Abstract
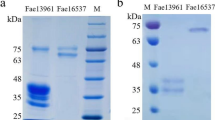
Endophytic bacteria inhabit all or part of their life cycle within the tissues of healthy plants, without causing any apparent symptoms of disease. They are treasure trove of several hydrolytic enzymes with distinct characteristics. Esterase is one of such enzymes and this study aims to characterize esterase produced by endophytic actinobacteria Cellulomonas fimi DB19 isolated from Zanthoxylum armatum with its capacity to degrade diesel oil. The enzyme was purified with purification fold 8.22 and specific activity 124.72 U/mg with 16.43% recovery. The purified enzyme showed a single protein band on SDS-PAGE having molecular mass of approximately 39 kDa. The Km and Vmax value for p-nitrophenyl acetate were 2.23 mM and 22.04 U/mL, respectively. The enzyme was stable in the pH range 6–9 with its optimal activity at pH 8.0. The enzyme was stable at 40 °C and retained more than 80% activity after incubation for two h. The enzyme activity was positively influenced in the presence of Na+, Ba2+, Ca2+, and negatively by Mn2+, and Mg2+. The EDTA and PMSF inhibited the enzyme activity and retained its activity in the presence of SDS, H2O2, β-mercaptoethanol, and organic solvents. Application of the isolate in degradation of diesel showed that its growth and degradation capacity enhanced in media supplemented with 0.2–4% of diesel oil with maximum at 3% of diesel oil. Furthermore, esterase activity was greater in media containing diesel than control which is suggesting the plausible role of esterase produced by Cellulomonas fimi DB19 in the degradation of diesel oil.




Similar content being viewed by others
Availability of data and materials
All data are included in the manuscript.
References
Abdalla MA, Matasyoh JC (2014) Endophytes as producers of peptides: an overview about the recently discovered peptides from endophytic microbes. Nat Prod Bioprospect 4(5):257–270. https://doi.org/10.1007/s13659-014-0038-y
Ali N, Dashti N, Khanafer M, Awadhi HA, Radwan S (2020) Bioremediation of soils saturated with spilled crude oil. Sci Rep 10:1116. https://doi.org/10.1038/s41598-019-57224-x
Asemoloye MD, Tosi S, Daccò C, Wang X, Xu S, Marchisio MA, Gao W, Jonathan SG, Pecoraro L (2020) Hydrocarbon degradation and enzyme activities of Aspergillus oryzae and Mucor irregularis isolated from Nigerian crude oil-polluted sites. Microorganisms 8(12):1912. https://doi.org/10.3390/microorganisms8121912
Ateslier ZBB, Metin K (2006) Production and partial characterization of novel thermostable esterase from a thermophilic Bacillus sp. Enzyme Microb Technol 38(5):628–635. https://doi.org/10.1016/j.enzmictec.2005.07.015
Barman D, Dkhar MS (2020) Seasonal variation influence endophytic actinobacterial communities of medicinal plants from tropical deciduous forest of Meghalaya and characterization of their plant growth-promoting potentials. Curr Microbiol 77(8):1689–1698. https://doi.org/10.1007/s00284-020-01988-3
Bhattacharjee K, Banerjee S, Joshi SR (2012) Diversity of Streptomyces spp. in Eastern Himalayan region-computational RNomics approach to phylogeny. Bioinformation 8(12):548–554. https://doi.org/10.6026/97320630008548
Bhattacharjee K, Chrungoo NK, Joshi SR (2021) Cryopreservation design for bacterial cell: a non-conventional gizmatic approach. Proc Natl Acad Sci India Sect B Biol Sci 91:811–820. https://doi.org/10.1007/s40011-021-01266-7
Brault G, Shareck F, Hurtubise Y, LepineDoucet FN (2012) Isolation and characterization of EstC, a new cold-active esterase from Streptomyces coelicolor A3(2). PLoS ONE 7(3):e32041. https://doi.org/10.1371/journal.pone.0032041
Chandra P, Enespa SR, Arora PK (2020) Microbial lipases and their industrial applications: a comprehensive review. Microb Cell Factories 19:169. https://doi.org/10.1186/s12934-020-01428-8
EI-Sharkawy TA, Huisingh D (1971) Electrophoretic analysis of esterases and other soluble proteins from representatives of phytopathogenic bacterial genera. J Gen Microbiol 68:149–154. https://doi.org/10.1099/00221287-68-2-149
Ek-Ramos MJ, Gomez-Flores R, Orozco-Flores AA, Rodríguez-Padilla C, González-Ochoa G, Tamez-Guerra P (2019) Bioactive products from plant-endophytic gram-positive bacteria. Front Microbiol 10:463. https://doi.org/10.3389/fmicb.2019.00463
Essoussi I, Ghodhbane-Gtari F, Amairi H, Sghaier H, Jaouani A, Brusetti L, Daffonchio D, Boudabous A, Gtari M (2010) Esterase as an enzymatic signature of Geodermatophilaceae adaptability to Sahara desert stones and monuments. J Appl Microbiol 108(5):1723–1732. https://doi.org/10.1111/j.1365-2672.2009.04580.x
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39(4):783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Frankowski H, Gu YH, Heo JH, Milner R, del Zoppo GJ (2012) Use of gel zymography to examine matrix metalloproteinase (gelatinase) expression in brain tissue or in primary glial cultures. Methods Mol Biol 814:221–233. https://doi.org/10.1007/978-1-61779-452-0_15
Holt JG, Krieg NR, Staley JT, Williams ST (1994) Bergey’s manual of determinative bacteriology, 9th edn. Lippincott Williams and Wilkins, Baltimore
Hong S, Lee C, Jang SH (2012) Purification and properties of an extracellular esterase from a cold-adapted Pseudomonas mandelii. Biotechnol Lett 34(6):1051–1055. https://doi.org/10.1007/s10529-012-0866-y
Iqbal A, Arshad M, Karthikeyan R, Gentry TJ, Rashid J, Ahmed I, Schwab AP (2019) Diesel degrading bacterial endophytes with plant growth promoting potential isolated from a petroleum storage facility. 3 Biotech 9(1):35. https://doi.org/10.1007/s13205-018-1561-z
Jaouani A, Neifar A, Hamza A, Chaabouni S, Martinez MJ, Gtari M (2012) Purification and characterization of a highly thermostable esterase from the actinobacterium Geodermatophilus obscurus strain G20. J Basic Microbiol 52:653–660. https://doi.org/10.1002/jobm.201100428
Jin P, Pei X, Du P, Yin X, Xiong X, Wu H, Zhou X, Wang Q (2012) Overexpression and characterization of a new organic solvent-tolerant esterase derived from soil metagenomic DNA. Bioresour Technol 116:234–240. https://doi.org/10.1016/j.biortech.2011.10.087
Kademi A, Ait-Abdelkader N, Fakhreddine L, Baratti J (2000) Purification and characterization of a thermostable esterase from the moderate thermophilic bacterium Bacillus circulans. Appl Microbiol Biotechnol 54:173–179. https://doi.org/10.1007/s002530000353
Kadri T, Rouissi T, Magdouli S, Brar SK, Hegde K, Khiari Z, Daghrir R, Lauzon JM (2018) Production and characterization of novel hydrocarbon degrading enzymes from Alcanivorax borkumensis. Int J Biol Macromol 112:230–240. https://doi.org/10.1016/j.ijbiomac.2018.01.177
Kaiser P, Raina C, Parshad R, Johri S, Verma V, Andrabi KI, Qazi GN (2006) A novel esterase from Bacillus subtilis (RRL 1789): purification and characterization of the enzyme. Protein Expr Purif 45:262–268. https://doi.org/10.1016/j.pep.2005.08.030
Kang CH, Oh KH, Lee MH, Oh TK, Kim BH, Yoon JH (2011) A novel family VII esterase with industrial potential from compost metagenomic library. Microb Cell Factories 10:41. https://doi.org/10.1186/1475-2859-10-41
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16(2):111–120. https://doi.org/10.1007/BF01731581
Kumar L, Singh B, Adhikari DK, Mukherjee J, Ghosh D (2012) A thermoalkaliphilic halotolerant esterase from Rhodococcus sp LKE-028 (MTCC 5562): enzyme purification and characterization. Process Biochem 47(6):983–991. https://doi.org/10.1016/j.procbio.2012.03.020
Kyselkova M, Kopecky J, Felfoldi T, Cermak L, Omelka M, Grundmann GL, Moenne-Loccoz Y, Sagova-Mareckova M (2008) Development of a 16S rRNA gene-based prototype microarray for the detection of selected actinomycetes genera. Antonie Van Leeuwenhoek 94(3):439–453. https://doi.org/10.1007/s10482-008-9261-z
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Lee CW, Kwon S, Park SH, Kim BY, Yoo W, Ryu BH, Kim HW, Shin SC, Kim S, Park H, Kim TD, Lee JH (2017) Crystal structure and functional characterization of an esterase (EaEST) from Exiguobacterium antarcticum. PLoS ONE 12(1):e0169540. https://doi.org/10.1371/journal.pone.0169540
Li XJ, Zheng RC, Wu ZM, Ding X, Zheng YG (2014) Thermophilic esterase from Thermomyces lanuginosus: molecular cloning, functional expression and biochemical characterization. Protein Expr Purif 101:1–7. https://doi.org/10.1016/j.pep.2014.05.006
Liang H, Xing Y, Chen J, Zhang D, Guo S, Wang C (2012) Antimicrobial activities of endophytic fungi isolated from Ophiopogon japonicus (Liliaceae). BMC Complement Altern Med 12:238. https://doi.org/10.1186/1472-6882-12-238
Liu Y, Xu H, Yan Q, Yang S, Duan X, Jiang Z (2013) Biochemical characterization of a first fungal esterase from Rhizomucor miehei showing high efficiency of ester synthesis. PLoS ONE 8(10):e77856. https://doi.org/10.1371/journal.pone.0077856
Lowry OH, Rosebrough NJ, Farr A, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Lumactud R, Shen SY, Lau M, Fulthorpe R (2016) Bacterial endophytes isolated from plants in natural oil seep soils with chronic hydrocarbon contamination. Front Microbiol 7:755. https://doi.org/10.3389/fmicb.2016.00755
Luo Q, Shen XR, Zhang JG, Fan ZQ, He Y (2012) Isolation, identification and biodegradation ability of diesel oil degrading Pseudomonas sp. strain C7 from bilge water. Afr J Microbiol Res 6:1033–1040. https://doi.org/10.5897/AJMR11.1301
Lv XY, Guo LZ, Song L, Fu Q, Zhao K, Li AX, Luo XL, Lu WD (2010) Purification and characterization of a novel extracellular carboxylesterase from the moderately halophilic bacterium Thalassobacillus sp. strain DF-E4. Ann Microbiol 61:281–290. https://doi.org/10.1007/s13213-010-0135-z
Marchut-Mikolajczyk O, Drożdżyński P, Pietrzyk D, Antczak T (2018) Biosurfactant production and hydrocarbon degradation activity of endophytic bacteria isolated from Chelidonium majus L. Microb Cell Factories 17:171. https://doi.org/10.1186/s12934-018-1017-5
Marchut-Mikolajczyk O, Drożdżyński P, Struszczyk-Świta K (2020) Biodegradation of slop oil by endophytic Bacillus cereus EN18 coupled with lipase from Rhizomucor miehei (Palatase®). Chemosphere 250:126203. https://doi.org/10.1016/j.chemosphere.2020.126203
Moliterni E, Jimenez-Tusset RG, Villar Rayo M, Rodriguez L, Fernandez FJ, Villasenor J (2012) Kinetics of biodegradation of diesel fuel by enriched microbial consortia from polluted soils. Int J Environ Sci Technol 9:749–758. https://doi.org/10.1007/s13762-012-0071-5
Nxumalo CI, Ngidi LS, Shandu JSE, Maliehe TS (2020) Isolation of endophytic bacteria from the leaves of Anredera cordifolia CIX1 for metabolites and their biological activities. BMC Complement Med Ther 20(1):300. https://doi.org/10.1186/s12906-020-03095-z
Palanisamy N, Ramya J, Kumar S, Vasanthi N, Chandran P, Khan S (2014) Diesel biodegradation capacities of indigenous bacterial species isolated from diesel contaminated soil. J Environ Health Sci Eng 12(1):142. https://doi.org/10.1186/s40201-014-0142-2
Panda T, Gowrishankar BS (2005) Production and applications of esterases. Appl Microbiol Biotechnol 67:160–169. https://doi.org/10.1007/s00253-004-1840-y
Pawlik M, Cania B, Thijs S, Vangronsveld J, Piotrowska-Seget Z (2017) Hydrocarbon degradation potential and plant growth-promoting activity of culturable endophytic bacteria of Lotus corniculatus and Oenothera biennis from a long-term polluted site. Environ Sci Pollut Res 24:19640–19652. https://doi.org/10.1007/s11356-017-9496-1
Pereira MR, Mercaldi GF, Maester TC, Balan A, de Macedo Lemos EG (2015) Est16, a new esterase isolated from a metagenomic library of a microbial consortium specializing in diesel oil degradation. PLoS ONE 10(7):e0133723. https://doi.org/10.1371/journal.pone.0133723
Phuyal N, Jha PK, Raturi PP, Gurung S, Rajbhandary S (2019) Essential oil composition of Zanthoxylum armatum leaves as a function of growing conditions. Int J Food Prop 22:1873–1885. https://doi.org/10.1080/10942912.2019.1687517
Plumer DT (1978) An introduction to practical biochemistry. McGraw Hill, London
Qin S, Li J, Chen HH, Zhao GZ, Zhu WY, Jiang CL, Xu LH, Li WJ (2009) Isolation, diversity, and antimicrobial activity of rare actinobacteria from medicinal plants of tropical rain forests in Xishuangbanna, China. Appl Environ Microbiol 75(19):6176–6186. https://doi.org/10.1128/AEM.01034-09
Rao L, Xue Y, Zheng Y, Lu JR, Ma Y (2013) A novel alkaliphilic Bacillus esterase belongs to the 13th bacterial lipolytic enzyme family. PLoS ONE 8(4):e60645. https://doi.org/10.1371/journal.pone.0060645
Robinson PK (2015) Enzymes: principles and biotechnological applications. Essays Biochem 59:1–41. https://doi.org/10.1042/bse0590001
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Samaga PV, Rai VR (2016) Diversity and bioactive potential of endophytic fungi from Nothapodytes foetida, Hypericum mysorense and Hypericum japonicum collected from western ghats of India. Ann Microbiol 66:229–244. https://doi.org/10.1007/s13213-015-1099-9
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sana B, Ghosh D, Saha M, Mukherjee J (2007) Purification and characterization of extremely dimethylosulfoxide tolerant esterase from salt-tolerant Bacillus species isolated from the marine environment of the Sundarbans. Process Biochem 42(12):1571–1578. https://doi.org/10.1016/j.procbio.2007.05.026
Sauvetre A, May R, Harpaintnera R, Poschenriederc C, Schröder P (2018) Metabolism of carbamazepine in plant roots and endophytic rhizobacteria isolated from Phragmites australis. J Hazard Mater 342:85–95. https://doi.org/10.1016/j.jhazmat.2017.08.006
Schütte M, Fetzner S (2007) EstA from Arthrobacter nitroguajacolicus Rü61a, a thermo-and solvent-tolerant carboxylesterase related to class C beta-lactamases. Curr Microbiol 54:230–236. https://doi.org/10.1007/s00284-006-0438-2
Sharma A, Sharma T, Sharma T, Sharma S, Kanwar SS (2019) Role of microbial hydrolases in bioremediation. In: Kumar A, Sharma S (eds) Microbes and enzymes in soil health and bioremediation. Springer, Singapore, pp 149–164
Shirling EB, Gottlieb D (1966) Methods for characterization of Streptomyces species. Int J Syst Evol Microbiol 16:313–340. https://doi.org/10.1099/00207713-16-3-313
Singh M, Kumar A, Singh R, Pandey KD (2017) Endophytic bacteria: a new source of bioactive compounds. 3 Biotech 7(5):315. https://doi.org/10.1007/s13205-017-0942-z
Strobel G, Daisy B, Castillo U, Harper J (2004) Natural products from endophytic microorganisms. J Nat Prod 67:257–268. https://doi.org/10.1021/np030397v
Takeda Y, Aono R, Doukyu N (2006) Purification, characterization, and molecular cloning of organic-solvent-tolerant cholesterol esterase from cyclohexane-tolerant Burkholderia cepacia strain ST-200. Extremophiles 10:269–277. https://doi.org/10.1007/s00792-005-0494-8
Tan RX, Zou WX (2001) Endophytes: a rich source of functional metabolites. Nat Prod Rep 18:448–459. https://doi.org/10.1039/b100918o
Xin L, Ying YH (2013) Purification and characterization of an extracellular esterase with organic solvent tolerance from a halotolerant isolate, Salimicrobium sp. LY19. BMC Biotechnol 13:108. https://doi.org/10.1186/1472-6750-13-108
Xue J, Wu Y, Liu Z, Li M, Sun X, Wang H, Liu B (2017) Characteristic assessment of diesel-degrading bacteria immobilized on natural organic carriers in marine environment: the degradation activity and nutrient. Sci Rep 7(1):8635. https://doi.org/10.1038/s41598-017-08832-y
Acknowledgements
The authors are highly thankful to the State Biotech Hub (SBTHub), North-Eastern Hill University, Shillong for providing the required instrumentation facilities. DB is also thankful to the Department of Science and Technology, Government of India for providing financial assistance in the form of fellowship (INSPIRE Fellow).
Funding
The authors have no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Contributions
Both the authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by DB. The first draft of the manuscript was written by DB, and manuscript was reviewed and edited by both the authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Barman, D., Dkhar, M.S. Characterization and purification of esterase from Cellulomonas fimi DB19 isolated from Zanthoxylum armatum with its possible role in diesel biodegradation. Arch Microbiol 204, 580 (2022). https://doi.org/10.1007/s00203-022-03210-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03210-3