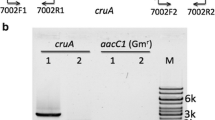
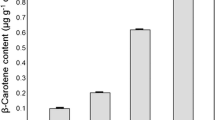
Abstract
Gloeobacter violaceus is a cyanobacterium isolated from other groups by lack of thylakoids and unique structural features of its photosynthetic protein complexes. Carotenoid biosynthesis has been investigated with respect to the carotenoids formed and the genes and enzymes involved. Carotenoid analysis identified ß-carotene as major carotenoid and echinenone as a minor component. This composition is quite unique and the cellular amounts are up to 10-fold lower than in other unicellular cyanobacteria. Carotenoid biosynthesis is up-regulated in a light-dependent manner. This enhanced biosynthesis partially compensates for photooxidation especially of ß-carotene. The sequenced genome of G. violaceus was analyzed and several gene candidates homologous to carotenogenic genes from other organisms obtained. Functional expression of all candidates and complementation in Escherichia coli led to the identification of all genes involved in the biosynthesis of the G. violaceus carotenoids with the exception of the lycopene cyclase gene. An additional diketolase gene was found that functioned in E. coli but is silent in G. violaceus cells. The biggest difference from all other cyanobacteria is the existence of a single bacterial-type 4-step desaturase instead of the poly cis cyanobacterial desaturation pathway catalyzed by two cyanobacterial-type desaturases and an isomerase. The genes for these three enzymes are absent in G. violaceus.




Similar content being viewed by others
References
Albrecht M, Steiger S, Sandmann G (2001) Expression of a ketolase gene mediates the synthesis of canthaxanthin in Synechococcus leading to tolerance against photoinhibition, pigment degradation and UV-B sensitivity of photosynthesis. Photochem Photobiol 73:551–555
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipmann DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Borovkov AY, Rivkin MI (1997) XcmI-Containing vector for direct cloning of PCR products. Biotechniques 22:812–814
Bramley PM, Sandmann G (1985) In vitro and in vivo biosynthesis of xanthophylls by the cyanobacterium Aphanocapsa. Phytochem 24:2919–2922
Breitenbach J, Vioque A, Sandmann G (2001a) Gene sll0033 from Synechocystis 6803 encodes a carotene isomerase involved in the biosynthesis of all-E lycopene. Z Naturforsch 56C:915–917
Breitenbach J, Braun G, Steiger S, Sandmann G (2001b) Chromatographic performance on a C30-bonded stationary phase of mono hydroxycarotenoids with variable chain length or degree of desaturation and of lycopene isomers synthesized by different carotene desaturases. J Chromatogr A 936:59–69
Fernández-Gonzalez B, Sandmann G, Vioque A (1997) A new type of asymmetrically acting ß-carotene ketolase is required for the synthesis of echinenone in the cyanobacterium Synechocystis sp PCC 6803. J Biol Chem 272:9728–9733
Fernández-Gonzalez B, Martínez-Férez IM, Vioque A (1998) Characterization of two carotenoid gene promoters in the cyanobacterium Synechocystis sp PCC6803. Biochim Biophys Acta 1443:343–351
Frank HA, Cogdell RJ (1996) Carotenoids in photosynthesis. Photochem Photobiol 63:257–264
Frigaard N, Bryant DA (2004) Seeing green bacteria in a new light: genomics-enabled studies of the photosynthetic apparatus in green sulfur bacteria and filamentous anoxygenic phototrophic bacteria. Arch Microbiol 182:265–276
Frigaard N, Maresca J, Yunker C, Jones A, Byrant DA (2004) Genetic manipulation of carotenoid biosynthesis in the green sulfur bacterium Chlorobium tepidum. J Bacteriol 8:5210–5220
Gantt E (1994) Supramolecular membrane organisation. In: Bryant DA (ed) Advances in photosynthesis. Kluwer, Dordrecht, pp 119–138
Goodwin TW (1980) The biochemistry of the carotenoids, 2nd edn. Chapman & Hall, London
Guglielmi G, Cohen-Bazire G, Bryant DA (1981) The structure of Gloeobacter violaceus and its phycobilisomes. Arch Microbiol 129:181–189
Hertzberg S, Liaaen-Jensen S, Siegelman HW (1971) The carotenoids of blue-green algae. Phytochem 10:3121–3127
Honda D, Yokota D, Sugiyama J (1999) Detection of seven major evolutionary lineages in cyanobacteria based on the 16S rRNA gene sequence analysis with new sequences of five marine Synechococcus strains. J Mol Evol 48:723–739
Inoue H, Tsuchiya T, Satoh S, Miyashita H, Kaneko T, Tabata S, Tanaka A, Mimuro M (2004) Unique constitution of photosystem I with a novel subunit in the cyanobacterium Gloeobacter violaceus PCC 7421. FEBS Lett 578:275–279
Jöstingmeyer P, Koenig F (1998) Gloeobacter violaceus - investigation of carotenoids and carotenoid associated proteins. In: Garab G (ed) Photosynthesis: mechanisms and effects, vol I. Kluwer, Dordrecht, pp 229–232
Krubasik P, Sandmann G (2000) A carotenogenic gene cluster from Brevibacterium linens with novel lycopene cyclase genes involved in the synthesis of aromatic carotenoids. Mol Gen Genet 263:423–432
Linden H, Sandmann G, Chamovitz D, Hirschberg J, Böger P (1990) Biochemcial characterization of Synechococcus mutants selected against the bleaching herbicide norflurazon. Pestic Biochem Physiol 36:46–51
Masamoto K, Furukawa KI (1997) Accumulation of zeaxanthin in cells of the cyanobacterium Synechococcus sp. strain PCC 7942 grown under high irradiance. J Plant Physiol 151:257–261
Masamoto K, Misawa N, Kaneko T, Kikuno R, Toh H (1998) ß-Carotene hydroxylase gene from the cyanobacterium Synechocystis sp. strain PCC6803. Plant Cell Physiol 39:560–564
Masamoto K, Wada H, Kaneko T, Takaichi S (2001) Identification of a gene required for cis-to-trans carotene isomerization in carotenogenesis of the cyanobacterium Synechocystis sp PCC 6803. Plant Cell Physiol 42:1398–1402
Misawa N, Satomi Y, Kondo K, Yokoyama A, Kajiwara S, Saito T, Ohtani T, Miki W (1995) Structure and functional analysis of a marine bacterial carotenoid biosynthesis gene cluster and astaxanthin biosynthetic pathway proposed at the gene level. J Bacteriol 177:6575–6584
Nakamura Y, Kaneko T, Sato S, Mimuro M, Miyashita H, Tsuchiya T, Sasamoto S, Watanabe A, Kawashima K, Kishida Y, Kiyokawa C, Kohara M, Matsumoto M, Matsuno A, Nakazaki N, Shimpo S, Takeuchi C, Yamada M, Tabata S (2003) Complete genome structure of Gloeobacter violaceus PCC 7421, a cyanobacterium that lacks thylakoids. DNA Res 10:137–145
Nelissen B, Van de Peer Y, Wilmotte A, De Wachter R (1995) An early origin of plastids within the cyanobacterial divergence is suggested by evolutionary trees based on complete 16S rRNA sequences. Mol Biol Evol 12:1166–1173
Rippka R, Waterbury JB, Cohen-Bazire G (1974) A cyanobacterium which lacks thylakoids. Arch Microbiol 100:419–436
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 11:1–61
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning, a laboratory manual, 2nd edn. Cold Spring Harbour Laboratory Press, New York
Sandmann G (2001) Carotenoid biosynthesis and biotechnological application. Arch Biochem Biophys 385:4–12
Sandmann G (2002) Molecular evolution of carotenoid biosynthesis from bacteria to plants. Physiol Plantarum 116:431–440
Sandmann G, Fraser PD (1993) Differential inhibition of phytoene desaturase from diverse origins and analysis of resistant cyanobacterial mutants. Z Naturforsch 48c:307–311
Schumann G, Nürnberger H, Sandmann G, Krügel H (1996) Activation and analysis of cryptic crt genes for carotenoid biosynthesis from Streptomyces griseus. Mol Gen Genet 252:658–666
Selstam E, Campbell D (1996) Membrane lipid composition of the unusual cyanobacterium Gloeobacter violaceus sp PCC 7421, which lacks sulfoquinovosyl diacylglycerol. Arch Microbiol 166:132–135
Simkin AJ, Zhu C, Kuntz M, Sandmann G (2003) Light-dark regulation of carotenoid biosynthesis in pepper (Capsicum annuum) leaves. J Plant Physiol 160:439–443
Steiger S, Sandmann G (2004) Cloning of two carotenoid ketolase genes from Nostoc punctiforme for the heterologous production of canthaxanthin and astaxanthin. Biotechnol Lett 26:813–817
Steiger S, Schäfer L, Sandmann G (1999) High-light upregulation of carotenoids and their antioxidative properties in the cyanobacterium Synechocystis PCC 6803. J Photochem Photobiol B 521:14–18
Steiger S, Mazet A, Sandmann G (2003) Heterologous expression, purification, and enzymatic characterization of the acyclic carotenoid 1,2-hydratase from Rubrivivax gelatinosus. Arch Biochem Biophys 414:51–58
Stransky H, Hager A (1970) Das Carotenoid Muster und die Verbreitung des lichtinduzierten Xanthophyll-Cyclus in verschiedenen Algenklassen. IV, Cyanophyceae und Rhodophyceae. Arch Microbiol 72:84–96
Takaichi S, Maoka T, Masamoto K (2001) Myxoxanthophyll in Synechocystis sp PCC 6803 is myxol 2′-dimethyl-fucoside, (3R,2′S)-myxol 2′-(2,4-di-O-methyl-alpha-L-fucoside), not rhamnoside. Plant Cell Physiol 42:756–762
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Acknowledgements
We thank Dr. T. Kaneko, Kazusa DNA Research Institute for a sample of Gloeobacter violaceus DNA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steiger, S., Jackisch, Y. & Sandmann, G. Carotenoid biosynthesis in Gloeobacter violaceus PCC4721 involves a single crtI-type phytoene desaturase instead of typical cyanobacterial enzymes. Arch Microbiol 184, 207–214 (2005). https://doi.org/10.1007/s00203-005-0004-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-005-0004-5