–259
Lallement G, Mestries JC, Privat A, Brochier G, Baubichon D, Carpentier P, Kamenka JM, Sentenac-Roumanou H, Burckhart MF, Peoc’h M (1997) GK 11: promising additional neuroprotective therapy for organophosphate poisoning. Neurotoxicology 18:851–856
Lemercier G, Carpentier P, Sentenac-Roumanou H, Morelis P (1983) Histological and histochemical changes in the central nervous system of the rat poisoned by an irreversible anticholinesterase organophosphorus compound. Acta Neuropathol (Berl) 61:123–129
Lennox WJ, Harris LW, Talbot BG, Anderson DR (1985) Relationship between reversible acetylcholinesterase inhibition and efficacy against soman lethality. Life Sci 37:793–798
Marrs TC (2003) Diazepam in the treatment of organophosphorus ester pesticide poisoning. Toxicol Rev 22:75–81
McDonough JH Jr, Shih TM (1993) Pharmacological modulation of soman-induced seizures. Neurosci Biobehav Rev 17:203–215
McDonough JH, Shih T-M (1997) Neuropharmacological Mechanisms of Nerve Agent-induced Seizure and Neuropathology. Neurosci Biobehav Rev 21:559–579
McIntosh TK, Vink R, Yamakami I, Faden AI (1989) Magnesium protects against neurological deficit after brain injury. Brain Res 482:252–260
Myhrer T, Enger S, Aas P (2006) Pharmacological therapies against soman-induced seizures in rats 30 min following onset and anticonvulsant impact. Eur J Pharmacol 548:83–89
Myhrer T, Enger S, Aas P (2010) Roles of perirhinal and posterior piriform cortices in control and generation of seizures: a microinfusion study in rats exposed to soman. Neurotoxicology 31:147–153
Pajoumand A, Shadnia S, Rezaie A, Abdi M, Abdollahi M (2004) Benefits of magnesium sulphate in the management of acute human poisoning by organophosphorus insecticides. Hum Exp Toxicol 23:565–569
Papadopoulos V, Baraldi M, Guilarte TR, Knudsen TB, Lacapere JJ, Lindemann P, Norenberg MD, Nutt D, Weizman A, Zhang MR, Gavish M (2006) Translocator protein (18 kDa): new nomenclature for the peripheral-type benzodiazepine receptor based on its structure and molecular function. Trends Pharmacol Sci 27:402–409
Peter JV, Moran JL, Pichamuthu K, Chacko B (2008) Adjuncts and alternatives to oxime therapy in organophosphate poisoning–is there evidence of benefit in human poisoning? A review. Anaesth Intensive Care 36:339–350
Petras JM (1994) Neurology and neuropathology of soman-induced brain injury: an overview. J Exp Anal Behav 61:319–329
Raveh L, Weissman BA, Cohen G, Alkalay D, Rabinovitz I, Sonego H, Brandeis R (2002) Caramiphen and scopolamine prevent soman-induced brain damage and cognitive dysfunction. Neurotoxicology 23:7–17
Raveh L, Brandeis R, Gilat E, Cohen G, Alkalay D, Rabinovitz I, Sonego H, Weissman BA (2003) Anticholinergic and antiglutamatergic agents protect against soman-induced brain damage and cognitive dysfunction. Toxicol Sci 75:108–116
Raveh L, Rabinovitz I, Gilat E, Egoz I, Kapon J, Stavitsky Z, Weissman BA, Brandeis R (2008) Efficacy of antidotal treatment against sarin poisoning: the superiority of benactyzine and caramiphen. Toxicol Appl Pharmacol 227:155–162
Shih T, McDonough JH Jr, Koplovitz I (1999) Anticonvulsants for soman-induced seizure activity. J Biomed Sci 6:86–96
Shih TM, Duniho SM, McDonough JH (2003) Control of nerve agent-induced seizures is critical for neuroprotection and survival. Toxicol Appl Pharmacol 188:69–80
Sibai BM (1990) Magnesium sulphate is the ideal anticonvulsant in preeclampsia-eclampsia. Am J Obstet Gynecol 162:1141–1145
Singh G, Avasthi G, Khurana D, Whig J, Mahajan R (1998) Neurophysiological monitoring of pharmacological manipulation in acute organophosphate (OP) poisoning. The effects of pralidoxime, magnesium sulphate and pancuronium. Electroencephalogr Clin Neurophysiol 107:140–148
Slutsky I, Abumaria N, Wu LJ, Huang C, Zhang L, Li B, Zhao X, Govindarajan A, Zhao MG, Zhuo M, Tonegawa S, Liu G (2010) Enhancement of learning and memory by elevating brain magnesium. Neuron 65:165–177
Tattersall J (2009) Seizure activity post organophosphate exposure. Front Biosci 14:3688–3711
Taylor P (2005) Anticholinesterase agents. In: Hardman JG, Limbird EL, Gilman AG (eds) The pharmacological basis of therapeutics, 10th edn. McGraw-Hill Inc, New York, pp 175–191
Weissman BA, Raveh L (2003) Peripheral benzodiazepine receptors: on mice and human brain imaging. J Neurochem 84:432–437
Wolf G, Keilhoff G, Fischer S, Hass P (1990) Subcutaneously applied magnesium protects reliably against quinolinate-induced N-methyl-D-aspartate (NMDA)-mediated neurodegeneration and convulsions in rats: are there therapeutical implications. Neurosci Lett 117:207–211
Wolf G, Fischer S, Hass P, Abicht K, Keilhoff G (1991) Magnesium sulphate subcutaneously injected protects against kainate-induced convulsions and neurodegeneration: in vivo study on the rat hippocampus. Neuroscience 43:31–34
Young AB, Fagg GE (1990) Excitatory amino acid receptors in the brain: membrane binding and receptor autoradiographic approaches. Trends Pharmacol Sci 11:126–133
Zaret GM (1983) Possible treatment of pre-eclampsia with calcium channel blocking agents. Med Hypotheses 12:303–319
Zimmer LA, Ennis M, Shipley MT (1997) Soman-induced seizures rapidly activate astrocytes and microglia in discrete brain regions. J Comp Neurol 378:482–492
Acknowledgments
The authors wish to thank Dr. Ben-Avi Weissman for his helpful advice and Rita Sahar, G.Yacov, S. Baranes, and Y. Kapon for their excellent technical assistance.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katalan, S., Lazar, S., Brandeis, R. et al. Magnesium sulfate treatment against sarin poisoning: dissociation between overt convulsions and recorded cortical seizure activity. Arch Toxicol 87, 347–360 (2013). https://doi.org/10.1007/s00204-012-0916-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-012-0916-1