Abstract
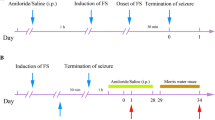
The effects of GM1 monosialoganglioside pretreatment on brain damage resulting from soman-induced seizure activity were examined in this study. Male Sprague-Dawley rats were infused with GM1 via an osmotic minipump connected through a permanent cannula implanted intracerebroventricularly and challenged with soman (83 μg/kg, i.e., 1.25 × LD50) 4 d after initiation of GM1 infusion. Electrocorticographic recordings were monitored via indwelling cortical electrodes. Twenty-seven hours after soman administration, anesthetized rats were euthanized via transcardial perfusion with buffered paraformaldehyde. Brains were processed for hematoxylin and eosin (H&E), cresyl violet (CV), and acetylcholinesterase (AChE) histochemistry, and glial fibrillary acidic protein (GFAP) and microtubule-associated protein 2 (MAP2) immunohistochemistry. All soman-challenged rats not infused with GM1 (n=14) developed status epilepticus (SE). GM1-infused, soman-challenged rats (n=11) showed initial signs of seizures; however, only five developed SE. The remaining six recovered and had no brain damage. In addition, the latter group showed a significantly higher residual AChE reactivity in the basolateral amygdala compared to rats that developed SE. Quantitative image analysis of MAP2-immunostained brain sections from the five GM1-infused rats that developed SE showed a 85.9±14.1% reduction in cross-sectional area of necrosis in the piriform cortex when compared to the somanchallenged rats that were not infused with GM1. This was paralleled by a pronounced decrease in morphological evidence of damage on H&E and CV-stained serial sections. Considerable brain region/treatment group variability was seen with GFAP immunostaining. The present findings demonstrate that GM1 pretreatment interferes with the development of SE and significantly alleviates brain damage resulting from soman-induced seizures.
Similar content being viewed by others
References
Ando S. (1983) Gangliosides in the nervous system.Neurochem. Int. 5, 507–537.
Bakhit C., Armanini M., Bennett G. L., Wong W. L., Hansen S. E. and Taylor R. (1991) Increase in glia-derived nerve growth factor following destruction of hippocampal neurons.Brain Res. 560, 76–83.
Ballough G. P. H., Martin L. J., Cann F. J., Graham J. S., Smith C. D., Kling C. E., et al. (1995) Microtubule-associated protein 2 (MAP2): a sensitive marker of seizure-related brain damage.J. Neurosci. Methods 61, 23–32.
Bernhardt R. and Matus A. (1984) Light and electron microscopic studies of the distribution of microtubule-associated protein 2 in rat brain: a difference between dendritic and axonal cytoskeletons,J. Comp. Neurol. 226, 203–221.
Braitman D. J. and Sparenborg S. (1989) MK-801 protects against seizures induced by the cholinesterase inhibitor soman.Brain Res. Bull. 23, 145–148.
Carolei A., Fieschi C., Bruno R., and Toffano G. (1991) Monosialoganglioside GM1 in cerebral ischemia.Cerebrovasc. Brain Metab. Rev. 3, 134–157.
Carpentier P., Delamanche I. S., Le Bert M., Blanchet G., and Bouchaud C. (1990) Seizurerelated opening of the blood-brain barrier induced by soman: possible correlation with the acute neuropathology observed in poisoned rats.NeuroToxicology 11, 493–508.
Choi D. W. (1987) Ionic dependence of glutamate neurotoxicity.J. Neurosci. 7(2), 369–379.
Choi D. W. (1992) Bench to bedside: the glutamate connection.Science 258, 241–243.
Connor J. A., Wadman W. J., and Hockberger P. E. (1988) Sustained dendritic gradients of Ca2+ induced by excitatory amino acid in CA1 hippocampal neurons.Science 240, 649–653.
Costa E., Armstrong D. M., Guidotti A., Kharlamov A., Kiedrowski L., Manev H., et al. (1994) Gangliosides in the protection against glutamate excitotoxicity, inProgress in Brain Research, vol. 101 (Svennerholm L., Asbury A. K., Reisfeld R. A., Sandhoff D., Suzuki K., Tettamanti G., et al., eds.), Elsevier, New York, pp. 357–373.
Facci L., Leon A., and Skaper S. D. (1990a) Hypoglycemic neurotoxicity in vitro; involvement of excitatory amino acid receptors and attenuation by monosialoganglioside GM1.Neuroscience 37, 709–716.
Facci L., Leon A., and Skaper S. D. (1990b) Excitatory amino acid neurotoxicity in cultured retinal neurons: involvement ofN-methyl-d-aspartate (NMDA) and non-NMDA receptors and effect of ganglioside GM1.J. Neurosci. Res. 27, 202–210.
Favaron M., Manev H., Alho H., et al. (1988) Gangliosides prevent glutamate and kainate neurotoxicity in primary neuronal cultures of neonatal rat cerebellum and cortex.Proc. Natl. Acad. Sci. USA 85, 7351–7355.
Greene J. G. and Greenamyre J. T. (1996) Bioenergetics and glutamate excitotoxicity.Prog. Neurobiol. 48, 613–634.
Hicks R. R., Smith D. H., and McIntosh T. K. (1995) Temporal response and effects of excitatory amino acid antagonism on microtubule-associated protein 2 immunoreactivity following experimental brain injury in rats.Brain Res. 678, 151–160.
Hsu S.-M., Raine L., and Fanger H. (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures.J. Histochem. Cytochem. 29, 577–580.
Hugon J., Vallet J. M., and Dumas M. (1996) Role of glutamate and excitotoxicity in neurologic diseases.Rev. Neurol. 154, 239–248.
Karnovsky M. J. and Roots L. (1964) A “direct-coloring” thiocholine method for cholinesterases.J. Histochem. Cytochem. 12, 219–221.
Kitagawa K., Matsumoto M., Ninobe M., Mikoshiba K., Hata R., Ueda H., et al. (1989) Microtubule-associated protein 2 as a sensitive marker for cerebral ischemic damage-immunohistochemical investigation of dendritic damage.Neuroscience 31, 401–411.
Kraig R. P., Lascola C. D., and Caggiano A. (1995) Glial response to brain ischemia, inNeuroglia (Kettenmann H. and Ransom B. R., eds.), pp. 964–976, Oxford University Press, New York.
Ledeen R. W. (1984) Biology of gangliosides: neuritogenic and neuronotrophic properties.J. Neurosci. Res. 12, 147–159.
Lemercier G., Carpentier P., Sentenac-Roumanou H., and Morelis P. (1983) Histological and histochemical changes in the central nervous system of the rat poisoned by an irreversible anticholinesterase organophosphorus compound.Acta Neuropathol. 61, 123–129.
Lombardi G. and Moroni F. (1992) GM1 ganglioside reduces ischemia-induced excitatory amino acid output: a microdialysis study in the gerbil hippocampus.Neurosci. Lett. 134, 171–174.
Mahadik S. P., Vilim F., Korenovsky A., and Karpiak S. E. (1988) GM1 ganglioside protects nucleus basalis from excitotoxin damage: reduced cortical cholinergic losses and animal mortality.J. Neurosci. Res. 20, 479–483.
Malhotra S. K., Shnitka T. K., and Elbrink J. (1990) Reactive astrocytes: a review.Cytobios 61, 133–160.
Manev H., Costa E., Wroblewski J. T., and Guidotti A. (1990a) Abusive stimulation of excitatory amino acid receptors: a strategy to limit neurotoxicity.FASEB J. 4, 2789–2797.
Manev H., Favaron M., Vicini S., Guidotti A., and Costa E. (1990b) Glutamate-induced neuronal death in primary cultures of cerebellar granule cells: protection by synthetic derivatives of endogenous sphingolipids.J. Pharmacol. Exper. Ther. 252, 419–427.
Manev, H., Guidotti A., and Costa E. (1991) Neuroprotective effects of glycosphingolipids in in-vitro and in-vivo models of neurotoxicity, inProceedings of the 1991 Medical Defense Bioscience Review, pp. 401–405, DTIC Accession #AD B158-588.
Manev H., Guidotti A., and Costa E. (1993) Protection by gangliosides against glutamate excitotoxicity.Adv. Lipid Res. 25, 269–288.
Matus A. (1994) MAP2, inMicrotubules (Hyams J. S. and Lloyd C. W., eds.), Wiley-Liss, New York, NY, pp. 155–166.
Maysinger D., Herrera-Marschitz M., Ungerstedt U., and Cuello A. C. (1990) Acetylcholine release in vivo: effects of chronic treatment with monosialoganglioside GM1.Neuropharmacology 29, 151–159.
McDonough J. H., Jr., and Shih T.-M. (1993) Pharmacological modulation of soman-induced seizures.Neurosci. Biobehav. Rev. 17, 203–215.
McDonough J. H., Jr., McLeod C. G., Jr., and Nipwoda T. (1987) Direct microinjection of soman or VX into the amygdala produces repetitive limbic convulsions and neuropathology.Brain Res. 435, 123–137.
McLeod C. G., Jr., Singer A. W., and Harrington D. G. (1984) Acute neuropathology in soman poisoned rats.NeuroToxicology 5, 53–58.
Montgomery D. L. (1994) Astrocytes: form, function, and roles in disease: Review.Vet. Pathol. 31, 145–167.
O'Callaghan J. P. and Jensen K. F. (1992) Enhanced expression of glial fibrillary acidic protein and the cupric silver degeneration reaction can be used as sensitive and early indicators of neurotoxicity.NeuroToxicology 13, 113–122.
Olney J. W., De Gubareff T., and Labruyere J. (1983) Seizure-related brain damage induced by cholinergic agents.Nature 301, 520–522.
Paxinos G. and Watson C. (1986)The Rat Brain in Stereotaxic Coordinates, 2nd ed., Academic, New York, NY.
Pazdernik T. L., Cross R., Giesler M., Nelson S., Samson F., and McDonough J., Jr. (1985) Delayed effects of soman: brain glucose use and pathology.NeuroToxicology 6, 61–70.
Pechan P. A., Yoshida T., Panahian N., Moskowitz M. A., and Breakefield X. O. (1995) Genetically modified fibroblasts producing NGF protect hippocampal neurons after ischemia in the rat.NeuroReport 6, 669–672.
Petrali J. P., Maxwell D. M., Lenz D. E., and Mills K. R. (1991) Effect of an anticholinesterase compound on the ultrastructure and function of the rat blood-brain barrier: a review and experiment.J. Submicrosc. Cytol. Pathol. 23, 331–338.
Petras J. M. (1981) Soman neurotoxicity.Fundam. Appl. Toxicol. 1, 242.
Phillis J. W. and O'Regan M. H. (1995) GM1 ganglioside inhibits ischemic release of amino acid neurotransmitters from rat cortex.NeuroReport 23, 2010–2016.
Polo A., Kirschner G., Guidotti A., and Costa E. (1994) Brain content of glycosphin-golipids after oral administration of monosialogangliosides GM1 and LIGA20 to rats.Mol. Chem. Neuropathol. 21, 41–53.
Powers M. and Clark G. (1955) An evaluation of cresyl echt violet as a Nissl stain.Stain Technol. 30, 83–92.
Rabin S. J. and Mocchetti I. (1995) GM1 ganglioside activates the high-affinity nerve growth factor receptortrkA.J. Neurochem. 65, 347–354.
Randall R. D. and Thayer S. A. (1992) Glutamate-induced calcium transient triggers delayed calcium overload and neurotoxicity in rat hippocampal neurons.J. Neurosci. 12, 1882–1895.
Saqr H. E., Pearl D. K., and Yates A. J. (1993) A review and predictive models of ganglioside uptake by biological membranes.J. Neurochem. 61, 395–411.
Semkova I., Schilling M., Henrich-Noack P., Rami A., and Krieglstein J. (1996a) Clenbuterol protects mouse cerebral cortex and rat hippocampus from ischemic damage and attenuates glutamate neurotoxicity in cultured hippocampal neurons by induction of NGF.Brain Res. 717, 44–54.
Semkova I., Wolz P., Schilling M., and Krieglstein J. (1996b) Selegiline enhances NGF synthesis and protects central nervous system neurons from excitotoxic and ischemic damage.Eur. J. Pharmacol. 315, 19–30.
Silver B., Weber J., and Fisher M. (1996) Medical therapy for ischemic stroke: review.Clin. Neuropharm. 19, 101–128.
Skaper S. D., Facci L., Milani D., and Leon A. (1989) Monosialoganglioside GM1 protects anoxia-induced neuronal death in vitro.Exp Neurol. 106, 297–305.
Skaper S. D., Facci L., and Leon A. (1990) Gangliosides attenuate the delayed neurotoxicity of aspartic acid in vitro.Neurosci. Lett. 117, 154–159.
Skaper S. D., Leon A., and Facci L. (1991) Ganglioside GM1 prevents death induced by excessive excitatory neurotransmission in cultured hippocampal pyramidal neurons.Neurosci. Lett. 126, 98–101.
Sparenborg S., Brennecke L. H., Jaax N. K., and Braitman D. J. (1992) Dizocilpine (MK-801) arrests status epilepticus and prevents brain damage induced by soman.Neuropharmacology 31, 357–368.
Switzer R. C. (1991) Strategies for assessing neurotoxicity,Neurosci. Biobehav. Rev. 15, 89–93.
Tan W. K. M., Williams C. E., Gunn A. J., Mallard E. C., and Gluckman P. D. (1993) Pretreatment with monosialoganglioside GM1 protects the brain of fetal sheep against hypoxic/ischemic injury without causing systemic compromise.Pediatr. Res. 34, 18–22.
Tan W. K. M., Williams C. E., Mallard E. C., and Gluckman P. D. (1994) Monosialoganglioside GM1 treatment after a hypoxic/ischemic episode reduces the vulnerability of the fetal sheep brain to subsequent injuries.Am. J. Obstet. Gynecol. 170, 663–670.
Toffano G., Benvegnu D., Bonetti A. C., Facci L., Leon A., Orlando P., et al. (1980) Interactions of GM1 ganglioside with crude rat brain neuronal membranes.J. Neurochem. 35, 861–866.
Torgner I. and Kvamme E. (1990) Synthesis of transmitter glutamate and the glial-neuron interrelationship.Mol. Chem. Neuropathol. 12, 11–17.
Tuor U. I., Del Bigio M. R., and Chumas P. D. (1996) Brain damage due to cerebral hypoxia/ischemia in the neonate: pathology and pharmacological modification.Cerebrovasc. Brain Metab. Rev. 8, 159–193.
Watson R. E., Wiegand S. J., Clough R. W., and Hoffman G. E. (1986) Use of cryoprotectant to maintain long-term peptide immunoreactivity and tissue morphology.Peptides 7, 155–159.
Wiche G. (1989) High-Mr microtubule-associated proteins: properties and functions: review article.Biochem. J. 259, 1–12.
Wojcik M., Ulas J., and Oderfeld-Nowak B. (1982) The stimulating effect of ganglioside injections on the recovery of choline acetyltransferase and acetylcholinesterase activities in the hippocampus of the rat after septal lesions.Neuroscience 7, 495–499.
Yoshida K. and Toya S. (1997) Neurotrophic activity in cytokine-activated astrocytes.Keio J. Med. 46, 55–60.
Yu R. K., Goldenring J. R., Kim J. Y. H., and DeLorenzo R. J. (1986) Gangliosides as differential modulators of membrane-bound protein kinase systems, inFidia Research Series: Gangliosides and Neuronal Plasticity, vol. 6 (Tettamanti G., Ledeen R. W., Sandhoff K., Nagai Y., and Toffano G., eds.), pp. 95–104, Liviana, Padova.
Author information
Authors and Affiliations
Additional information
The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as the views of the Department of the Army. In conducting the research described in this article, the investigators adhered to the “Guide for the Care and Use of Laboratory Animals” as adopted and promulgated by the National Institutes of Health publication 96-01.
Rights and permissions
About this article
Cite this article
Ballough, G.P.H., Cann, F.J., Smith, C.D. et al. GM1 monosialoganglioside pretreatment protects against soman-induced seizure-related brain damage. Molecular and Chemical Neuropathology 34, 1–23 (1998). https://doi.org/10.1007/BF02815133
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02815133