Abstract
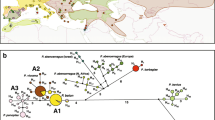
Ophiocordyceps sinensis Berk. is a fungal parasite that parasitizes the larvae of Hepialidae and is endemic to the Qinghai–Tibet Plateau (QTP). The phylogeny and divergence time of O. sinensis and its host insects were analyzed for 137 individuals from 48 O. sinensis populations based on the elongation factor 1 alpha (EF-1α) gene. Lower nucleotide variation, with only 7 and 16 EF-1α haplotypes, was detected in O. sinensis and its host insects, respectively. The isolated and broad distribution patterns coexisted in both O. sinensis and its host insects on the QTP. The divergence time estimates show that O. sinensis and its host insects originated later than 14.33 million years (Myr) and earlier than 23.60 Myr in the Miocene period, and the major differentiation occurred later than 4 Myr. Their origin and differentiation match well with the second and third uplifts of the QTP, respectively. The host insects from the O. sinensis populations distributed around Qinghai Lake are inferred as an ancient and relict species that has survived various geological events of the QTP. It is suitable to estimate the divergence times of both O. sinensis and its host insects from the same individuals using one gene: EF-1α. Our findings of the origin, phylogeny, and evolution of the endemic species also support the epoch of geological events on the QTP.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed in the current study are available from the corresponding author upon reasonable request.
References
An ZS, Kutzbach JE, Prell WL, Porter SC (2001) Evolution of Asian monsoons and phased uplift of the Himalaya-Tibetan plateau since ate Miocene times. Nature 411:62–66. https://doi.org/10.1038/35075035
Bandelt HJ, Forster P, Rohl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Berbeea ML, Taylorb JW (2010) Dating the molecular clock in fungi-how close are we? Fungal Biol Rev 24:1–16. https://doi.org/10.1016/j.fbr.2010.03.001
Blackwell M (2010) Fungal evolution and taxonomy. Biocontrol 55:7–16. https://doi.org/10.1007/s10526-009-9243-8
Bouckaert R, Heled J, Kühnert D et al (2014) Beast 2: a software platform for Bayesian evolutionary analysis. PLoS Comput Biol 10:e1003537. https://doi.org/10.1371/journal.pcbi.1003537
Braby MF, Roger V, Pierce NE (2006) Molecular phylogeny and systematics of the Pieridae (Lepidoptera: Papilionoidea): higher classification and biogeography. Zool J Linn Soc 147:239–275. https://doi.org/10.1111/j.1096-3642.2006.00218.x
Caterino MS, Cho S, Sperling FAH (2000) The current state of insect molecular systematics: a thriving Tower of Babel. Annu Rev of Entomol 45:1–54. https://doi.org/10.1146/annurev.ento.45.1.1
Chen YJ, Zhang YP, Yang YX et al (1999) Genetic diversity and taxonomic implication of Cordyceps sinensis as revealed by RAPD markers. Biochem Genet 37:201–213. https://doi.org/10.1023/a:1018738706123
Chen YQ, Wang N, Qu L et al (2001) Determination of the anamorph of Cordyceps sinensis inferred from the analysis of the ribosomal DNA internal transcribed spacers and 5.8S rDNA. Biochem Syst Ecol 29:597–607. https://doi.org/10.1016/S0305-1978(00)00100-9
Chen YQ, Hu B, Xu F et al (2004) Genetic variation of Cordyceps sinensis, a fruit-body-producing entomopathogenic species from different geographical regions in China. FEMS Microbiol Lett 230:153–158. https://doi.org/10.1016/s0378-1097(03)00889-9
Cheng Z, Geng Y, Liang HH et al (2007) Phylogenetic relationships of host insects of Cordyceps sinensis inferred from mitochondrial cytochrome b sequences. Progress in Nat Sci 17:789–797. https://doi.org/10.1080/10002007088537474
Clouse RM, Sharma PP, Giribet G et al (2013) Elongation factor-1α, a putative single-copy nuclear gene, has divergent sets of paralogs in an arachnid. Mol Phylogenet and Evol 68:471–481. https://doi.org/10.1016/j.ympev.2013.04.025
Dai YD, Wu CK, Wang YB et al (2019) Phylogeographic structures of the host insects of Ophiocordyceps sinensis. Zoology 134:27–37. https://doi.org/10.1016/j.zool.2019.03.003
Dai YD, Wu CK, Yuan F et al (2020) Evolutionary biogeography on Ophiocordyceps sinensis: an indicator of molecular phylogeny to geochronological and ecological exchanges. Geosci Front 11:807–820. https://doi.org/10.1016/j.gsf.2019.09.001
Djernaes MA, Damgaard J (2006) Exon-intron structure, paralogy and sequenced regions of elongation factor-1 alpha in Hexapoda. Arthropod Syst Phylo 64:45–52. https://doi.org/10.4028/www.scientific.net/KEM.592-593.197
Favre A, Päckert M, Pauls SU et al (2015) The role of the uplift of the Qinghai-Tibetan Plateau for the evolution of Tibetan biotas. Biol Rev 90:236–253. https://doi.org/10.1111/brv.12107
Hao JJ, Cheng Z, Liang HH et al (2009) Genetic differentiation and distributing pattern of Cordyceps sinensis in China revealed by rDNA ITS sequences. Chin Tradit Herb Drugs 40:112–116
Hu WG, Zhang Q, Li DY et al (2014) Diversity and community structure of fungi through a permafrost core profile from the Qinghai-Tibet Plateau of China. J Basic Microbiol 54:1331–1341. https://doi.org/10.1002/jobm.201400232
Kinjo N, Zang M (2001) Morphological and phylogenetic studies on Cordyceps sinensis distributed in southwestern China. Mycoscience 42:567–574. https://doi.org/10.1007/BF02460956
Klopfstein S, Ronquist F (2013) Convergent intron gains in hymenopteran elongation factor-1α. Mole Phylogenet Evol 67:266–276. https://doi.org/10.1016/j.ympev.2013.01.015
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Li JJ, Fang XM (1998) Research on the uplift of Qinghai-Tibet Plateau and its environment changes. Chin Sci Bull 43:1569–1574
Li SP, Tsim KWK (2004) The biological and pharmacological properties of Cordyceps sinensis, a traditional Chinese medicine that has broad clinical applications. In: Wachtel-Galor S (ed) Herbal and traditional medicine: molecular aspects of health. CRC Press, New York, pp 657–686
Liang HH, Cheng Z, Yang XL et al (2005) Genetic variation and affinity of Cordyceps sinensis in Qinghai province based on analysis of morphologic characters and inter-simple sequence repeat markers. Chin Tradit Herb Drugs 36:1859–1864
Liang HH, Cheng Z, Yang XL et al (2008) Genetic diversity and structure of Cordyceps sinensis populations from extensive geographical regions in China as revealed by inter-simple sequence repeat markers. J Microbiol 46:549–556. https://doi.org/10.1007/s12275-008-0107-1
Lin CP, Danforth BN (2004) How do insect nuclear and mitochondrial gene substitution patterns differ? Insights from Bayesian analyses of combined datasets. Mol Phylogenet Evol 30:686–702. https://doi.org/10.1016/S1055-7903(03)00241-0
Liu DF, Jiang GF (2005) The application of nuclear genes sequences in insect molecular systematics. Acta Zootaxonomica Sinica 30:484–492. https://doi.org/10.3969/j.issn.1000-0739.2005.03.007
Lo HC, Hsieh C, Lin FY et al (2013) A systematic review of the mysterious caterpillar fungus Ophiocordyceps sinensis in DongChongXiaCao and related bioactive ingredients. J Tradit Complement Med 3:16–32. https://doi.org/10.1016/S2225-4110(16)30164-X
Meyer W, Irinyi L, Hoang MTV et al (2019) Database establishment for the secondary fungal DNA barcode translational elongation factor 1α (TEF1α). Genome 62:160–169. https://doi.org/10.1139/gen-2018-0083
Nikoh N, Fukatsu T (2000) Interkingdom host jumping underground: phylogenetic analysis of entomoparasitic fungi of the genus Cordyceps. Mol Biol Evol 17:629–638. https://doi.org/10.1093/oxfordjournals.molbev.a026341
Pandey A, Mohanty PS, Arya P et al (2010) Genetic diversity among the isolates of Cordyceps sinensis of higher Himalayan meadows of India. Int J Mol Sci 1:242–245
Quan QM, Wang QX, Zhou XL et al (2014a) Comparative phylogenetic relationships and genetic structure of the caterpillar fungus Ophiocordyceps sinensis and its host insects inferred from multiple gene sequences. J Microbiol 52:99–105. https://doi.org/10.1007/s12275-014-3391-y
Quan QM, Chen LL, Wang X et al (2014b) Genetic diversity and distribution patterns of host insects of caterpillar fungus Ophiocordyceps sinensis in the Qinghai-Tibet Plateau. PLoS ONE 9:e92293. https://doi.org/10.1371/journal.pone.0092293
Rambaut A, Drummond AJ (2009) Tracer: MCMC trace analysis tool, version 1.5. http://beast.bio.ed.ac.uk/Tracer. Accessed 5 Feb 2022
Roger AJ, Sandblom O, Doolittle WF et al (1999) An evaluation of elongation factor 1α as a phylogenetic marker for eukaryotes. Mol Biol Evol 16:218–233. https://doi.org/10.1093/oxfordjournals.molbev.a026104
Rozas J, Ferrer-Mata A, Sanchez-DelBarrio JC et al (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34:3299–3302. https://doi.org/10.1093/molbev/msx248
Sato K, Sato M (2017) Multiple ways to prevent transmission of paternal mitochondrial DNA for maternal inheritance in animals. J Biochem 162(4):247–253. https://doi.org/10.1093/jb/mvx052
Shashidhar MG, Giridhar P, Sankar KU et al (2013) Bioactive principles from cordyceps sinensis: a potent food supplement - a review. J Funct Foods 5:1013–1030. https://doi.org/10.1016/j.jff.2013.04.018
Shi YF, Li JJ, Li BY et al (1999) Uplift of the Qinghai-Xizang (Tibetan) Plateau and East Asia environmental change during late Cenozoic. Acta Geogr Sin 54:10–21
Singh R, Negi PS, Ahmed Z (2009) Genetic variability assessment in medicinal caterpillar fungi Cordyceps spp. (Ascomycetes) in Central Himalayas India. Int J Med Mushrooms 11:185–189
Song A, Yang JC, Dong WN et al (2021) The advances in biogeography research on Tibetan Plateau alpine bioregion. J Glaciol Geocryol 43:786–797. https://doi.org/10.7522/j.issn.1000-0240.2021.0020
Spatafora JW, Sung GH, Sung JM et al (2007) Phylogenetic evidence for an animal pathogen origin of ergot and the grass endophytes. Mol Ecol 16:1701–1711. https://doi.org/10.1111/j.1365-294X.2007.03225.x
Sun H (2002) Evolution of Arctic-Tertiary flora in Himalayan-Hengduan Mountains. Acta Bot Yunnanica 24:671–688
Sung GH, Hyweljones NL, Sung JM et al (2007) Phylogenetic classification of Cordyceps and the clavicipitaceous fungi. Stud Mycol 57:5–59. https://doi.org/10.3114/sim.2007.57.01
Sung GH, Poinar GOJ, Spatafora JW (2008) The oldest fossil evidence of animal parasitism by fungi supports a Cretaceous diversification of fungal–arthropod symbioses. Mol Phylogenet Evol 49:495–502. https://doi.org/10.1016/j.ympev.2008.08.028
Thomas JA (2005) Monitoring change in the abundance and distribution of insects using butterflies as indicator groups. Philos Trans R Soc Lond B Biol Sci 360:339–357. https://doi.org/10.1098/rstb.2004.1585
Thomas JA, Morris MG (1994) Patterns, mechanisms and rates of extinction among invertebrates in the United Kingdom. Philos Trans R Soc Lond B Biol Sci 344:47–54. https://doi.org/10.1098/rstb.1994.0050
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Tian LH, Hu B, Zhou H et al (2010) Molecular phylogeny of the entomopathogenic fungi of the genus Cordyceps (Ascomycota: Clavicipitaceae) and its evolutionary implications. J Syst Evol 48:435–444. https://doi.org/10.1111/j.1759-6831.2010.00100.x
Wahlberg N, Leneveu J, Kodandaramaiah U et al (2009) Nymphalid butterflies diversify following near demise at the Cretaceous/Tertiary boundary. Proc R Soc B 276:4295–4302. https://doi.org/10.1098/rspb.2009.1303
Wang GD (1995) Cordyceps species: ecology, cultivation and application. Science and Technology Reference Press, Beijing, p 307
Wang XL, Yao YJ (2011) Host insect species of Ophiocordyceps sinensis: a review. ZooKeys 127:43–49. https://doi.org/10.3897/zookeys.127.802
Wen J, Zhang JQ, Nie ZL et al (2014) Evolutionary diversifications of plants on the Qinghai-Tibetan Plateau. Front Genet 45:4. https://doi.org/10.3389/fgene.2014.00004
Xia EH, Yang DR, Jiang JJ et al (2017) The caterpillar fungus, Ophiocordyceps sinensis, genome provides insights into highland adaptation of fungal pathogenicity. Sci Rep 7:1806. https://doi.org/10.1038/s41598-017-01869-z
Xiao W, Yang JL, Ping Z et al (2009) Non-support of species complex hypothesis of Cordyceps sinensis by targeted rDNA-ITS sequence analysis. Mycosystema 28:724–730. https://doi.org/10.13346/j.mycosystema.2009.05.028
Xu B, Luo D, Li ZM et al (2019) Evolutionary radiations of cushion plants on the Qinghai-Tibet Plateau: insights from molecular phylogenetic analysis of two subgenera of Arenaria and Thylacospermum (Caryophyllaceae). Taxon 68:1003–1020. https://doi.org/10.1002/tax.12127
Xu ZX, Li S, Chen LL et al (2020a) Effects of fungus–host associations on nucleoside differences among Ophiocordyceps sinensis populations on the Qinghai-Tibet Plateau of China. Arch Microbiol 202:2323–2328. https://doi.org/10.1007/s00203-020-01919-7
Xu ZX, Zhu YG, Xuan LY et al (2020b) Haplotype diversity of NADPH-cytochrome P450 reductase gene of Ophiocordyceps sinensis and the effect on fungal infection in host insects. Microorganisms 8:968. https://doi.org/10.3390/microorganisms8070968
Yang DR, Li CD, Shu C et al (1996) Studies on the Chinese species of the genus Hepialus and their geographical distribution. Acta Entomol Sin 39:413–422. https://doi.org/10.16380/j.kcxb.1996.04.012
Yao YJ (2014) Conservation and rational use of the natural resources of Cordyceps sinensis. Science News 15:28–29
Yuan BY, Chen KZ, Bowler JM et al (1990) The formation and evolution of the Qinghai Lake. Quaternary Sci 3:233–243
Yuan SH, Liu YJ, Ge XH et al (2008) Uplift period of the northern margin of the Qinghai-Tibet Plateau: evidences from the Altyn Mountains and Qaidam Basin. Acta Petrologica ET Mineralogica 27:413–421
Zhang YW, Chen YJ, Shen FR et al (1999) Study of genetic divergence in Cordyceps sinensis and C. crassispora from northwest of Yunnan by using RAPD. Mycosystema 18:176–183
Zhang YJ, Xu LL, Zhang S et al (2009) Genetic diversity of Ophiocordyceps sinensis, a medicinal fungus endemic to the Tibetan Plateau: implications for its evolution and conservation. BMC Ecol Evol 9:290. https://doi.org/10.1186/1471-2148-9-290
Zhang YJ, Li EW, Wang CS et al (2012) Ophiocordyceps sinensis, the flagship fungus of China, terminology, life strategy and ecology. Mycology 3:2–10. https://doi.org/10.1080/21501203.2011.654354
Zhang YJ, Zhang S, Li YL et al (2014) Phylogeography and evolution of a fungal-insect association on the Tibetan Plateau. Mol Ecol 23:5337–5355. https://doi.org/10.1111/mec.12940
Zhang P, Cui SH, Liu B et al (2018) Discriminatory power evaluation of nuclear ribosomal rna barcoding sequences through Ophiocordyceps sinensis related samples. Front Microbiol 9:2498. https://doi.org/10.3389/fmicb.2018.02498
Zhang M, Gao ZM, Yin J et al (2019) Complete mitochondrial genome of two Thitarodes species (Lepidoptera, Hepialidae), the host moths of Ophiocordyceps sinensis and phylogenetic implications. Int J Biol Macromol 140:794–807. https://doi.org/10.1016/j.ijbiomac.2019.08.182
Zhu JS, Halpern GM, Jones K (1998) The scientific rediscovery of an ancient Chinese herbal medicine: Cordyceps sinensis Part I. J Altern Complement Med 4:289–303. https://doi.org/10.1089/acm.1998.4.3-289
Zhu ZX, Xie F, Zhang N (2011) Genetic diversity of Ophiocordyceps sinensis in Gansu Province based on inter-simple sequence repeat (ISSR) analyses. Mycosystema 30:501–507
Funding
This work was supported by the National Natural Science Foundation of China (81673526).
Author information
Authors and Affiliations
Contributions
ZC conceived this study. SL, ZX and LC carried out the experimental work and data analysis. YZ and ZC guided the experiments. SL, ZX and WW drafted the manuscript. ZC reviewed and edited the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or nonfinancial interests to disclose. The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
203_2023_3444_MOESM1_ESM.png
Supplementary file1 Time-calibrated phylogeny of O. sinensis and its host insects using the EF-1α gene sequences based on P. coccophagus. The dataset of the EF-1α gene sequences included the seven haplotypes of O. sinensis, the 16 haplotypes of the host insects, 30 species of the genus Ophiocordyceps, and 49 species of the family Nymphalidae (Table S4). P. coccophagus (102 Myr; 99–105 Myr) was used as the crown calibration point (Sung et al., 2008) (PNG 335 KB)
203_2023_3444_MOESM2_ESM.png
Supplementary file2 Time-calibrated phylogeny of O. sinensis and its host insects using the EF-1α gene sequences based on Nymphalidae. The dataset of the EF-1α gene sequences included the seven haplotypes of O. sinensis, the 16 haplotypes of the host insects, 30 species of the genus Ophiocordyceps, and 49 species of the family Nymphalidae (Table S4). Nymphalidae (90 Myr; 80–110 Myr) was used as the crown calibration point (Wahlberg et al., 2009) (PNG 324 KB)
203_2023_3444_MOESM3_ESM.png
Supplementary file3 Time-calibrated phylogeny of O. sinensis and its host insects using the EF-1α gene sequences based on four fossil evidence calibration points, i.e., (A) bird—mammal (300 Myr; standard deviation of 30 Myr); (B) plant—opisthokonts (1700 Myr; standard deviation of 300 Myr); (C) fly—mosquito (235 Myr; standard deviation of 24 Myr); (D) Arabidopsis–rice (200 Myr; 100–200 Myr) (Berbeea and Taylorb, 2010). The EF-1α gene sequences dataset of 42 samples, included the 7 haplotypes of O. sinensis, the 16 haplotypes of the host insects, and the other 23 taxa that vary throughout plants, animals, and fungi, and Plasmodium falciparum (GenBank accession No. X60488.1) was selected as an outgroup (Table S5) (PNG 114 KB)
203_2023_3444_MOESM4_ESM.png
Supplementary file4 Time-calibrated phylogeny of O. sinensis using the nrDNA ITS gene sequences based on P. coccophagus. The dataset of fungal nrDNA ITS gene sequences included one haplotype of O. sinensis, 23 species of genus Ophiocordyceps (Table S6). P. coccophagus (102 Myr; 99–105 Myr) was used as the crown calibration point (Sung et al., 2008) (PNG 70 KB)
203_2023_3444_MOESM5_ESM.png
Supplementary file5 Time-calibrated phylogeny of the host insects using the mitochondrial COI gene sequences based on Nymphalidae. The dataset of mitochondrial COI gene sequences included 38 haplotypes of the host insects and 63 species of the family Nymphalidae (Table S7). Nymphalidae (90 Myr; 80–110 Myr) was used as the crown calibration point (Wahlberg et al., 2009) (PNG 88 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, S., Zhu, Y., Xu, Z. et al. The phylogeny and divergence time of Ophiocordyceps sinensis and its host insects based on elongation factor 1 alpha. Arch Microbiol 205, 98 (2023). https://doi.org/10.1007/s00203-023-03444-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03444-9