Abstract
Summary
This study highlights an unmet need in osteoporosis management, suggesting that beyond bone mineral density and fracture history, gender, fracture type, and age should be considered for fracture risk assessment. Following fragility fracture, men, patients with a spine or hip fracture, and those aged ≥ 65 have a higher disease burden.
Introduction
The objective of this study was to characterize osteoporosis-related fracture incidence and identify predictors of subsequent fractures and mortality.
Methods
This retrospective cohort study, conducted within Kaiser Permanente Southern California, included patients aged ≥ 50 years with qualifying fractures from 1/1/2007 to 12/31/2016, identified from diagnosis/procedure codes. Rates for fracture incidence, mortality, and resource utilization in the year post-fracture are reported. Associations between index fracture types and demographic/clinical characteristics, and mortality, subsequent fracture, and rehospitalization outcomes were estimated.
Results
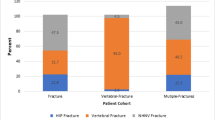
Of 63,755 eligible patients, 66.7% were ≥ 65 years and 69.1% female. Index fractures included nonhip/nonspine (64.4%), hip (25.3%), and spine (10.3%). Age-adjusted subsequent fracture rate/100 person-years was higher for those with an index spine (14.5) versus hip fracture (6.3). Hospitalization rate/100 person-years was highest for patients ≥ 65 (31.8) and for spine fractures (43.5). Men (vs women) had higher age-adjusted rates of hospitalization (19.4; 17.7), emergency room visits (73.8; 66.3), and use of rehabilitation services (31.7; 27.2). The 30-day age-adjusted mortality rate/100 person-years was 46.7, 32.4, and 15.5 for spine, hip, and nonspine/nonhip fractures. The 1-year age-adjusted mortality rate/100 person-years was 14.7 for spine and 15.6 for hip fractures. In multivariable analyses, spine and hip fractures (vs nonhip/nonspine fractures) were significant predictors of 1-year mortality, all-cause and osteoporosis-related hospitalization, and nursing home use (all P-values < 0.0001).
Conclusion
Morbidity is high in the year following a fragility fracture and men, patients with a spine or hip fracture, and those aged ≥ 65 have a greater disease burden.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis and associated fractures are an important public health burden. The subsequent effects of fractures for patients may include increased mortality and morbidity, functional decline, and loss of independence [1, 2], and the economic burden is significant regardless of the type of healthcare plan [3, 4]. Compared to patients without a fracture, those incurring a fracture have a higher total cost burden ($47,163 vs $16,034) in the year after index fracture. Furthermore, there is an incremental cost in the year following index fracture for patients incurring a second fracture compared to those who do not have a subsequent fracture ($78,137 vs $16,807). As the US population ages, the number of Americans at risk for fractures is projected to increase by 32% from 2010 to 2030 [5]. Many patients remain underdiagnosed and undertreated [3, 4], possibly due to the presence of other symptomatic comorbidities considered to require more urgent management. These other conditions (i.e., cardiovascular and respiratory disease in particular), however, may contribute to increased fracture risk and worse outcomes if the fracture patient is not adequately managed. Prior studies have focused on fracture-specific resource utilization and costs [5, 6]; however, other effects of all osteoporosis-related fractures, not just hip fractures, in the post-fracture period are less clear. Previous epidemiological studies have shown that about half of fractures occur in patients with bone mineral density (BMD) ≤−2.5 [7,8,9,10]. The evaluation of variations in outcomes for patients with different risk profiles and comorbidities in addition to BMD is therefore important. The objective of this study was to characterize osteoporosis-related fracture incidence, evaluate outcomes including subsequent fractures and mortality, and identify predictors of outcomes.
Methods
A retrospective cohort study was conducted within Kaiser Permanente Southern California, an integrated healthcare organization serving approximately 4.7 million members throughout Southern California. Eligible patients were women and men aged ≥ 50 years who sustained an osteoporosis-related fracture (excluding fingers, toes, face, and skull fractures) between January 1, 2007, and December 31, 2016 (identification period). Patients were required to have had a minimum of 2 years of continuous health plan enrollment prior to the index fracture (pre-index) to allow for determination of fracture history and other pre-existing medical conditions associated with increased fracture risk. Since patients also have pharmacy benefits as part of their health plan coverage, the gathering of information related to prescription medication use is facilitated and comprehensive. Patients with a diagnosis of Paget disease or malignancy (not including nonmelanoma skin cancer) during the pre-index period were excluded.
An algorithm was used to identify nontraumatic osteoporotic-related fractures [11]. The algorithm identifies fracture events using ICD diagnosis codes, procedure codes, and physician HCPCS codes in combination with visit types. Qualifying fractures were those identified during an inpatient stay or during an ambulatory visit accompanied by a repair procedure code. Fractures were categorized as spine only, pelvis only, shoulder (clavicle, humerus) only, radius/ulna only, hip only, other femur only, tibia/fibula only, ankle only, and multiple fracture sites (Online Resource Table 1). The earliest qualifying fracture observed during the study period was the index fracture, and the date of that first occurrence was the index date. Once an incident fracture was identified, the episode for that fracture continued until a gap of > 90 days between fracture-related encounters was observed. A qualifying fracture that occurred at the same anatomic site after 90 days was considered a new fracture. Fractures that occurred at a different anatomic site anytime following the index fracture were considered new fractures.
Clinical outcomes of interest included subsequent fracture within the year after index fracture; mortality following index fracture; 30-day and 1-year post-index fracture mortality; time to death; and time to subsequent fracture. The follow-up period was until the earliest of outcome of interest, death, disenrollment from KPSC, the end of the 1-year follow-up period, or the end of the study period (through December 31, 2017). Osteoporosis-related and all-cause healthcare resource utilization were assessed including hospitalizations, ambulatory care encounters including office visits, emergency department visits, telephone encounters, and use of nursing home, home healthcare services, and rehabilitation services. Primary reason for hospitalization and utilization were considered osteoporosis-related if there was a diagnosis of osteoporosis (ICD-9 733.0x; ICD-10 M81.0), fracture, or aftercare of fracture (ICD-9 V54.xx; ICD-10 Z47.89) associated with the healthcare encounter. High utilization was defined as the top 20% of users of healthcare services (including inpatient and outpatient encounters and associated procedures and services). Rates of hospitalization, ambulatory care encounters (outpatient, urgent care, emergency department, telephone), and mortality were age-adjusted by the US 2010 census and reported as events per 100 person-years.
For all descriptive analyses, the mean and standard deviation are provided for continuous variables; the frequency and percentage are provided for categorical variables. Crude and age-adjusted subsequent fracture rates are provided as events per 100 person-years including rates for subgroups of age, gender, and index fracture sites. Cumulative incidence plots were used to estimate time-to-event for subsequent fracture, mortality, and rehospitalization outcomes by anatomic site of fractures. Multivariable Cox proportional hazards models were used to estimate associations between index fracture types and demographic/clinical characteristics, and the mortality, subsequent fracture, and rehospitalization outcomes. Stepwise logistic regression models were used to identify significant predictors of health outcomes (with significance entry and exit set at 0.25 and 0.15, respectively).
Results
A total of 63,755 patients met the study eligibility criteria (Table 1). The majority of patients (66.7%) were ≥ 65 years of age, White (65.9%), and female (69.1%). Index fractures included nonhip/nonspine (n = 41,080; 64.4%), hip (n = 16,103; 25.3%), and spine (n = 6,572; 10.3%). Of patients with a BMD T-score data available (n = 13,282), 50.3% (n = 6,684) had a T-score ≤ − 2.5, considered to be in the osteoporotic range. In the 2-year period prior to their index fracture, 23.1% of patients were previously diagnosed and treated for osteoporosis while 44.7% of patients did not have osteoporosis diagnosis or treatment. The mean (SD) gap between the most recent BMD assessment and index fracture was 326.1 (225.2) days. Of those treated for osteoporosis with medication in the 2 years prior to their index fracture, 85% were on alendronate. One-third of all patients had a history of falls and 19.2% were hospitalized for any reason in the year prior to index fracture. Approximately one-third had a Charlson Comorbidity Index (CCI) score > 3 and the most prevalent comorbidities were cardiovascular disease (70.1%), musculoskeletal pain (59.3%), osteoarthritis (26.5%), diabetes (23.8%), and renal disease (23%). In the 90-day period prior to their index fractures, the use of medications associated with increased fall risk included opioids (32.1%), beta blockers (21.7%), selective serotonin reuptake inhibitors (SSRIs) (15.2%), and benzodiazepines/barbiturate/muscle relaxants (15.3%).
Subsequent fractures were more likely among patients who were older at the time of index fracture (45.6% for 50–64 years vs 75.6% for 85 + years). This finding held for women, but, among men, the 50–64 age group had a higher rate of a second fracture (6.3/100 person-years) compared to the 65–74 (5.7/100 person-years) and 75–84 (6.2/100 person-years) age groups. Women 50–64 had the lowest rate of nonhip/nonspine fracture rates (2.9/100 person-years) compared to women in other age categories (3.9 for 65–74; 5.9 for 75–84; 8.2 for 85 +). Age-adjusted subsequent fracture rate at any anatomic site was highest following an index spine versus index hip fracture (14.5 vs 6.3/100 person-years) (Table 2). Those with an index hip fracture had the highest rate of a second fracture of the hip (1.9/100 person-years) and those with an index spine fracture had the highest rate of subsequent spine fractures (2.8/100 person-years). Age-adjusted subsequent fracture incidence rates were higher for men versus women overall (6.2 vs 4.3/100 person-years), for spine fractures (16.3 vs 12.7/100 person-years), and for nonhip/nonspine (4.8 vs 3.6/100 person-years) fractures (Table 2).
Regarding mortality, 3.6% and 12.8% of patients died within the 30-day and 1-year post-index fracture periods, respectively. While the proportion of patients who died within the 30-day period was comparable for spine versus hip fractures (6.4% vs 6.4%), 1-year mortality was somewhat higher for hip versus spine (23.9% vs 20.6%). The 30-day age-adjusted mortality rate was 46.7/100 person-years for spine, 32.4/100 person-years for hip, and 15.5/100 person-years for nonspine/nonhip fractures. The 1-year age-adjusted mortality rate was 14.7/100 person-years for spine and 15.6/100 person-years for hip fractures. Although the same trend was observed in gender-specific stratified analyses, the mortality rate was greater for men compared to women in all age groups.
The osteoporosis-related hospitalization rate in the 1-year post-index fracture period was higher for patients aged ≥ 65 vs < 65 (31.8 vs 11.3/100 person-years). Patients with spine fractures had the highest (unadjusted) osteoporosis-related hospitalization rate in the 1-year post-index period (43.5/100 person-years) versus hip (39.5) and nonhip/nonspine fractures (16.7). Of all patients with any fracture, 13,988 (22%) used nursing home, home healthcare, or rehabilitation services. The age-adjusted rates per 100 person-years for these services in the year following the index fracture were highest for patients who incurred a hip fracture (65.6) versus those with spine (44.6) and nonhip/nonspine fractures (19.1). Age-adjusted osteoporosis-related hospitalization, emergency room, and rehabilitation service use per 100 person-years were higher for men (19.4, 73.8, and 31.7, respectively) versus women (17.7, 66.3, and 27.2), while outpatient services, telephone encounters, and urgent care use were higher for women (1630.8, 192.9, and 41.4, respectively) compared to men (1550.5, 181.3, and 36.6).
In the time-to-event analyses, after adjusting for important confounders associated with increased risk of falls, fractures, or mortality compared to patients with nonhip/nonspine fractures, those with a spine fracture were significantly more likely to have a subsequent hip fracture (HR [95% CI]: 1.26 [1.02, 1.55]; P = 0.032) or a subsequent fracture of any kind (2.11 [1.92, 2.31]; P < 0.0001); however, these findings should be interpreted with caution since the proportional hazard assumptions for subsequent fracture outcomes were not met for the model (Fig. 1). Compared to patients with nonhip/nonspine fractures, those with spine or hip fractures were significantly more likely to have an osteoporosis-related hospitalization, an all-cause hospitalization, nursing home use, and death within 1 year (P < 0.0001 for all comparisons) (Table 3 and Fig. 2). The following were associated with high use of healthcare resources: being overweight or obese; having a prior diagnosis or treatment for osteoporosis; CCI > 3; mobility impairment; and use of oral corticosteroids, benzodiazepine/barbiturates/muscle relaxants, and proton-pump inhibitors (P < 0.0001 for all). Furthermore, compared to Whites, Hispanics were significantly more likely to use urgent care (OR [95% CI]: 1.21 [1.15–1.27]; P < 0.0001), and Blacks were significantly more likely to use emergency rooms (OR [95% CI]: 1.36 [1.27–1.46]; P < 0.0001).
Discussion
The current study evaluated osteoporotic fractures in a demographically diverse population within an integrated healthcare organization. Subsequent fracture incidence, mortality, and healthcare resource utilization following index fracture were assessed. Overall, the majority of index fractures were nonhip/nonspine fractures. Forty-three percent of women in the 50–64 year old age group, a population of patients typically not considered for routine osteoporosis screening, experienced index fractures of this type. Similarly, men, a group that is often not considered for osteoporosis screening, appeared to have a higher burden of subsequent fracture and mortality compared to women across all age groups. The difference persisted after age adjustment and may be reflective of the lower recognition or treatment of osteoporosis after index fracture in men compared to women.
At the time of the index fracture, consistent with previous findings, a substantial proportion (44.7%) of patients did not have an osteoporosis diagnosis or treatment [3, 4]. Mean gap in days from last DXA scan to fractures was less than 2 years, which is consistent with the HEDIS quality of care measures for management of osteoporosis. We did not evaluate the occurrence of other imaging tests (i.e., x-ray or CT scans) or the calculation of risk prediction scores by the physician which may have occurred. Of patients who had BMD T-scores available, only half had a score within the osteoporotic range (≤ − 2.5). Known predictors of fracture include age, previous fracture, family history, and steroid use [12] and it should also be noted that a high proportion of patients in the current study had comorbidities such as cardiovascular disease, musculoskeletal pain, osteoarthritis, diabetes, and renal disease. Our findings provide further evidence that these additional factors (beyond bone density) should be considered when assessing fracture risk.
A quarter of the patients were on treatment at the time of fracture, indicating that treatments reduce, but do not eliminate, the risk of subsequent fracture. Consistent with these findings, Imel et al. showed that, despite adherence (≥ 80% medication possession ratio), approximately one-third of patients on antiresorptive therapies do not reach the treatment goals of fracture risk reduction or maintenance of BMD values [13]. Our findings suggest a need for better adherence to risk-based management strategy as per evidence-based guidelines [14].
The rate of subsequent fractures was highest following spine fractures and associations between index spine fractures, mortality rate, and healthcare resource utilization were significant. While disease burden associated with hip fractures is well recognized, the burden associated with spine fractures is underappreciated. In a 5-year prospective cohort study for both men and women with hip fracture, survival time was significantly shorter than that of the general population (P < 0.0001) [15]. Nonhip major fractures and vertebral fractures were also associated with significantly shorter survival (P = 0.003 for women, P < 0.0001 for men). According to a multinational observational study, which included patient perspectives, there is substantial loss of quality of life for patients with hip or vertebral fractures for at least 18 months following the occurrence of fracture, with reported mean (SD) difference between Health State Utility Values (HSUV) before and after hip, vertebral, and distal forearm fracture of 0.89 (0.40), 0.67 (0.45), and 0.48 (0.34), respectively (P < 0.001 for all fracture types). Eighteen months after fracture, mean HSUVs were lower than before the fracture in patients with hip fracture (0.66 vs 0.77 P < 0.001) and vertebral fracture (0.70 vs 0.83 P < 0.001) [16].
The occurrence of any fracture at any site is a significant predictor of a second fracture within the first-year post-index fracture. These findings highlight the importance of secondary fracture prevention, especially in groups typically not considered at risk such as women aged 50–64, men, and those with normal or osteopenic bone density. Early identification and treatment of high-risk patients are important in secondary fracture prevention and reduction in the overall burden of disease, which aligns with the current National Osteoporosis Foundation strategy for reduction of osteoporotic disease burden [17].
The 30-day mortality rate observed in this study is consistent with that reported in other studies, which have varied from 3.5 to 10% following a hip fracture [18, 19]. Previous studies have shown increased risk of mortality following hip fracture in patients with poor baseline functional level [20]; however, we did not have access to functional status in the current study and could not evaluate its impact on overall mortality rate post-fracture. In the current study, fewer than 10% of the study population had indicators of poor function or frailty associated with increased mortality (i.e., muscle atrophy/weakness, sarcopenia). The observed higher use of resources following hip fracture is consistent with other studies and likely associated with a higher cost of care, although the cost of care was not evaluated in the current study [21].
The study has several potential limitations, primarily those associated with the use of secondary data in observational research. First, we only had access to data that was previously collected. While the electronic health record did provide information on variables that are not typically available from claims databases, including race, BMI, and BMD T-score, it did not include other predictors of fracture risk, including lifestyle risk factors such as smoking and alcohol intake, or family history. Data on asymptomatic vertebral fractures were not available as only clinical vertebral fractures that resulted in a healthcare encounter were available. Additionally, our research was limited to death occurring in-hospital and was more likely to have included more severe cases of osteoporosis with higher associated disease burden. Furthermore, disease burden from patient perspective, which could be substantial, was not available, so the overall disease burden may be underestimated. Finally, findings from the time-to-event analysis should be interpreted with caution since the proportional hazard assumptions for subsequent fracture outcomes were not met for the model.
This study has several strengths. Our study was broad in scope in consideration of all fracture sites and included a large number of demographically diverse patients with complete data on all health encounters within a closed health system. We reduced information bias by ascertaining information for all patients using the same methods, and extracting from the electronic health record, minimizing any differential information acquisition for patients with and without the outcomes of interest. We further minimized selection bias as the study cohort included all members of qualifying age with relevant fractures from the overall KPSC membership during the study period.
In summary, our findings provide new insights on the unmet medical need in osteoporosis management and suggest consideration of risk factors beyond BMD and history of fracture for subsequent fracture risk assessment. Guidelines for earlier treatment of men to reduce subsequent fracture risk and mortality are warranted. Further evaluation of outcomes in patients with normal BMD versus osteoporosis range would also be valuable. Payers and providers alike can use the data to develop risk prediction models for improved population health management.
Availability of data and material
For this study, the Investigators will provide these research data in a controlled manner to outside researchers who are willing to enter into formal research relationships to ensure that the data will be used for scientific purposes in the public interest, that patient privacy will be protected, and that all other risks to participants will be minimized. All such requests will be reviewed by the Investigators for scientific merit, human subjects considerations, and Kaiser Permanente Southern California (KPSC) legal obligations. Primary data will reside locally within KPSC Department of Research & Evaluation. After approval of the request, a data sharing agreement will be created, approved, and signed. Conditions may be placed on the use of the data, including but not limited to, no distribution to third parties, a KPSC researcher to be included on the study team, proper acknowledgement and citation of the data providers (as indicated in the data sharing agreement) and the grant funding, exclusive use by the data recipient in connection with a specific research project, for which the recipient has sole responsibility and which is explicitly described, and agreement not to use the data in any effort to establish the identity of the study subjects. The data recipient will be subject to applicable federal, state, and local laws or regulations and institutional policies providing additional protections for human subjects.
Code availability
KPSC will make the code available upon request, if the request is deemed appropriate by the Investigator team.
References
Cauley JA (2013) Public health impact of osteoporosis. J Gerontol A Biol Sci Med Sci 68:1243–1251. https://doi.org/10.1093/gerona/glt093
Dempster DW (2011) Osteoporosis and the burden of osteoporosis-related fractures. Am J Manag Care 17:S164-169
Williams SA, Chastek B, Sundquist K et al (2020) Economic burden of osteoporotic fractures in US managed care enrollees. Am J Manag Care. 26:e142–e149. https://doi.org/10.37765/ajmc.2020.43156
Williams SA, Daigle SG, Weiss R, Wang Y, Arora T, Curtis JR (2021) Economic burden of osteoporosis-related fractures in the US medicare population. Ann Pharmacother 55(7):821–829. https://doi.org/10.1177/1060028020970518
Wright NC, Looker AC, Saag KG et al (2014) The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res 29:2520–2526. https://doi.org/10.1002/jbmr.2269
Budhia S, Mikyas Y, Tang M, Badamgarav E (2012) Osteoporotic fractures: a systematic review of U.S. healthcare costs and resource utilization. Pharmacoeconomics. 30:147–170. https://doi.org/10.2165/11596880-000000000-00000
Miller PD, Siris ES, Barrett-Connor E et al (2002) Prediction of fracture risk in postmenopausal white women with peripheral bone densitometry: evidence from the National Osteoporosis Risk Assessment. J Bone Miner Res 17:2222–2230. https://doi.org/10.1359/jbmr.2002.17.12.2222
Schuit SCE, van der Klift M, Weel AE et al (2004) Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone 34:195–202. https://doi.org/10.1016/j.bone.2003.10.001
Siris ES, Miller PD, Barrett-Connor E et al (2001) Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women: results from the National Osteoporosis Risk Assessment. JAMA 286:2815–2822. https://doi.org/10.1001/jama.286.22.2815
Sornay-Rendu E, Munoz F, Garnero P, Duboeuf F, Delmas PD (2005) Identification of osteopenic women at high risk of fracture: the OFELY study. J Bone Miner Res 20:1813–1819. https://doi.org/10.1359/JBMR.050609
Wright NC, Daigle SG, Melton ME, Delzell ES, Balasubramanian A, Curtis JR (2019) The design and validation of a new algorithm to identify incident fractures in administrative claims data. J Bone Miner Res 34:1798–1807. https://doi.org/10.1002/jbmr.3807
U.S. Department of Health and Human Services (2019). The surgeon general’s report on bone health and osteoporosis. U.S. Department of Health and Human Services. Available at: https://www.bones.nih.gov/sites/bones/files/pdfs/sgrbonehealtheng_2019_final.pdf. Accessed May 28, 2021.
Imel EA, Eckert G, Modi A et al (2016) Proportion of osteoporotic women remaining at risk for fracture despite adherence to oral bisphosphonates. Bone 83:267–275. https://doi.org/10.1016/j.bone.2015.11.021
Camacho PM, Petak SM, Binkley N et al (2020) American Association of Clinical Endocrinologists/American College of Endocrinology Clinical Practice Guidelines for the Diagnosis and Treatment of Postmenopausal Osteoporosis-2020 Update. Endocr Pract 26:564–570. https://doi.org/10.4158/GL-2020-0524SUPPL
Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA (1999) Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet 353:878–882. https://doi.org/10.1016/S0140-6736(98)09075-8
Svedbom A, Borgstom F, Hernlund E et al (2018) Quality of life for up to 18 months after low-energy hip, vertebral, and distal forearm fractures-results from the ICUROS. Osteoporos Int 29:557–566. https://doi.org/10.1007/s00198-017-4317-4
Cosman F, de Beur SJ, LeBoff MS et al (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 25:2359–2381. https://doi.org/10.1007/s00198-014-2794-2
Carretta E, Bochicchio V, Rucci P, Fabbri G, Laus M, Fantini MP (2011) Hip fracture: effectiveness of early surgery to prevent 30-day mortality. Int Orthop 35:419–424. https://doi.org/10.1007/s00264-010-1004-x
Daugaard CL, Jørgensen HL, Riis T, Lauritzen JB, Duus BR, van der Mark S (2013) Is mortality after hip fracture associated with surgical delay or admission during weekends and public holidays? A retrospective study of 38,020 patients. Acta Orthop 83:609–613. https://doi.org/10.3109/17453674.2012.747926
Haentjens P, Magaziner J, Colón-Emeric, et al (2010) Meta-analysis: excess mortality after hip fractures among older women and men. Ann Int Med 152:380–390. https://doi.org/10.7326/0003-4819-152-6-201003160-00008
Nikitovic M, Wodchis WP, Krahn MD, Cadarette SM (2013) Direct health-care costs attributed to hip fractures among seniors: a matched cohort study. Osteoporos Int 24:659–669. https://doi.org/10.1007/s00198-012-2034-6
Acknowledgements
All listed authors meet the criteria for authorship set forth by the International Committee for Medical Journal Editors. Editorial support (assembling tables and figures, collating author comments, copyediting, fact checking, and referencing) and graphic services were provided by AOIC, LLC (Sarah Hummasti, PhD), and were funded by Radius Health, Inc.
Funding
Funding for this study was provided by Radius Health, Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was reviewed and approved by the Kaiser Permanente Southern California IRB.
Consent to participate
A waiver of informed consent was received from the Kaiser Permanente Southern California IRB.
Consent for publication
Not applicable.
Conflicts of interest
Financial: Annette L. Adams is an employee of Kaiser Permanente which received funding from Radius Health, Inc. to conduct the study. Denison S. Ryan is an employee of Kaiser Permanente which received funding from Radius Health, Inc. to conduct the study. Bonnie H. Li is an employee of Kaiser Permanente which received funding from Radius Health, Inc. to conduct the study. Setareh A. Williams is an employee and shareholder of Radius Health, Inc. Yamei Wang is an employee and shareholder of Radius Health, Inc. Richard J. Weiss is a former employee and shareholder of Radius Health, Inc. Dennis M. Black provided consultancy for EffRx Pharmaceuticals, received research funding from Radius Health, Inc., and is on a data safety monitoring board for Eli Lilly (non-bone related).
Personal: The authors declare that they have no personal conflicts.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Data was previously presented Virtual Academy of Managed Care Pharmacy 2020 Annual Meeting ■ Houston, TX, USA ■ April 21‒24, 2020.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Adams, A.L., Ryan, D.S., Li, B.H. et al. Outcomes post fragility fracture among members of an integrated healthcare organization. Osteoporos Int 33, 783–790 (2022). https://doi.org/10.1007/s00198-021-06205-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-06205-w