Abstract
Summary
Liver cirrhosis leads to bone loss. To date, information on bone quality (three-dimensional microarchitecture) and, thus, bone strength is scarce. We observed decreased bone quality at both assessed sites, independent of disease severity. Therefore, all patients should undergo early-stage screening for osteoporosis.
Introduction
Recent studies found low bone mineral density in cirrhosis, but data on bone microstructure are scarce. This study assessed weight-bearing and non-weight-bearing bones in patients with cirrhosis and healthy controls. The primary objective was to evaluate trabecular and cortical microarchitecture.
Methods
This was a single-center study in patients with recently diagnosed hepatic cirrhosis. Thirty-two patients and 32 controls participated in this study. After determining the type of cirrhosis, the parameters of bone microarchitecture were assessed by high-resolution peripheral quantitative computed tomography.
Results
Both cortical and trabecular microarchitectures showed significant alterations. At the radius, trabecular bone volume fraction was 17% lower (corrected p = 0.028), and, at the tibia, differences were slightly more pronounced. Trabecular bone volume fraction was 19% lower (p = 0.024), cortical bone mineral density 7% (p = 0.007), and cortical thickness 28% (p = 0.001), while cortical porosity was 32% higher (p = 0.023), compared to controls. Areal bone mineral density was lower (lumbar spine − 13%, total hip − 11%, total body − 9%, radius − 17%, and calcaneus − 26%). There was no correlation between disease severity and microarchitecture. Areal bone mineral density (aBMD) measured by dual-energy X-ray absorptiometry (DXA) correlated well with parameters of cortical and trabecular microarchitecture.
Conclusions
Hepatic cirrhosis deteriorates both trabecular and cortical microarchitecture, regardless of disease severity. Areal bone mineral density is diminished at all sites as a sign of generalized affection. In patients with hepatic cirrhosis, regardless of its origin or disease severity, aBMD measurements are an appropriate tool for osteologic screening.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic liver disease is a common medical issue with a globally rising prevalence and high risk of cirrhosis. In hepatic cirrhosis, pathological metabolic changes result in various comorbidities, including a higher risk of osteoporosis [1].
Previous studies have found decreased areal bone mineral density (aBMD) at the lumbar spine [2, 3]. At the hip, no significant differences were observed as compared with controls [2,3,4]. However, even though dual X-ray absorptiometry (DXA) is widely used to assess fracture risk, the assessment of aBMD alone has some limitations. For instance, in a study in patients with diabetes, aBMD alone was unable to explain the increased fracture risk, and, therefore, adding important information by assessing bone microarchitecture clearly improved the diagnostic value in this investigation [5]. Still, high-resolution peripheral quantitative computed tomography (HR-pQCT) remains not systematically used in clinical routine.
To date, the literature on bone microarchitecture in liver cirrhosis has remained scarce. Several investigations have addressed histomorphometric evaluation in chronic liver disease, but the studies were inconsistent concerning etiology and stages of hepatic disease. In addition, use of control groups has been infrequent. Only two studies evaluated patients with cirrhosis. Trabecular bone volume fraction (Tb.BV/TV) was reduced [6, 7], although other studies observed normal values in chronic liver disease. Likewise, comments on detailed trabecular microarchitecture remain rare. One study [6] observed decreased trabecular thickness (Tb.Th) in cirrhosis. Information on cortical bone in patients with cirrhosis is scarce, as well. Cortical thickness (Ct.Th) was decreased [6], and cortical porosity (Ct.Po) has not yet been evaluated.
Histomorphometry is associated with several disadvantages. First, biopsies need to be taken; an invasive procedure with such potential complications as fractures or infections. Second, the biopsies are mostly performed at the iliac crest. Therefore, this method provides no reliable information on the microarchitecture of other skeletal regions, such as weight-bearing long bones, as their structure may clearly differ from that at the ilium. Third, thin samples of static histomorphometry taken from large specimens are limited to a two-dimensional (2D) view. Values of 2D histomorphometry do not consistently correlate with those measured by three-dimensional (3D) radiological methods, as the microarchitecture at the iliac crest shows high inter-individual variability and 2D histomorphometry is limited to distinct histological sections, thereby only providing a glimpse of a small part of the bone [8].
Through the use of non-invasive 3D HR-pQCT [9], investigators can assess both weight-bearing (distal tibia) and non-weight-bearing (distal radius) bones in vivo and, thus, examine cortical and trabecular microarchitecture independently from one another. HR-pQCT results correlate strongly with both microarchitecture and failure loads at the lumbar spine and proximal femur [10]. To date, 3D bone microarchitecture has not been explored in hepatic cirrhosis.
The primary objective of this study was to evaluate trabecular and cortical bone microarchitecture, assessed by HR-pQCT, in patients with hepatic cirrhosis compared with age- and sex-matched healthy controls.
Our secondary objectives were to evaluate aBMD at the lumbar spine, hip, total body, and calcaneus, trabecular bone score values, and serum markers of bone turnover.
Subjects and methods
Subjects
This single-center study was conducted at the Medical Department II of St. Vincent Hospital, an academic teaching hospital of the Medical University of Vienna and a referral center for gastrointestinal, metabolic, and bone diseases in Vienna, Austria. Female and male patients with recently diagnosed hepatic cirrhosis, who were admitted to this center to determine the type of cirrhosis and initiate liver-specific therapy, were screened for eligibility.
This study was approved by the Local Ethics Committee (Approval no. 003-02-2013), and the participants’ written informed consent was obtained prior to any patient-related procedures. This study was conducted in accordance with the Declaration of Helsinki.
The inclusion criteria were diagnosis of liver cirrhosis based on clinical presentation, medical history, imaging (sonography, computed tomography [CT] scan, or magnetic resonance imaging), and laboratory parameters, in line with the current guidelines for the diagnosis of hepatic disorders [11, 12]. In case of uncertainty, a biopsy was performed.
The etiologies of cirrhosis were the following: (1) alcoholic liver disease, defined as daily ethanol ingestion greater than 40 g in men and 20 g in women over the last years and no other causes for cirrhosis; (2) chronic viral hepatitis B or C, based on positive polymerase chain reaction results for the respective virus; (3) autoimmune hepatitis, based on a score equal or greater than 6 in the simplified autoimmune hepatitis score and responsiveness to corticosteroids; (4) non-alcoholic fatty liver disease, defined as the presence of hepatic steatosis detected by histology and no cause for secondary hepatic fat accumulation, such as significant alcohol consumption, use of steatogenic medication, or hereditary disorders; and (5) storage diseases, such as hemochromatosis based on histology and typical laboratory findings. Six patients with primary biliary cirrhosis or primary sclerosing cholangitis were screened but excluded, as they did not fulfill the eligibility criteria concerning comorbidities.
The exclusion criteria were age < 18 years; pregnancy or lactation; history of high-energy trauma; gastrointestinal and endocrine diseases (inflammatory bowel disease, i.e., Crohn’s disease, ulcerative colitis); chronic kidney disease (chronic kidney disease stages II–V, renal association); insulin-dependent diabetes mellitus type I or II; hypo- or hyperthyroid metabolic status (substitution was not an exclusion criterion); biliary obstruction; oncological and rheumatic diseases (rheumatoid arthritis, spondyloarthritis, systemic lupus erythematosus); congestive heart failure (New York Heart Association Classes II–IV); human immunodeficiency virus infection; drug abuse; immobility, wheelchair dependence, or bedridden state; body mass index < 20 and > 35 kg/m2; and treatment with the following medications: any prior liver-specific medication (except nutrition supplements, e.g., silibinin), any medication that could cause elevation of liver enzymes (e.g., methotrexate, chronic paracetamol use), adrenal or anabolic steroids (e.g., > 5 mg prednisolone per day), glitazones, anticonvulsants, long-term use of anticoagulants (> 10 years), and/or any osteoporosis treatment (including calcium and/or vitamin D supplementation).
The healthy controls (subject to the same exclusion criteria but no history and no laboratory evidence of liver disease) were recruited from active and retired hospital staff.
Radiology
Vertebral fractures were detected by digital radiographs according to the Genant criteria. The patients received an abdominal ultrasound to assess ascites and to exclude malignancies.
Two technologists certified by the International Osteoporosis Foundation/International Society for Clinical Densitometry applied DXA to assess aBMD.
The lumbar spine (L1–L4), non-dominant radius (except for previous fracture), total body, total hip, and femoral neck were scanned after daily calibration with standardized phantoms (iDXA®, GE Healthcare Lunar, Madison, WI, USA, latest software version). Fractured vertebrae were excluded from all investigations. The in vivo precision of error, expressed as coefficient of variation (CV), was 0.41% for the lumbar spine and 0.53% for the hip [13].
In addition, in a subgroup of 19 patients and 28 controls, trabecular bone score was automatically assessed from lumbar DXA via TBS iNsight® (Medimaps SA, Canéjan, France, latest software version). In 9 patients and 8 controls, calcaneal BMD was assessed by dual X-ray and laser (DXL; Calcscan®, Demtech AB, Taby, Sweden, latest software version). According to the literature, the CV was 1.19% [14]. Due to technical issues (a deletion of raw data at the DXA device due to a software update, technical failure of DXL over a period of several months, and lack of technical support by the Swedish hardware provider), we were unable to obtain the trabecular bone score and calcaneal BMD from all subjects. These subjects were not selected deliberately.
Bone microarchitecture
Two specifically trained technologists performed the HR-pQCT examinations (XtremeCT, Scanco Medical, Brütisellen, Switzerland) following daily calibrations with a standardized phantom (Moehrendorf, Germany) and according to the manufacturer’s standard in vivo protocol. The non-dominant (except for previous fracture) distal radius and distal tibia were scanned while immobilized in a carbon-fiber cast. The manufacturer’s standard software was used for all measurements and evaluations, and the reference lines were set manually according to the anteroposterior scout view. There were 110 CT slices (82 μm voxel size), with the first slice 9.5 mm proximal to the reference line. Each measurement lasted 2.8 min, and the effective dose was lower than 3 μSv. The Scanco SOP scale was used to exclude scans with motion artifacts graded higher than 3.
The following parameters of volumetric BMD (vBMD), bone microstructure, and bone geometry were assessed: cortical BMD, Tb.BV/TV, trabecular number (Tb.N/mm), Tb.Th, trabecular separation (Tb.Sp), Ct.Th, and cortical porosity (Ct.Po).
Clinical chemistry and serum markers of bone turnover
Parameters of bone turnover were obtained after overnight fasting (sampling between 8 and 10 a.m.) at the ISO 9001-certified laboratory (Labcon Ltd) at the St. Vincent Hospital. The following parameters were determined: calcium, alkaline phosphatase, phosphorus, C-terminal telopeptide of type I collagen (CTX), 25-OH vitamin D, intact parathyroid hormone (iPTH), and procollagen type 1 N-terminal peptide (P1NP). Furthermore, the following parameters related to liver disease were determined: aspartate aminotransferase, alanine aminotransferase, gamma glutamyl transferase, creatinine, albumin, bilirubin, international normalized ratio, and partial thromboplastin time (IDS-iSYS microparticle immunoassay system, Immunodiagnostics Systems Ltd., Boldon, UK; Architect ci8200 platform, Abbott Laboratories, Abbott Park, IL, USA; and ACL TOP 500 CTS, Instrumentation Laboratory, Bedford, MA, USA). Serum calcium was adjusted for low albumin.
Statistical analysis
Sample size calculations are based on standard deviations from a pilot evaluation of Tb.BV/TV in 8 patients with cirrhosis (3 due to alcoholic liver disease, 3 due to viral hepatitis, and 2 due to non-alcoholic fatty liver disease). In order to detect a mean difference in Tb.BV/TV of 0.03 with a power of 80%, 32 patients were to be included per group.
Central tendency and dispersion measurements were calculated for all variables. Group differences between patients and controls and between patients with alcoholic liver disease and non-alcoholic etiology were carried out using two-sample t-tests. Distributional assumptions for t-tests were checked visually by quantile–quantile plots.
Multiple linear regression models were performed to evaluate the relationship between cortical and trabecular microarchitecture parameters and aBMD measures, serum markers of bone turnover (alkaline phosphatase, iPTH, and CTX), and Child-Pugh and Model for End-Stage Liver Disease (MELD) scores. Separate models were derived for each microarchitecture parameter as dependent variable and BMD or serum markers of bone turnover measures as explanatory variables. Model fits were quantified by R2 values and p values. For each model, the improvement in model fit by MELD and Child-Pugh scores was tested using F-tests.
All tests were two-sided, and p values less than 0.05 were considered statistically significant. As bone microarchitecture is described by multiple parameters, corresponding p values were adjusted to control a false discovery rate (FDR) of 0.05. All statistical analyses were performed with the statistical software R version 3.50 (R Development Core Team, 2018).
Results
Study demographics
In total, 32 patients (12 women) and 32 healthy age- and sex-matched controls (12 women) with a median age of 62 and 60 years, respectively, were included—16 patients with alcoholic liver disease and 16 with non-alcoholic etiology (8 with viral cirrhosis, 5 with non-alcoholic fatty liver disease, 2 with hemochromatosis, and one with autoimmune hepatitis). The demographic data were similar between patients and healthy controls with the exception of higher alcohol intake in patients (Table 1).
The Child-Pugh and MELD scores were similar between the groups; yet, the packyears of smoking and alcohol intake were higher among subjects with alcoholic liver disease.
Spinal X-ray identified low-trauma vertebral fractures in 13 patients with cirrhosis (40%), all of whom were asymptomatic (morphometric). Of these patients, five also had a history of at least one low-trauma non-vertebral fracture. None of the controls had a prevalent low-trauma vertebral or non-vertebral fracture.
Bone microarchitecture
At the radius, the patients and controls had similar cortical BMD values. In addition, no differences in Ct.Th or Ct.Po were found. However, Tb.BV/TV was significantly lower in cirrhotic patients, and Tb.Th only showed a trend to a decrease.
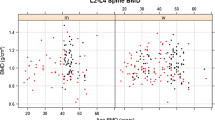
Alterations were more pronounced at the tibia. Among the patients, cortical BMD and Ct.Th were significantly lower with increased Ct.Po. Tb.BV/TV was significantly lower in the patients, as well, but there was only a trend towards higher Tb.Sp and lower Tb.Th and Tb.N (Table 2 and Fig. 1).
Images of a patient with hepatic cirrhosis, compared with a matched healthy control. a Sample of the distal radius; b sample of the distal tibia; c three-dimensional reconstruction of the distal radius; d three-dimensional reconstruction of the distal tibia; e three-dimensional visualization of cortical porosity. The images demonstrate both trabecular thinning and rarefication (higher separation between trabeculae), cortical thinning, as well as higher cortical porosity
Between the patients with and without low-trauma vertebral fractures, no differences in trabecular microarchitecture were observed. However, cortical BMD was lower at the tibia (759 vs. 805 mg/cm3, uncorrected p = 0.03), and there was a trend towards higher Ct.Po at the tibia (10.84 vs. 9.09%, uncorrected p = 0.06) in patients with low-trauma vertebral fractures.
Areal BMD and trabecular bone score
Areal BMD was lower in patients at all skeletal sites (lumbar spine, total hip, femoral neck, total body BMD, radius, and calcaneus). At both the lumbar spine and the hip, the Z scores were significantly lower in the patients compared with the controls: the difference was 1.25 at the lumbar spine and 0.85 at the hip.
Changes at the total hip and femoral neck were similar (Table 3).
The trabecular bone score values were decreased by 17% in the patients compared with the controls. Patients with and without a history of low-trauma vertebral fractures had similar trabecular bone score values (p = 0.75).
Clinical chemistry and serum markers of bone turnover
The liver enzymes were increased in the patient group, and the coagulation parameters were altered in terms of prolonged partial thromboplastin time with increased international normalized ratio. The patients had higher serum phosphorus and lower adjusted calcium values, even though both parameters were within normal ranges. 25-OH vitamin D and CTX were similar, and both P1NP and alkaline phosphatase were higher in the patient group (Table 4).
No differences in bone microarchitecture, markers of bone turnover, aBMD, or the trabecular bone score were observed between subjects with alcoholic liver disease and the non-alcoholic group.
Correlations (regression analysis)
As calculated by multiple regression analysis, significant correlations between most parameters of trabecular and cortical microarchitecture (except cortical BMD) were observed with aBMD of the lumbar spine and hip. The strongest correlation was observed for tibial Tb.BV/TV (R2 = 0.5, p < 0.001) and tibial Tb.N (R2 = 0.51, p < 0.001).
In the multiple regression analysis, there was no significant correlation between microarchitecture and the Child-Pugh score or the MELD score, even when adjusted for both aBMD (lumbar spine, hip, total body, and radius) and serum markers of bone turnover (alkaline phosphatase, iPTH, and CTX). Therefore, differences in microarchitecture were independent of disease severity (e.g., tibial Tb.BV/TV F2 = 0.09, p = 0.77; tibial Tb.N F2 = 0.79, p = 0.38).
Discussion
To date, this is the first study to evaluate bone microarchitecture in hepatic cirrhosis via state-of-the-art non-invasive HR-pQCT. We observed significantly impaired microarchitecture at both the trabecular and cortical compartments, as well as differences in aBMD and serum markers of bone turnover.
To our knowledge, only two studies have performed histomorphometry in patients with cirrhosis [6, 7]. Other investigations in patients with chronic liver disease varied concerning etiology and severity [15, 16]. Additionally, no detailed parameters of cortical microstructure or bone geometry have so far been evaluated in patients with cirrhosis. Dynamic histomorphometry in cirrhosis has suggested reduced formation, increased resorption, and normal mineralization [6] with no signs of osteomalacia [7].
We assessed bone microarchitecture at both weight-bearing and non-weight-bearing sites and observed increased Ct.Po and decreased Ct.Th in hepatic cirrhosis. Ct.Th and Ct.Po are closely related to cortical integrity and bone strength [17]. Thus, Ct.Th is decreased in chronic kidney disease patients with sustained fractures [18] and Ct.Po increased in diabetics, especially in those with prevalent fragility fractures [19]. Moreover, we observed impaired trabecular microarchitecture with decreased Tb.BV/TV and a trend towards lower Tb.Th and elevated Tb.Sp. Impaired trabecular microarchitecture is associated with fragility fractures; chronic kidney disease patients who had sustained fractures have lower Tb.BV/TV, and Tb.N is decreased with increased Tb.Sp [18].
Based on the deteriorations in both trabecular and cortical microarchitecture, we assume increased susceptibility for fragility fractures in hepatic cirrhosis. Recent studies have identified both vertebral and non-vertebral fractures in a high percentage of patients with chronic liver disease, the prevalence varying between 7 and 35% [20]. Moreover, in up to 49% of the patients with advanced liver disease, there was at least one self-reported low-trauma fracture [21], and in a study on hypogonadal cirrhotic patients, 71% had sustained vertebral or non-vertebral fractures, including the hip, regardless of trauma intensity [22]. Compared with these studies, a relatively high percentage of our patients (40%) had sustained at least one low-trauma fracture. However, not all of the previous studies have incorporated spinal X-rays, and vertebral fractures may have, thus, been underdiagnosed.
In line with current studies investigating patients in the same age group [2, 3], we observed lower vertebral aBMD. In contrast to other investigations [2,3,4], we also found decreased aBMD at the total hip, femoral neck, the total body scan, as well as at the calcaneus, implying that hepatic cirrhosis affects the entire skeleton, independent of etiology.
Trabecular bone score, which is automatically calculated from gray scale levels derived from DXA measurements, is capable of reflecting vertebral trabecular microarchitecture [23]. Lower trabecular bone scores accurately predict impaired trabecular microarchitecture in various patient groups, such as those with pre- or postmenopausal osteoporosis [23], osteogenesis imperfecta [24], chronic kidney disease [25], or diabetes [26]. To date, this is the first evaluation of trabecular bone score in hepatic cirrhosis and, compared with the aforementioned patient groups with increased fracture risk, trabecular bone score was even more strongly decreased in our patients (17%).
In hepatic cirrhosis, several metabolic alterations contribute to bone loss, including increased oncofetal fibronectin [27]; decreased levels of insulin-like growth factor 1 synthesized in the liver [28]; upregulation of proinflammatory cytokines [28]; chronic cholestasis with retained bilirubin and bile acids [29]; hypogonadism [30]; and higher alcohol consumption, smoking, lower physical activity, and malnutrition with muscle wasting [28].
Low total calcium levels in hepatic cirrhosis are common in the literature [6, 31], even though other authors have reported normal levels [2]. In our patients, the adjusted total calcium levels were within normal ranges but significantly lower than in the controls. In addition, phosphorus levels were higher in our patients than the controls but still within normal ranges. In a normal population, higher phosphorus levels within normal ranges were related to increased fracture risk [32]. In our study, iPTH, 25-OH vitamin D, and CTX were similar in the patients and the controls and thereby unlikely to explain increased phosphorus levels. Finally, patients with hepatic cirrhosis are likely to consume high quantities of processed food containing high amounts of phosphorus additives [33].
In our patients, the standard serum markers of bone turnover (CTX, P1NP) did not reflect the unfavorable changes that lead to the deterioration of both cortical and trabecular microarchitecture. Increased P1NP levels in our patients could be explained by altered hepatic collagen metabolism [34]. Therefore, collagen-based serum parameters do not appear to be valuable markers of bone turnover in hepatic cirrhosis.
As tested by multiple regression analysis, we observed no correlation between bone microarchitecture and the severity of liver disease, as reflected by the Child-Pugh or MELD scores, even when adjusted for aBMD and serum markers of bone turnover. These findings indicate a loss of structural integrity already at the early stages. Similarly, histomorphometric studies have reported no differences between different Child-Pugh stages [7].
The impact of the etiology of cirrhosis is under debate in the literature. Some studies have observed no differences in fracture risk [1], but others have suggested stronger bone loss in cholestatic than viral cirrhosis [35].
Similar to our work, a recent study in non-cirrhotic primary biliary cholangitis patients of a similar age [36] observed deranged microarchitecture of both cortical and trabecular bones. In detail, Ct.Th, BV/TV, and Tb.Th were decreased with unchanged Tb.N at both the radius and tibia. Ct.Po and serum markers of bone turnover were not determined. In contrast to our findings, disease stage was related to the degree of microstructural derangement.
Some limitations of this study are to be mentioned. First, we were unable to precisely define the duration of hepatic cirrhosis. However, we provided Child-Pugh and MELD scores to reflect disease severity. Second, we were dependent on the subjects’ statements concerning units of alcohol intake and packyears of smoking. Third, due to technical issues, we could not determine trabecular bone score and calcaneal aBMD in all subjects. Fourth, we determined only one parameter of bone formation (P1NP). Fifth, the threshold-based algorithm of image segmentation could possibly have had an influence on the portions of trabecular and cortical bone. Sixth, our study possibly lacked the power to test for differences between different etiologies. Finally, we could have underestimated differences in Ct.Po, as smaller pores are possibly not recognized at a voxel size of 82 μm.
In conclusion, both cortical and cancellous bone microarchitectures were affected at all assessed sites and structural deteriorations were independent of disease severity. Especially at the tibia, cortical bone was affected with both decreased Ct.Th and increased Ct.Po. Areal BMD was diminished at all measured sites as a sign of generalized bone disease in patients with hepatic cirrhosis. This is supported by differences in bone microarchitecture, non-invasively measured by HR-pQCT. A clear statement of this study is the implementation of DXA as an easily obtainable screening method for osteoporosis in this patient group to evaluate an individual fracture risk. We, therefore, generally recommend osteologic screening, including DXA of the lumbar spine and hip, for patients with hepatic cirrhosis, independent of its etiology or disease severity.
Abbreviations
- aBMD:
-
Areal bone mineral density
- Ct.Po:
-
Cortical porosity
- Ct.Th:
-
Cortical thickness
- CTX:
-
C-terminal telopeptide of type I collagen
- CV:
-
Coefficient of variation
- DXA:
-
Dual X-ray absorptiometry
- DXL:
-
Dual X-ray and laser
- FDR:
-
False discovery rate
- HR-pQCT:
-
High-resolution peripheral quantitative computed tomography
- iPTH:
-
Intact parathyroid hormone
- MELD:
-
Model for End-Stage Liver Disease
- P1NP:
-
Procollagen type 1 N-terminal peptide
- Tb.BV/TV:
-
Trabecular bone volume fraction
- Tb.N:
-
Trabecular number
- Tb.Sp:
-
Trabecular separation
- Tb.Th:
-
Trabecular Thickness
- vBMD:
-
Volumetric BMD
References
Guarino M, Loperto I, Camera S, Cossiga V, di Somma C, Colao A, Caporaso N, Morisco F (2016) Osteoporosis across chronic liver disease. Osteoporos Int 27:1967–1977. https://doi.org/10.1007/s00198-016-3512-z
Santos LAA, Lima TB, Augusti L, Franzoni LC, Yamashiro FS, Bolfi F, Nunes VS, Dorna MS, de Oliveira CV, Caramori CA, Silva GF, Romeiro FG (2016) Handgrip strength as a predictor of bone mineral density in outpatients with cirrhosis. J Gastroenterol Hepatol 31:229–234. https://doi.org/10.1111/jgh.13062
Lupoli R, Di Minno A, Spadarella G et al (2016) The risk of osteoporosis in patients with liver cirrhosis: a meta-analysis of literature studies. Clin Endocrinol 84:30–38. https://doi.org/10.1111/cen.12780
Culafić D, Djonic D, Culafic-Vojinovic V, Ignjatovic S, Soldatovic I, Vasic J, Beck TJ, Djuric M (2015) Evidence of degraded BMD and geometry at the proximal femora in male patients with alcoholic liver cirrhosis. Osteoporos Int 26:253–259. https://doi.org/10.1007/s00198-014-2849-4
Burghardt AJ, Issever AS, Schwartz AV, Davis KA, Masharani U, Majumdar S, Link TM (2010) High-resolution peripheral quantitative computed tomographic imaging of cortical and trabecular bone microarchitecture in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab 95:5045–5055. https://doi.org/10.1210/jc.2010-0226
Guichelaar MMJ, Malinchoc M, Sibonga J et al (2002) Bone metabolism in advanced cholestatic liver disease: analysis by bone histomorphometry. Hepatology 36:895–903. https://doi.org/10.1053/jhep.2002.36357
Jorge-Hernandez JA, Gonzalez-Reimers CE, Torres-Ramirez A, Santolaria-Fernandez F, Gonzalez-Garcia C, Batista-Lopez JN, Pestana-Pestana M, Hernandez-Nieto L (1988) Bone changes in alcoholic liver cirrhosis—a histomorphometrical analysis of 52 cases. Dig Dis Sci 33:1089–1095. https://doi.org/10.1007/BF01535783
Cohen A, Dempster DW, Müller R, Guo XE, Nickolas TL, Liu XS, Zhang XH, Wirth AJ, van Lenthe GH, Kohler T, McMahon DJ, Zhou H, Rubin MR, Bilezikian JP, Lappe JM, Recker RR, Shane E (2010) Assessment of trabecular and cortical architecture and mechanical competence of bone by high-resolution peripheral computed tomography: comparison with transiliac bone biopsy. Osteoporos Int 21:263–273. https://doi.org/10.1007/s00198-009-0945-7
Boutroy S, Bouxsein ML, Munoz F, Delmas PD (2005) In vivo assessment of trabecular bone microarchitecture by high-resolution peripheral quantitative computed tomography. J Clin Endocrinol Metab 90:6508–6515. https://doi.org/10.1210/jc.2005-1258
Kroker A, Plett R, Nishiyama KK, McErlain DD, Sandino C, Boyd SK (2017) Distal skeletal tibia assessed by HR-pQCT is highly correlated with femoral and lumbar vertebra failure loads. J Biomech 59:43–49. https://doi.org/10.1016/j.jbiomech.2017.05.011
O’Shea RS, Dasarathy S, McCullough AJ et al (2010) Alcoholic liver disease. Hepatology 51:307–328. https://doi.org/10.1002/hep.23258
Chalasani N, Younossi Z, Lavine JE, Diehl AM, Brunt EM, Cusi K, Charlton M, Sanyal AJ (2012) The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 55:2005–2023. https://doi.org/10.1002/hep.25762
Hind K, Oldroyd B, Truscott JG (2010) In vivo precision of the GE Lunar iDXA densitometer for the measurement of total-body, lumbar spine, and femoral bone mineral density in adults. J Clin Densitom 13:413–417. https://doi.org/10.1016/j.jocd.2010.06.002
Thorpe JA, Steel SA (2006) The DXL Calscan heel densitometer: evaluation and diagnostic thresholds. Br J Radiol 79:336–341. https://doi.org/10.1259/bjr/22191429
Stellon AJ, Webb A, Compston J, Williams R (1987) Low bone turnover state in primary biliary cirrhosis. Hepatology 7:137–142. https://doi.org/10.1002/hep.1840070127
Diamond TH, Stiel D, Lunzer M, McDowall D, Eckstein RP, Posen S (1989) Hepatic osteodystrophy. Static and dynamic bone histomorphometry and serum bone Gla-protein in 80 patients with chronic liver disease. Gastroenterology 96:213–221
Sornay-Rendu E, Boutroy S, Munoz F, Delmas PD (2007) Alterations of cortical and trabecular architecture are associated with fractures in postmenopausal women, partially independent of decreased BMD measured by DXA: the OFELY study. J Bone Miner Res 22:425–433. https://doi.org/10.1359/JBMR.061206
Nickolas TL, Stein E, Cohen A, Thomas V, Staron RB, McMahon DJ, Leonard MB, Shane E (2010) Bone mass and microarchitecture in CKD patients with fracture. J Am Soc Nephrol 21:1371–1380. https://doi.org/10.1681/ASN.2009121208
Patsch JM, Burghardt AJ, Yap SP, Baum T, Schwartz AV, Joseph GB, Link TM (2013) Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J Bone Miner Res 28:313–324. https://doi.org/10.1002/jbmr.1763
Mansueto P, Carroccio A, Seidita A, di Fede G, Craxì A (2013) Osteodystrophy in chronic liver diseases. Intern Emerg Med 8:377–388. https://doi.org/10.1007/s11739-012-0753-5
Wibaux C, Legroux-Gerot I, Dharancy S, Boleslawski E, Declerck N, Canva V, Mathurin P, Pruvot FR, Cortet B (2011) Assessing bone status in patients awaiting liver transplantation. Joint Bone Spine 78:387–391. https://doi.org/10.1016/j.jbspin.2011.03.001
Diamond T, Stiel D, Lunzer M, Wilkinson M, Roche J, Posen S (1990) Osteoporosis and skeletal fractures in chronic liver disease. Gut 31:82–87. https://doi.org/10.1136/gut.31.1.82
Muschitz C, Kocijan R, Haschka J, Pahr D, Kaider A, Pietschmann P, Hans D, Muschitz GK, Fahrleitner-Pammer A, Resch H (2015) TBS reflects trabecular microarchitecture in premenopausal women and men with idiopathic osteoporosis and low-traumatic fractures. Bone 79:259–266. https://doi.org/10.1016/j.bone.2015.06.007
Kocijan R, Muschitz C, Haschka J, Hans D, Nia A, Geroldinger A, Ardelt M, Wakolbinger R, Resch H (2015) Bone structure assessed by HR-pQCT, TBS and DXL in adult patients with different types of osteogenesis imperfecta. Osteoporos Int 26:2431–2440. https://doi.org/10.1007/s00198-015-3156-4
Pérez-Sáez MJ, Herrera S, Prieto-Alhambra D, Vilaplana L, Nogués X, Vera M, Redondo-Pachón D, Mir M, Güerri R, Crespo M, Díez-Pérez A, Pascual J (2017) Bone density, microarchitecture, and material strength in chronic kidney disease patients at the time of kidney transplantation. Osteoporos Int 28:2723–2727. https://doi.org/10.1007/s00198-017-4065-5
Kim JH, Choi HJ, Ku EJ, Kim KM, Kim SW, Cho NH, Shin CS (2015) Trabecular bone score as an indicator for skeletal deterioration in diabetes. J Clin Endocrinol Metab 100:475–482. https://doi.org/10.1210/jc.2014-2047
Sens C, Altrock E, Rau K, Klemis V, von Au A, Pettera S, Uebel S, Damm T, Tiwari S, Moser M, Nakchbandi IA (2017) An O-glycosylation of fibronectin mediates hepatic osteodystrophy through α4β1 integrin. J Bone Miner Res 32:70–81. https://doi.org/10.1002/jbmr.2916
Nakchbandi IA, van der Merwe SW (2009) Current understanding of osteoporosis associated with liver disease. Nat Rev Gastroenterol Hepatol 6:660–670. https://doi.org/10.1038/nrgastro.2009.166
Hodgson SF, Dickson ER, Eastell R, Eriksen EF, Bryant SC, Riggs BL (1993) Rates of cancellous bone remodeling and turnover in osteopenia associated with primary biliary cirrhosis. Bone 14:819–827. https://doi.org/10.1016/8756-3282(93)90310-7
Foresta C, Schipilliti M, Ciarleglio FA, Lenzi A, D’Amico D (2008) Male hypogonadism in cirrhosis and after liver transplantation. J Endocrinol Investig 31:470–478. https://doi.org/10.1007/BF03346393
Guichelaar MMJ, Kendall R, Malinchoc M, Hay JE (2006) Bone mineral density before and after OLT: long-term follow-up and predictive factors. Liver Transpl 12:1390–1402. https://doi.org/10.1002/lt.20874
Campos-Obando N, Koek WNH, Hooker ER, van der Eerden BCJ, Pols HA, Hofman A, van Leeuwen JPTM, Uitterlinden AG, Nielson CM, Zillikens MC (2017) Serum phosphate is associated with fracture risk: the Rotterdam Study and MrOS. J Bone Miner Res 32:1182–1193. https://doi.org/10.1002/jbmr.3094
European Food Safety Authority (2013) Assessment of one published review on health risks associated with phosphate additives in food. EFSA J 11:3444. https://doi.org/10.2903/j.efsa.2013.3444
Monegal A, Navasa M, Peris P, Alvarez L, Pons F, Rodés J, Guañabens N (2007) Serum osteoprotegerin and its ligand in cirrhotic patients referred for orthotopic liver transplantation: relationship with metabolic bone disease. Liver Int 27:492–497. https://doi.org/10.1111/j.1478-3231.2007.01448.x
Wariaghli G, Allali F, El Maghraoui A, Hajjaj-Hassouni N (2010) Osteoporosis in patients with primary biliary cirrhosis. Eur J Gastroenterol Hepatol 22:1397–1401. https://doi.org/10.1097/MEG.0b013e3283405939
Schmidt T, Schmidt C, Schmidt FN, Butscheidt S, Mussawy H, Hubert J, Hawellek T, Oehler N, Barvencik F, Lohse AW, Schinke T, Schramm C, Amling M, Rolvien T (2018) Disease duration and stage influence bone microstructure in patients with primary biliary cholangitis. J Bone Miner Res 33:1011–1019. https://doi.org/10.1002/jbmr.3410
Acknowledgments
The authors are grateful to Dragana Simic, Patricia Gumbo, and Arastoo Nia for their assistance in data acquisition and to Prof. Tatjana Paternostro-Sluga for critical reading.
Funding
Open access funding provided by Medical University of Vienna.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure statement
CM has received speaker honoraria from Amgen, Novartis, Servier, Eli Lilly, Nycomed Pharma/Takeda, Kwizda Pharma, Boehringer Ingelheim, Actavis, and Daiichi Sankyo. He has also received educational grants/research support from the Austrian Society for Bone and Mineral Research, Roche Austria, Eli Lilly Austria, Eli Lilly International, and Amgen Austria. He has nothing to disclose concerning this manuscript.
RK has received speaker honoraria from Eli Lilly. He has nothing to disclose concerning this manuscript.
PP has received research support and/or honoraria from Amgen, Eli Lilly, Fresenius Kabi Austria, Servier Austria, Shire, and UCB Pharma. He has nothing to disclose concerning this manuscript.
HR has received speaker honoraria from Amgen, Novartis, Servier, Eli Lilly, and Nycomed Pharma/Takeda. He has also received educational grants/research support from the Austrian Society for Bone and Mineral Research, Roche Austria, Eli Lilly Austria, Eli Lilly International, and Amgen Austria. He has nothing to disclose concerning this manuscript.
All the other authors have nothing to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
OpenAccess This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Wakolbinger, R., Muschitz, C., Scheriau, G. et al. Bone microarchitecture and bone turnover in hepatic cirrhosis. Osteoporos Int 30, 1195–1204 (2019). https://doi.org/10.1007/s00198-019-04870-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-019-04870-6