Abstract
Summary
This paper provides a framework for the development of national guidelines for the management of glucocorticoid-induced osteoporosis in men and women aged 18 years and over in whom oral glucocorticoid therapy is considered for 3 months or longer.
Introduction
The need for updated guidelines for Europe and other parts of the world was recognised by the International Osteoporosis Foundation and the European Calcified Tissue Society, which set up a joint Guideline Working Group at the end of 2010.
Methods and results
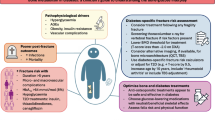
The epidemiology of GIO is reviewed. Assessment of risk used a fracture probability-based approach, and intervention thresholds were based on 10-year probabilities using FRAX. The efficacy of intervention was assessed by a systematic review.
Conclusions
Guidance for glucocorticoid-induced osteoporosis is updated in the light of new treatments and methods of assessment. National guidelines derived from this resource need to be tailored within the national healthcare framework of each country.




Similar content being viewed by others
References
Compston JE (2010) Management of glucocorticoid-induced osteoporosis. Nature Rev Rheumatol 6:82–88
American College of Rheumatology ad hoc Committee on Glucocorticoid-induced Osteoporosis (2001) Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis: 2001 update. Arthritis Rheum 44:1496–1503
Royal College of Physicians (2002) Glucocorticoid-induced osteoporosis. Guidelines on prevention and treatment. Bone and Tooth Society of Great Britain, National Osteoporosis Society and Royal College of Physicians. Royal College of Physicians, London
Brown JP, Josse RG, Scientific Advisory Council of the Osteoporosis Society of Canada (2002) Clinical practice guidelines for the diagnosis and management of osteoporosis in Canada. CMAJ 167:S1–S34
Devogelaer JP, Goemaere S, Boonen S, Body JJ, Kaufman JM, Reginster JY, Rozenberg S, Boutsen Y (2006) Evidence-based guidelines for the prevention and treatment of glucocorticoid-induced osteoporosis: a consensus document of the Belgian Bone Club. Osteoporos Int 17:8–19
Geusens PP, Lems WF, Verhaar HJ, Leusink G, Goemaere S, Zmierczack H, Compston JE (2006) Review and evaluation of the Dutch guidelines for osteoporosis. J Eval Clin Pract 12:539–548
Gourlay M, Franceschini N, Sheyn Y (2007) Prevention and treatment strategies for glucocorticoid-induced osteoporosis. Clin Rheumatol 26:144–153
Hoes JN, Jacobs JW, Boers M, Boumpas D, Buttgereit F, Caeyers N, Choy EH, Cutolo M, Da Silva JA, Esselens G, Guillevin L, Hafstrom I, Kirwan JR, Rovensky J, Russell A, Saag KG, Svensson B, Westhovens R, Zeidler H, Bijlsma JW (2007) EULAR evidence-based recommendations on the management of systemic glucocorticoid therapy in rheumatic diseases. Ann Rheum Dis 66:1560–1567
Nawata H, Soen S, Takayanagi R, Tanaka I, Takaoka K, Fukunaga M, Matsumoto T, Suzuki Y, Tanaka H, Fujiwara S, Miki T, Sagawa A, Nishizawa Y, Seino Y, Subcommittee to Study Diagnostic Criteria for Glucocorticoid-Induced Osteoporosis (2005) Guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society for Bone and Mineral Research (2004). J Bone Miner Metab 23:105–109
National Osteoporosis Foundation (2008) Clinician's guide to prevention and treatment of osteoporosis. National Osteoporosis Foundation, Washington, DC, www.nof.org. Accessed 15 August 2011
Grossman JM, Gordon R, Ranganath VK, Deal C, Caplan L, Chen W, Curtis JR, Furst DE, McMahon M, Patkar NM, Volkmann E, Saag KG (2010) American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arth Care Res 62:1515–1526
Díez-Pérez A, Hooven FH, Adachi JD, Adami S, Anderson FA, Boonen S, Chapurlat R, Compston JE, Cooper C, Delmas P, Greenspan SL, LaCroix AZ, Lindsay R, Netelenbos JC, Pfeilschifter J, Roux C, Saag KG, Sambrook P, Silverman S, Siris ES, Watts NB, Nika G, Gehlbach SH (2011) Regional differences in treatment for osteoporosis. The Global Longitudinal Study of Osteoporosis in Women (GLOW). Bone 49:493–498
van Staa TP, Leufkens HG, Cooper C (2002) The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int 13:777–787
van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Oral corticosteroids and fracture risk: relationship to daily and cumulative doses. Rheumatol Phys Med 39:1383–1389
van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Use of oral corticosteroids and risk of fractures. J Bone Miner Res 15:933–1000
Hoes JN, Jacobs JW, Hulsmans HM, De Nijs RN, Lems WF, Bruyn GA, Geusens PP, Bijlsma JW (2010) High incidence rate of vertebral fractures during chronic prednisone treatment, in spite of bisphosphonate or alfacalcidol use. Extension of the alendronate or alfacalcidol in glucocorticoid-induced osteoporosis-trial. Clin Exp Rheumatol 28:354–359
Kanis JA, Johansson H, Oden A, Johnell O, de Laet C, Melton LJ III, Tenenhouse A, Reeve J, Silman AJ, Pols HA, Eisman JA, McCloskey EV, Mellstrom D (2004) A meta-analysis of prior corticosteroid use and fracture risk. J Bone Miner Res 19:893–899
van Staa TP, Leufkens HG, Cooper C (2001) Use of inhaled corticosteroids and risk of fractures. J Bone Miner Res 16:581–588
de Vries F, Bracke M, Leufkens HG, Lammers JW, Cooper C, van Staa TP (2007) Fracture risk with intermittent high-dose oral glucocorticoid therapy. Arthritis Rheum 56:208–214
Canalis E, Mazziotti G, Giustina A, Bilezikian JP (2007) Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 18:1319–1328
Hofbauer LC, Rauner M (2009) Minireview: live and let die: molecular effects of glucocorticoids on bone cells. Mol Endocrinol 23:1525–1531
van de Berg BC, Malghem J, Lecouvet FE, Devogelaer JP, Maldague B, Houssiau FA (1999) Fat conversion of femoral marrow in glucocorticoid-treated patients: a cross-sectional and longitudinal study with magnetic resonance imaging. Arthritis Rheum 42:1405–1411
Mak W, Shao X, Dunstan CR, Seibel MJ, Zhou H (2009) Biphasic glucocorticoid-dependent regulation of Wnt expression and its inhibitors in mature osteoblastic cells. Calcif Tissue Int 85:538–545
Ohnaka K, Taniguchi H, Kawate H, Nawata H, Takayanagi R (2004) Glucocorticoid enhances the expression of dickkopf-1 in human osteoblasts: novel mechanism of glucocorticoid-induced osteoporosis. Biochem Biophys Res Commun 21:259–264
Wang FS, Ko JY, Yeh DW, Ke HC, Wu HL (2008) Modulation of Dickkopf-1 attenuates glucocorticoid induction of osteoblast apoptosis, adipocytic differentiation, and bone mass loss. Endocrinology 149:1793–1801
Weinstein RS, Jilka RL, Parfitt AM, Manolagas SC (1998) Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids. Potential mechanisms of their deleterious effects on bone. J Clin Invest 102:274–282
Cherian PP, Xia X, Jiang JX (2008) Role of gap junction, hemichannels, and connexin 43 in mineralizing in response to intermittent and continuous application ofparathyroid hormone. Cell Commun Adhes 15:43–54
Plotkin LI, Lezcano V, Thostenson J, Weinstein RS, Manolagas SC, Bellido T (2008) Connexin 43 is required for the anti-apoptotic effect of bisphosphonates on osteocytes and osteoblasts in vivo. J Bone Miner Res 23:1712–1721
Hofbauer LC, Gori F, Riggs BL, Lacey DL, Dunstan CR, Spelsberg TC, Khosla S (1999) Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblastic lineage cells: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology 140:4382–4389
Swanson C, Lorentzon M, Conaway HH, Lerner UH (2006) Glucocorticoid regulation of osteoclast differentiation and expression of receptor activator of nuclear factor-kappaB (NF-kappaB) ligand, osteoprotegerin, and receptor activator of NF-kappaB in mouse calvarial bones. Endocrinology 147:3613–3622
Kim HJ, Zhao H, Kitaura H, Bhattacharyya S, Brewer JA, Muglia LJ, Ross FP, Teitelbaum SL (2006) Glucocorticoids suppress bone formation via the osteoclast. J Clin Invest 116:2152–2160
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17:1–12
World Health Organization (2007) Assessment of osteoporosis at the primary health care level. At: http://www.who.int/chp/topics/Osteoporosis.pdf. Accessed 15 August 2011
Kanis JA on behalf of the World Health Organization Scientific Group (2008) Assessment of osteoporosis at the primary health-care level. Technical Report. WHO Collaborating Centre, University of Sheffield, UK. Available at: http://wwwshefacuk/FRAX/indexhtm. Accessed 15 August 2011
Kanis JA, Johnell O, Oden A, Johansson H, McCloskey EV (2008) FRAX™ and the assessment of fracture probability in men and women from the UK. Osteoporos Int 19:385–397
Kanis JA, Oden A, Johnell O, Johansson H, De Laet C, Brown JP, Burckhardt P, Cooper C, Christiansen C, Cummings S, Eisman JA, Fujiwara S, Glüer C, Goltzman D, Hans D, Krieg MA, La Croix A, McCloskey E, Mellstrom D, Melton LJ 3rd, Pols H, Reeve J, Sanders K, Schott AM, Silman A, Torgerson D, van Staa T, Watts NB, Yoshimura N (2007) The use of clinical risk factors enhances the performance of BMD in the prediction of hip and osteoporotic fractures in men and women. Osteoporos Int 18:1033–1046
Kanis JA, Johnell O, De Laet C, Jonsson B, Oden A, Oglesby A (2002) International variations in hip fracture probabilities: implications for risk assessment. J Bone Miner Res 17:1237–1244
Association Suisse contre l‘Ostéoporose (2010) Ostéoporose: Recommandations 2010. ASCO http://wwwsvgoch/content/documents/SVGO_Empfehlungen2010_V19April2010pdf. Accessed 15 August 2011
Socialstyrelsen (2010) Nationella riktlinjer för rörelseorganens sjukdomar 2010 - stöd för styrning och ledning. Preliminär version. Artikelnr 2010-11-15. Published at www.socialstyrelsen.se. Accessed 15 August 2011.
Compston JE, Cooper A, Cooper C, Francis R, Kanis JA, Marsh D, McCloskey EV, Reid DM, Selby P, Wilkins M, National Osteoporosis Guideline Group (NOGG) (2009) Guidelines for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK. Maturitas 62:105–108
Czerwinski E, Kanis JA, Trybulec B, Johansson H, Borowy P, Osieleniec J (2009) The incidence and risk of hip fracture in Poland. Osteoporos Int 20:1363–1368
Dawson-Hughes B (2008) A revised clinician's guide to the prevention and treatment of osteoporosis. J Clin Endocrinol Metab 93:2463–2465
Fujiwara S, Nakamura T, Orimo H, Hosoi T, Gorai I, Oden A, Johansson H, Kanis JA (2008) Development and application of a Japanese model of the WHO fracture risk assessment tool (FRAX™). Osteoporos Int 19:429–448
Kanis JA, Burlet N, Cooper C, Delmas PD, Reginster JY, Borgstrom F, Rizzoli R, European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) (2008) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 19:399–428
Kanis JA, McCloskey EV, Johansson H, Strom O, Borgstrom F, Oden A, National Osteoporosis Guideline Group (2008) Case finding for the management of osteoporosis with FRAX®—assessment and intervention thresholds for the UK. Osteoporos Int 19:1395–1408, Erratum 2009 Osteoporos Int 20: 499-502
Lippuner K, Johansson H, Kanis JA, Rizzoli R (2010) FRAX® assessment of osteoporotic fracture probability in Switzerland. Osteoporos Int 21:381–390
Neuprez A, Johansson H, Kanis JA, McCloskey EV, Odén A, Bruyère O, Hiligsmann M, Devogelaer JP, Kaufman JM, Reginster JY (2009) Rationalisation du remboursement des médicaments de l’ostéoporose: de la mesure isolée de la densité osseuse à l’intégration des facteurs cliniques de risque fracturaire. Validation de l’algorithme FRAX®. La Revue Médicale de Liège 64:612–619
Papaioannou A, Morin S, Cheung AM, Atkinson S, Brown JP, Feldman S, Hanley DA, Hodsman A, Jamal SA, Kaiser SM, Kvern B, Siminoski K, Leslie WD, Scientific Advisory Council of Osteoporosis Canada (2010) 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ 182:1864–1873
Kanis JA, Johnell O, Committee of Scientific Advisors of the International Osteoporosis Foundation (2005) Requirements for DXA for the management of osteoporosis in Europe. Osteoporos Int 16:229–238
Kanis JA, McCloskey E, Johansson H, Oden A, Leslie WD (2012) FRAX® with and without bone mineral density. Calcif Tissue Int 90:1–13
Leib ES, Saag KG, Adachi JD, Geusens PP, Binkley N, McCloskey EV, Hans DB, FRAX(®) Position Development Conference Members (2011) The impact of the use of glucocorticoids on the estimate by FRAX of the 10 year risk of fracture. J Clin Densitom 14:212–219
van Staa TP, Abenhaim L, Cooper C, Zhang B, Leufkens HG (2001) Public health impact of adverse bone effects of oral corticosteroids. Br J Clin Pharmacol 51:601–607
van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Fracture and oral corticosteroids: relationship to daily and cumulative dose. Rheumatol 39:1383–1389
Kanis JA, Johansson H, Oden A, McCloskey E (2011) Guidance for the adjustment of FRAX according to the dose of glucocorticoids. Osteoporos Int 22:809–816
Kanis JA, Hans D, Cooper C, Baim S, Bilezikian JP, Binkley N, Cauley JA, Compston JE, Dawson-Hughes B, El-Hajj Fuleihan G, Johansson H, Leslie WD, Lewiecki EM, Luckey M, Oden A, Papapoulos SE, Poiana C, Rizzoli R, Wahl DA, McCloskey EV, Task Force of the Frax Initiative (2011) Interpretation and use of FRAX in clinical practice. Osteoporos Int 22:2395–2411
Leslie WD, Lix LM, Johansson H, Oden A, McCloskey E, Kanis JA (2011) Spine-hip discordance and fracture risk assessment: a physician-friendly FRAX enhancement. Osteoporos Int 22:839–847
Nawata H, Soen S, Takayanagi R, Tanaka I, Takaoka K, Fukunaga M, Matsumoto T, Suzuki Y, Tanaka H, Fujiwara S, Miki T, Sagawa A, Nishizawa Y, Seino Y (2005) Guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society for Bone and Mineral Research (2004). J Bone Miner Metab 23:105–109
Agence Française de Sécurité Sanitaire des Produits de Santé (2006) Traitement medicamenteux de l’osteoporose post-ménopausique – Recommandations http://www.afssaps.fr/content/download/7843/78935/version/7/file/ostemrec.pdf (Updated 2008 http://sante-medecine.commentcamarche.net/faq/1118-les-traitementsde-l-osteoporose-post-menopausique-afssaps). Accessed 08 November 2011.
Adami S, Bertoldo F, Brandi M-L, Cepollaro C, Filipponi P, Fiore E, Frediani B, Giannini S, Gonnelli S, Isaia GC, Luisetto G, Mannarino E, Marcocci C, Masi L, Mereu C, Migliaccio S, Minisola S, Nuti R, Rini G, Rossini M, Varenna M, Ventura L, Bianchi G, Società Italiana dell'Osteoporosi, del Metabolismo Minerale e delle Malattie dello Scheletro (2009) Guidelines for the diagnosis, prevention and treatment of osteoporosis. Reumatismo 61:260–284
González Macias J, Guañabens N, Gomez Alonso C, del Rio Barquero L, Munoz Torres M, Delgado M, Perez Edo L, Bernardino Diaz Lopez J, Jodar Gimeno E, Hawkins Carranza F, Comité de Redacción, en representación del Comité de Expertos de la SEIOMM para la elaboración de las Guías (2008) Practice guidelines for postmenopausal, steroid-induced and male osteoporosis. Spanish Society for Bone and Mineral Research. Rev Clin Esp 208(Suppl1):1–24
Dawson-Hughes B, Tosteson AN, Melton LJ, Baim S, Favus MJ, Khosla S, Lindsay RL, National Osteoporosis Foundation Guide Committee (2008) Implications of absolute fracture risk assessment for osteoporosis practice guidelines in the USA. Osteoporos Int 19:449–458
Kanis JA, Borgstrom F, Zethraeus N, Johnell O, Oden A, Jonsson B (2005) Intervention thresholds for osteoporosis in the UK. Bone 36:22–32
European Community (1998) Report on osteoporosis in the European Community. 1998. EC, Strasbourg
Royal College of Physicians (1999) Osteoporosis: clinical guidelines for the prevention and treatment. Royal College of Physicians, London
Kanis JA, Delmas P, Burckhardt P, Cooper C, Torgerson D, European Foundation for Osteoporosis and Bone Disease (1997) Guidelines for diagnosis and management of osteoporosis. Osteoporos Int 7:390–406
Strom O, Borgstrom F, Kanis JA, Compston JE, Cooper C, McCloskey E, Jonsson B (2011) Osteoporosis; burden, health care provision and opportunities in the EU. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 6:59–155
Johansson H, Kanis JA, Oden A, Johnell O, Compston J, McCloskey EV (2011) A comparison of case finding strategies in the UK for the management of hip fractures. Osteoporos Int 23:907–915
Leslie WD, Morin S, Lix LM, Johansson H, Oden A, McCloskey E, Kanis JA, Manitoba Bone Density Program (2012) Fracture risk assessment without bone density measurement in routine clinical practice. Osteoporos Int 23:75–85
Johansson H, Kanis JA, Oden A, Johnell O, McCloskey E (2009) BMD, clinical risk factors and their combination for hip fracture prevention. Osteoporos Int 20:1675–1682
Johansson H, Oden A, Johnell O, Jonsson B, De Laet C, Oglesby A, McCloskey EV, Kayan K, Jalava T, Kanis JA (2004) Optimisation of BMD measurements to identify high risk groups for treatment—a test analysis. J Bone Miner Res 19:906–913
Kanis JA, Adams J, Borgström F, Cooper C, Jönsson B, Preedy D, Selby P, Compston J (2008) The cost-effectiveness of alendronate in the management of osteoporosis. Bone 42:4–15
Leslie WD, Lix LM, Majumdar SR, Johansson H, Oden A, McCloskey E, Kanis JA, Manitoba Bone Density Program (2011) High fracture probability with FRAX® usually indicates densitometric osteoporosis: implications for clinical practice. Osteoporos Int. doi:10.1007/s00198-00011-01592-00193
Kanis JA, Stevenson M, McCloskey EV, Davis S, Lloyd-Jones M (2007) Glucocorticoid-induced osteoporosis: a systematic review and cost-utility analysis. Health Technol Assess 11:1–256
Tosteson AN, Melton LJr, Dawson-Hughes B, Baim S, Favus MJ, Khosla S, Lindsay RL, National Osteoporosis Foundation Guide Committee (2008) Cost-effective osteoporosis treatment thresholds: the United States perspective. Osteoporos Int 19:437–447
Middleton ET, Gardiner ED, Steel SA (2009) Which women should be selected for vertebral fracture assessment? Comparing different methods of targeting VFA. Calcif Tissue Int 85:203–210
Compston J, Reid DM, Boisdron J, Brandi ML, Burlet N, Cahall D, Delmas PD, Dere W, Devogelaer JP, Fitzpatrick LA, Flamion B, Goel N, Korte S, Laslop A, Mitlak B, Ormarsdottir S, Ringe J, Rizzoli R, Tsouderos Y, van Staa T, Reginster JY, Group for the Respect of Ethics and Excellence in Science (2008) Recommendations for the registration of agents for prevention and treatment of glucocorticoid-induced osteoporosis: an update from the Group for the Respect of Ethics and Excellence in Science. Osteoporos Int 19:1247–1250
Adachi JD, Saag KG, Delmas PD, Liberman UA, Emkey RD, Seeman E, Lane NE, Kaufman JM, Poubelle PE, Hawkins F, Correa-Rotter R, Menkes CJ, Rodriguez-Portales JA, Schnitzer TJ, Block JA, Wing J, McIlwain HH, Westhovens R, Brown J, Melo-Gomes JA, Gruber BL, Yanover MJ, Leite MO, Siminoski KG, Nevitt MC, Sharp JT, Malice MP, Dumortier T, Czachur M, Carofano W, Daifotis AG (2001) Two-year effects of alendronate on bone mineral density and vertebral fracture in patients receiving glucocorticoids: a randomized, double-blind, placebo-controlled extension trial. Arthritis Rheum 44:202–211
de Nijs RN, Jacobs JW, Lems WF, Laan RF, Algra A, Huisman AM, Buskens E, de Laet CE, Oostveen AC, Geusens PP, Bruyn GA, Dijkmans BA, Bijlsma JW, Investigators STOP (2006) Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. N Engl J Med 355:675–684
Saag KG, Emkey R, Schnitzer TJ, Brown JP, Hawkins F, Goemaere S, Thamsborg G, Liberman UA, Delmas PD, Malice MP, Czachur M, Daifotis AG (1998) Alendronate for the prevention and treatment of glucocorticoid-induced osteoporosis. Glucocorticoid-Induced Osteoporosis Intervention Study Group. N Engl J Med 339:292–299
Stoch SA, Saag KG, Greenwald M, Sebba AI, Cohen S, Verbruggen N, Giezek H, West J, Schnitzer TJ (2009) Once-weekly oral alendronate 70 mg in patients with glucocorticoid-induced bone loss: a 12-month randomized, placebo-controlled clinical trial. J Rheumatol 36:1705–1714
Yilmaz L, Ozpran K, Gündüz OH, Ucan H, Yücel M (2001) Alendronate in rheumatoid arthritis patients treated with methotrexate and glucocorticoids. Rheumatol Int 20:65–69
Adachi JD, Bensen WG, Brown J, Hanley D, Hodsman A, Josse R, Kendler DL, Lentle B, Olszynski W, Ste-Marie LG, Tenenhouse A, Chines AA (1997) Intermittent etidronate therapy to prevent corticosteroid-induced osteoporosis. N Engl J Med 337:382–387
Cortet B, Hachulla E, Barton I, Bonvoisin B, Roux C (1999) Evaluation of the efficacy of etidronate therapy in preventing glucocorticoid-induced bone loss in patients with inflammatory rheumatic diseases. Rev Rheumatol 66:214–219
Geusens P, Dequecker J, Vanhoof J, Stalmans R, Boonen S, Joly J, Nijs J, Raus J (1998) Cyclical etidronate increases bone density in the spine and hip of postmenopausal women receiving long term corticosteroid treatment. A double blind, randomised placebo controlled study. Ann Rheum Dis 57:724–727
Jenkins EA, Walker-Bone KE, Wood A, McCrae FC, Cooper C, Cawley MI (1999) The prevention of corticosteroid-induced bone loss with intermittent cyclical etidronate. Scand J Rheumatol 28:152–156
Jinnouchi Y (2000) Efficacy of intermittent etidronate therapy for corticosteroid-induced osteoporosis in patients with diffuse connective tissue disease. Kurume Med J 47:219–224
Mulder H, Struys A (1994) Intermittent cyclical etidronate in the prevention of corticosteroid-induced bone loss. Br J Rheumatol 33:348–350
Pitt P, Li F, Todd P, Webber D, Pack S, Moniz C (1998) A double blind placebo controlled study to determine the effects of intermittent cyclical etidronate on bone mineral density in patients on long term oral corticosteroid treatment. Thorax 53:351–356
Roux C, Oriente P, Laan R, Hughes RA, Ittner J, Goemaere S, Di Munno O, Pouillès JM, Horlait S, Cortet B (1998) Randomized trial of effect of cyclical etidronate in the prevention of corticosteroid-induced bone loss. J Clin Endocrinol Metab 83:1128–1133
Skingle SJ, Crisp AJ (1994) Increased bone density in patients on steroids with etidronate. Lancet Infect Dis 344:543–544
Struys A, Snelder AA, Mulder H (1995) Cyclical etidronate reverses bone loss of the spine and proximal femur in patients with established corticosteroid-induced osteoporosis. Am J Med 99:235–242
Cohen S, Levy RM, Keller M, Boling E, Emkey RD, Greenwald M, Zizic TM, Wallach S, Sewell KL, Lukert BP, Axelrod DW, Chines AA (1999) Risedronate therapy prevents corticosteroid-induced bone loss: a twelve month, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum 42:2309–2318
Eastell R, Devogelaer J-P, Peel NFA, Chines AA, Bax DE, Sacco-Gibson N, Nagant de Deuxchaisnes C, Russell RG (2000) Prevention of bone loss with risedronate in glucocorticoid-treated rheumatoid arthritis patients. Osteoporos Int 11:331–337
Reid DM, Adami S, Devogelaer JP, Chines AA (2001) Risedronate increases bone density and reduces vertebral fracture risk within one year in men on corticosteroid therapy. Calcif Tissue Int 69:242–247
Reid DM, Hughes RA, Laan RF, Sacco-Gibson NA, Wenderoth DH, Adami S, Eusebio RA, Devogelaer JP (2000) Efficacy and safety of daily risedronate in the treatment of corticosteroid-induced osteoporosis in men and women: a randomized trial. European Corticosteroid-Induced Osteoporosis Treatment Study. J Bone Miner Res 15:1006–1013
Wallach S, Cohen S, Reid DM, Hughes RA, Hosking DJ, Laan RF, Doherty SM, Maricic M, Rosen C, Brown J, Barton I, Chines AA (2000) Effects of risedronate treatment on bone density and vertebral fracture in patients on corticosteroid therapy. Calcif Tissue Int 67:277–285
Reid DM, Devogelaer JP, Saag K, Roux C, Lau CS, Reginster JY, Papanastasiou P, Ferreira A, Hartl F, Fashola T, Mesenbrink P, Sambrook PN, HORIZON investigators (2009) Zoledronic acid and risedronate in the prevention and treatment of glucocorticoid-induced osteoporosis (HORIZON): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 373:1253–1263
Saag KG, Shane E, Boonen S, Marín F, Donley DW, Taylor KA, Dalsky GP, Marcus R (2007) Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med 357:2028–2039
Saag KG, Zanchetta JR, Devogelaer JP, Adler RA, Eastell R, See K, Krege JH, Krohn K, Warner MR (2009) Effects of teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: thirty-six-month results of a randomized, double-blind, controlled trial. Arthritis Rheum 60:3346–3355
Lakatos P, Nagy Z, Kiss L, Horvath CS, Takacs I, Foldes J, Speer G, Bossanyi A (2000) Prevention of corticosteroid-induced osteoporosis by alfacalcidol. Z Rheumatol 59(suppl 1):48–52
Reginster JY, Kuntz D, Verdickt W, Wouters M, Guillevin L, Menkes CJ, Nielsen K (1999) Prophylactic use of alfacalcidol in corticosteroid-induced osteoporosis. Osteoporos Int 9:75–81
Ringe JD, Cöster A, Meng T, Schacht E, Umbach R (1999) Treatment of glucocorticoid-induced osteoporosis with alfacalcidol/calcium versus vitamin D/calcium. Calcif Tissue Int 65:337–340
Sambrook PN, Kotowicz M, Nash P, Styles CB, Naganathan V, Henderson-Briffa KN, Eisman JA, Nicholson GC (2003) Prevention and treatment of glucocorticoid-induced osteoporosis: a comparison of calcitriol, vitamin D plus calcium, and alendronate plus calcium. J Bone Miner Res 18:919–924
Yamada H (1989) Long-term effect of 1a-hydroxyvitamin D, calcium and thiazide administration on glucocorticoid-induced osteoporosis. Folia Endocrinol Jap 65:603–614
Diamond T, McGuigan I, Schonell M, Levy S, Rae D (1997) A 2 year open randomised controlled trial comparing calcitriol to cyclical etidronate for the treatment of glucocorticoid-induced osteoporosis. J Bone Miner Res 12:S311
Dykman TR, Haralson KM, Gluck OS, Murphy WA, Teitelbaum SL, Hahn TJ, Hahn BH (1984) Effect of oral 1,25-dhydroxyvitamin D and calcium on glucocorticoid-induced osteopenia in patients with rheumatic diseases. Arthritis Rheum 27:1336–1343
Rittmaster RS, Bolognese M, Ettinger MP, Hanley DA, Hodsman AB, Kendler DL, Rosen CJ (2000) Enhancement of bone mass in osteoporotic women with parathyroid hormone followed by alendronate. J Clin Endocrinol Metab 85:2129–2134
Morris HA, Need AG, O'Loughlin PD, Horowitz M, Bridges A, Nordin BE (1990) Malabsorption of calcium in corticosteroid-induced osteoporosis. Calcif Tissue Int 46:305–308
Boutsen Y, Jamart J, Esselinckx W, Devogelaer J-P (2001) Primary prevention of glucocorticoid-induced osteoporosis with intravenous pamidronate and calcium: a prospective controlled 1-year study comparing a single infusion, an infusion given once every 3 months, and calcium alone. J Bone Miner Res 16:104–112
Sambrook P, Birmingham J, Kelly P, Kempler S, Nguyen T, Pocock N, Eisman J (1993) Prevention of corticosteroid osteoporosis. A comparison of calcium, calcitriol, and calcitonin. N Engl J Med 328:1747–1752
Sambrook PN (2000) Corticosteroid osteoporosis: practical implications of recent trials. J Bone Miner Res 15:1645–1649
Holick MF (2007) Optimal vitamin d status for the prevention and treatment of osteoporosis. Drugs Aging 24:1017–1029
Buckley LM, Leib ES, Cartularo KS, Vacek PM, Cooper SM (1996) Calcium and vitamin D3 supplementation prevents bone loss in the spine secondary to low-dose corticosteroids in patients with rheumatoid arthritis. A randomized, double-blind, placebo-controlled trial. Ann Intern Med 125:961–968
Amin S, LaValley MP, Simms RW, Felson DT (1999) The role of vitamin D in corticosteroid-induced osteoporosis: a meta-analytic approach. Arthritis Rheum 42:1740–1751
Homik J, Suarez-Almazor ME, Shea B, Cranney A, Wells G, Tugwell P (2000) Calcium and vitamin D for corticosteroid-induced osteoporosis. Cochrane Database Syst Rev 2:CD000952.
Richy F, Schacht E, Bruyere O, Ethgen O, Gourlay M, Reginster JY (2005) Vitamin D analogs versus native vitamin D in preventing bone loss and osteoporosis-related fractures: a comparative meta-analysis. Calcif Tissue Int 76:176–186
Abrahamsen B, Sahota O (2011) Do calcium plus vitamin D supplements increase cardiovascular risk? BMJ 342:d2080
Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, Clinton SK, Durazo-Arvizu RA, Gallagher JC, Gallo RL, Jones G, Kovacs CS, Mayne ST, Rosen CJ, Shapses SA (2011) The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab 96:53–58
Fleurence RL, Iglesias CP, Johnson JM (2007) The cost-effectiveness of bisphosphonates for the prevention and treatment of osteoporosis: a structured review of the literautre. Pharmacoeconomics 25:913–933
Zethraeus N, Borgstrom F, Strom O, Kanis JA, Jonsson B (2007) Cost-effectiveness of the treatment and prevention of osteoporosis–a review of the literature and a reference model. Osteoporos Int 18:9–23
Beukelman T, Saag KG, Curtis JR, Kilgore ML, Pisu M (2010) Cost-effectiveness of multifaceted evidence implementation programs for the prevention of glucocorticoid-induced osteoporosis. Osteoporos Int 21:1573–1584
Buckley LM, Hillner BE (2003) A cost effectiveness analysis of calcium and vitamin D supplementation, etidronate, and alendronate in the prevention of vertebral fractures in women treated with glucocorticoids. J Rheumatol 30:132–138
Solomon DH, Kuntz KM (2000) Should postmenopausal women with rheumatoid arthritis who are starting corticosteroid treatment be screened for osteoporosis? A cost-effectiveness analysis. Arthritis Rheum 43:1967–1975
van Staa TP, Geusens P, Zhang B, Leufkens HG, Boonen S, Cooper C (2007) Individual fracture risk and the cost-effectiveness of bisphosphonates in patients using oral glucocorticoids. Rheumatol 46:460–466
Ström O, Borgström F, Sen SS, Boonen S, Haentjens P, Johnell O, Kanis JA (2007) Cost-effectiveness of alendronate in the treatment of postmenopausal women in 9 European countries—an economic evaluation based on the fracture intervention trial. Osteoporos Int 18:1047–1061
Borgstrom F, Strom O, Coelho J, Johansson H, Oden A, McCloskey EV, Kanis JA (2010) The cost-effectiveness of risedronate in the UK for the management of osteoporosis using the FRAX. Osteoporos Int 21:495–505
Rizzoli R, Akesson K, Bouxsein M, Kanis JA, Napoli N, Papapoulos S, Reginster JY, Cooper C (2011) Subtrochanteric fractures after long-term treatment with bisphosphonates: a European Society on Clinical and Economic Aspects of Osteoporosis and Osteoarthritis, and International Osteoporosis Foundation Working Group Report. Osteoporos Int 22:373–390
Shane E, Burr D, Ebeling PR, Abrahamsen B, Adler RA, Brown TD, Cheung AM, Cosman F, Curtis JR, Dell R, Dempster D, Einhorn TA, Genant HK, Geusens P, Klaushofer K, Koval K, Lane JM, McKiernan F, McKinney R, Ng A, Nieves J, O'Keefe R, Papapoulos S, Sen HT, van der Meulen MC, Weinstein RS, Whyte M, American Society for Bone and Mineral Research (2010) Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 25:2267–2294
Giusti A, Hamdy NA, Dekkers OM, Ramautar SR, Dijkstra S, Papapoulos SE (2011) Atypical fractures and bisphosphonate therapy: a cohort study of patients with femoral fracture with radiographic adjudication of fracture site and features. Bone 48:966–971
Park-Wyllie LY, Mamdani MM, Juurlink DN, Hawker GA, Gunraj N, Austin PC, Whelan DB, Weiler PJ, Laupacis A (2010) Bisphosphonate use and the risk of subtrochanteric or femoral shaft fractures in older women. JAMA 305:783–789
Schilcher J, Michaëlsson K, Aspenberg P (2011) Bisphosphonate use and atypical fractures of the femoral shaft. N Engl J Med 364:1728–1737
Wang Z, Bhattacharyya T (2011) Trends in incidence of subtrochanteric fragility fractures and bisphosphonate use among the US elderly, 1996-2007. J Bone Miner Res 26:553–560
Armamento-Villareal R, Napoli N, Diemer K, Watkins M, Civitelli R, Teitelbaum S, Novack D (2009) Bone turnover in bone biopsies of patients with low-energy cortical fractures receiving bisphosphonates: a case series. Calcif Tissue Int 85:37–44
Edwards MH, McCrae FC, Young-Min SA (2010) Alendronate-related femoral diaphysis fracture-what should be done to predict and prevent subsequent fracture of the contralateral side? Osteoporos Int 21:701–703
Goh SK, Yang KY, Koh JS, Wong MK, Chua SY, Chua DT, Howe TS (2007) Subtrochanteric insufficiency fractures in patients on alendronate therapy: a caution. J Bone Joint Surg Br 89:349–353
Ing-Lorenzini K, Desmeules J, Plachta O, Suva D, Dayer P, Peter R (2009) Low-energy femoral fractures associated with the long-term use of bisphosphonates: a case series from a Swiss university hospital. Drug Saf 32:775–785
Kwek EB, Goh SK, Koh JS, Png MA, Howe TS (2008) An emerging pattern of subtrochanteric stress fractures: a long-term complication of alendronate therapy? Injury 39:224–231
Odvina CV, Zerwekh JE, Rao DS, Maalouf N, Gottschalk FA, Pak CY (2005) Severely suppressed bone turnover: a potential complication of alendronate therapy. J Clin Endocrinol Metab 90:1294–1301
Visekruna M, Wilson D, McKiernan FE (2008) Severely suppressed bone turnover and atypical skeletal fragility. J Clin Endocrinol Metab 93:2948–2952
Khan AA, Sándor GK, Dore E, Morrison AD, Alsahli M, Amin F, Peters E, Hanley DA, Chaudry SR, Lentle B, Dempster DW, Glorieux FH, Neville AJ, Talwar RM, Clokie CM, Mardini MA, Paul T, Khosla S, Josse RG, Sutherland S, Lam DK, Carmichael RP, Blanas N, Kendler D, Petak S, Ste-Marie LG, Brown J, Evans AW, Rios L, Compston JE, Canadian Taskforce on Osteonecrosis of the Jaw (2009) Bisphosphonate associated osteonecrosis of the jaw. J Rheumatol 36:478–490
Khosla S, Burr D, Cauley J, Dempster DW, Ebeling PR, Felsenberg D, Gagel RF, Gilsanz V, Guise T, Koka S, McCauley LK, McGowan J, McKee MD, Mohla S, Pendrys DG, Raisz LG, Ruggiero SL, Shafer DM, Shum L, Silverman SL, Van Poznak CH, Watts N, Woo S-B, Shane E (2007) Bisphosphonate–associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 22:1479–1491
Durie BG, Katz M, Crowley J (2005) Osteonecrosis of the jaw and bisphosphonates. N Engl J Med 353:99–102
Lazarovici TS, Yahalom R, Taicher S, Elad S, Hardan I, Yarom N (2009) Bisphosphonate-related osteonecrosis of the jaws: a single-center study of 101 patients. J Oral Maxillofac Surg 67:850–855
Woo SB, Hellstein JW, Kalmar JR (2006) Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws. Ann Intern Med 144:753–761
Patlas N, Golomb G, Yaffe P, Pinto T, Breuer E, Ornoy A (1999) Transplacental effects of bisphosphonates on fetal skeletal ossification and mineralization in rats. Teratology 60:68–73
Losada I, Sartori L, Di Gianantonio E, Zen M, Clementi M, Doria A (2010) Bisphosphonates in patients with autoimmune rheumatic diseases: can they be used in women of childbearing age? Autoimmun Rev 9:547–552
Levy S, Fayez I, Taguchi N, Han JY, Aiello J, Matsui D, Moretti M, Koren G, Ito S (2009) Pregnancy outcome following in utero exposure to bisphosphonates. Bone 44:428–430
Ornoy A, Wajnberg R, Diav-Citrin O (2006) The outcome of pregnancy following pre-pregnancy or early pregnancy alendronate treatment. Reprod Toxicol 22:578–579
Baim S, Wilson CR, Lewiecki EM, Luckey MM, Downs RJ, Lentle BC (2005) Precision assessment and radiation safety for dual-energy X-ray absorptiometry: position paper of the International Society for Clinical Densitometry. J Clin Densitom 8:371–378
Frost SA, Nguyen ND, Center JR, Eisman JA, Nguyen TV (2009) Timing of repeat BMD measurements: development of an absolute risk-based prognostic model. J Bone Miner Res 24:1800–1807
Gluer CC, Blake G, Lu Y, Blunt BA, Jergas M, Genant HK (1995) Accurate assessment of precision errors: how to measure the reproducibility of bone densitometry techniques. Osteoporos Int 19:1395–1408, Erratum 2009 Osteoporos Int 20: 499-502 5:262-270
Cummings SR, Karpf DB, Harris F, Genant HK, Ensrud K, LaCroix AZ, Black DM (2002) Improvement in spine bone density and reduction in risk of vertebral fractures during treatment with antiresorptive drugs. Am J Med 112:281–289
Hodgson SF, Watts NB, Bilezikian JP, Clarke BL, Gray TK, Harris DW, Johnston CC, Kleerekoper M, Lindsay R, Luckey MM, McClung MR, Nankin HR, Petak SM, Recker RR, AACE Osteoporosis Task Force (2003) American Association of Clinical Endocrinologists medical guidelines for clinical practice for the prevention and treatment of postmenopausal osteoporosis: 2001 edition, with selected updates for 2003. Endocr Pract 9:544–564
Lenchik L, Kiebzak GM, Blunt BA (2002) What is the role of serial bone mineral density measurements in patient management? J Clin Densitom 5:S29–38
Watts NB, Lewiecki EM, Bonnick SL, Laster AJ, Binkley N, Blank RD, Geusens PP, Miller PD, Petak SM, Recker RR, Saag KG, Schousboe J, Siris ES, Bilezikian JP (2009) Clinical value of monitoring BMD in patients treated with bisphosphonates for osteoporosis. J Bone Miner Res 24:1643–1646
Vasikaran S, Eastell R, Bruyère O, Foldes AJ, Garnero P, Griesmacher A, McClung M, Morris HA, Silverman S, Trenti T, Wahl DA, Cooper C, Kanis JA, IOF-IFCC Bone Marker Standards Working Group (2011) Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int 22:391–420
Eastell R, Krege JH, Chen P, Glass EV, Reginster JY (2006) Development of an algorithm for using PINP to monitor treatment of patients with teriparatide. Curr Med Res Opin 22:61–66
Kumagai S, Kawano S, Atsumi T, Inokuma S, Okada Y, Kanai Y, Kaburaki J, Kameda H, Suwa A, Hagiyama H, Hirohata S, Makino H, Hashimoto H (2005) Vertebral fracture and bone mineral density in women receiving high dose glucocorticoids for treatment of autoimmune diseases. J Rheumatol 32:863–869
Kaji H, Yamauchi M, Chihara K, Sugimoto T (2008) Glucocorticoid excess affects cortical bone geometry in premenopausal, but not postmenopausal, women. Calcif Tissue Int 82:182–190
Kaji H, Yamauchi M, Yamaguchi T, Sugimoto T (2010) Urinary deoxypyridinoline is a BMD-independent marker for prevalent vertebral fractures in postmenopausal women treated with glucocorticoid. Osteoporos Int 21:1585–1590
Honkanen R, Tuppurainen M, Kroger H, Alhava E, Puntila E (1997) Associations of early premenopausal fractures with subsequent fractures vary by sites and mechanisms of fractures. Calcif Tissue Int 60:327–331
Hosmer WD, Genant HK, Browner WS (2002) Fractures before menopause: a red flag for physicians. Osteoporos Int 13:337–341
Horowitz M, Wishart JM, Bochner M, Need AG, Chatterton BE, Nordin BE (1988) Mineral density of bone in the forearm in premenopausal women with fractured wrists. BMJ 297:1314–1315
Armamento-Villareal R, Villareal DT, Avioli LV, Civitelli R (1992) Estrogen status and heredity are major determinants of premenopausal bone mass. J Clin Invest 90:2464–2471
Cohen A, Fleischer J, Freeby MJ, McMahon DJ, Irani D, Shane E (2009) Clinical characteristics and medication use among premenopausal women with osteoporosis and low BMD: the experience of an osteoporosis referral center. J Womens Health 18:79–84
Moreira Kulak CA, Schussheim DH, McMahon DJ, Kurland E, Silverberg SJ, Siris ES, Bilezikian JP, Shane E (2000) Osteoporosis and low bone mass in premenopausal and perimenopausal women. Endocr Pract 6:296–304
Peris P, Guañabens N, Martínez J, de Osaba M, Monegal A, Alvarez L, Pons F, Ros I, Cerdá D, Muñoz-Gómez J (2002) Clinical characteristics and etiologic factors of premenopausal osteoporosis in a group of Spanish women. Semin Arthritis Rheum 32:64–70
Blum M, Harris SS, Must A, Phillips SM, Rand WM, Dawson-Hughes B (2001) Weight and body mass index at menarche are associated with premenopausal bone mass. Osteoporos Int 12:588–594
Hawker GA, Jamal SA, Ridout R, Chase C (2002) A clinical prediction rule to identify premenopausal women with low bone mass. Osteoporos Int 13:400–406
Sugiyama T, Suzuki S, Yoshida T, Suyama K, Tanaka T, Sueishi M, Tatsuno I (2010) Incidence of symptomatic vertebral fractures in women of childbearing age newly treated with high-dose glucocorticoid. Gend Med 7:218–229
Tatsuno I, Sugiyama T, Suzuki S, Yoshida T, Tanaka T, Sueishi M, Saito Y (2009) Age dependence of early symptomatic vertebral fracture with high-dose glucocorticoid treatment for collagen vascular diseases. J Clin Endocrinol Metab 94:1671–1677
Yirmiya R, Bab I (2009) Major depression is a risk factor for low bone mineral density: a meta-analysis. Biol Psychiatry 66:423–432
Solomon DH, Katz JN, Jacobs JP, La Tourette AM, Coblyn J (2002) Management of glucocorticoid-induced osteoporosis in patients with rheumatoid arthritis: rates and predictors of care in an academic rheumatology practice. Arthritis Rheum 46:3136–3142
Roux C, Reid DM, Devogelaer JP, Saag K, Lau CS, Reginster JY, Papanastasiou P, Bucci-Rechtweg C, Su G, Sambrook PN (2011) Post hoc analysis of a single IV infusion of zoledronic acid versus daily oral risedronate on lumbar spine bone mineral density in different subgroups with glucocorticoid-induced osteoporosis. Osteoporos Int. doi:10.1007/s00198-00011-01800-00191
Langdahl BL, Marin F, Shane E, Dobnig H, Zanchetta JR, Maricic M, Krohn K, See K, Warner MR (2009) Teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: an analysis by gender and menopausal status. Osteoporos Int 20:2095–2104
Yeap SS, Fauzi AR, Kong NC, Halim AG, Soehardy Z, Rahimah I, Chow SK, Goh EM (2008) A comparison of calcium, calcitriol, and alendronate in corticosteroid-treated premenopausal patients with systemic lupus erythematosus. J Rheumatol 35:2344–2347
Okada Y, Nawata M, Nakayamada S, Saito K, Tanaka Y (2008) Alendronate protects premenopausal women from bone loss and fracture associated with high-dose glucocorticoid therapy. J Rheumatol 35:2249–2254
Nakayamada S, Okada Y, Saito K, Tanaka Y (2004) Etidronate prevents high dose glucocorticoid induced bone loss in premenopausal individuals with systemic autoimmune diseases. J Rheumatol 31:163–166
Sato S, Ohosone Y, Suwa A, Yasuoka H, Nojima T, Fujii T, Kuwana M, Nakamura K, Mimori T, Hirakata M (2003) Effect of intermittent cyclical etidronate therapy on corticosteroid induced osteoporosis in Japanese patients with connective tissue disease: 3 year follow-up. J Rheumatol 30:2673–2679
Sato S, Takada T, Katsuki Y, Kimura N, Kaneko Y, Suwa A, Hirakata M, Kuwana M (2008) Longterm effect of intermittent cyclical etidronate therapy on corticosteroid-induced osteoporosis in Japanese patients with connective tissue disease: 7-year followup. J Rheumatol 35:142–146
Mok CC, Tong KH, To CH, Siu YP, Ma KM (2008) Risedronate for prevention of bone mineral density loss in patients receiving high-dose glucocorticoids: a randomized double-blind placebo-controlled trial. Osteoporos Int 19:357–364
Fujii N, Hamano T, Mikami S, Nagasawa Y, Isaka Y, Moriyama T, Horio M, Imai E, Hori M, Ito T (2007) Risedronate, an effective treatment for glucocorticoid-induced bone loss in CKD patients with or without concomitant active vitamin D (PRIUS-CKD). Nephrol Dial Transplant 22:1601–1607
Kikuchi Y, Imakiire T, Yamada M, Saigusa T, Hyodo T, Kushiyama T, Higashi K, Hyodo N, Yamamoto K, Suzuki S, Miura S (2007) Effect of risedronate on high-dose corticosteroid-induced bone loss in patients with glomerular disease. Nephrol Dial Transplant 22:1593–1600
Lambrinoudaki I, Chan DT, Lau CS, Wong RW, Yeung SS, Kung AW (2000) Effect of calcitriol on bone mineral density in premenopausal Chinese women taking chronic steroid therapy. A randomized, double blind, placebo controlled study. J Rheumatol 27:1759–1765
Bernstein CN, Seeger LL, Anton PA, Artinian L, Geffrey S, Goodman W, Belin TR, Shanahan F (1996) A randomized, placebo-controlled trial of calcium supplementation for decreased bone density in corticosteroid-using patients with inflammatory bowel disease: a pilot study. Aliment Pharmacol Ther 10:777–786
McDonald CF, Zebaze RM, Seeman E (2006) Calcitriol does not prevent bone loss in patients with asthma receiving corticosteroid therapy: a double-blind placebo-controlled trial. Osteoporos Int 17:1546–1551
Kung AW, Chan TM, Lau CS, Wong RW, Yeung SS (1999) Osteopenia in young hypogonadal women with systemic lupus erythematosus receiving chronic steroid therapy: a randomized controlled trial comparing calcitriol and hormonal replacement therapy. Rheumatology (Oxford) 38:1239–1244
Acknowledgments
We are grateful to Amanda Sherwood and Heidi-Mai Warren from the European Calcified Tissue Society and Judy Stenmark from the International Osteoporosis Foundation, for administrative support. This paper has been consulted by the IOF Committee of National Societies and the following associations: EFFORT, AO Trauma, International Society of Orthopaedic Surgery and Traumatology, Endocrine Society, International Society of Endocrinology, AACE, ILAR, EULAR, ACR, International Menopause Society, International Association of Gerontology and Geriatrics, European Union Geriatric Medicine Society, American Geriatrics Society and ASBMR.
Conflict of interest
Adachi J.D. is consultant/speaker for Amgen, Eli Lilly, GSK, Merck, Novartis, Pfizer, Procter & Gamble, Roche, Sanofi Aventis and Warner Chilcott and received grants for clinical trials from Amgen, Bristol-Myers Squibb, Eli Lilly, Merck, Novartis, Pfizer, Procter & Gamble, Sanofi Aventis, Roche and Warner Chilcott.
Agnusdei D. is a stock holder and employee of Eli Lilly and Company.
Bilezikian J.P. receives consultancies and speaker fees for Amgen, Lilly, Merck and Novartis; consultancies for Radius Pharmaceuticals, NPS Pharmaceuticals and GSK; and research support from NPS Pharmaceuticals.
Boonen S. receives research support and/or consulting fees from Amgen, Merck, Novartis, Sanofi Aventis and Warner Chilcott.
Cooper C. receives lecture fees and consultancy from ABBH, Amgen, Eli Lilly, GSK/Roche, MSD, Novartis, Nycomed and Servier.
Diez Perez A. receives honoraria as consultant or speaker from Amgen, Eli Lilly and MSD as well as research funds from Alliance of Bone Health and Servier.
Eastell R. is a member of the Procter & Gamble National Advisory Board, Medical Research Council, European Calcified Tissue Society, Biobank MSK Advisory Board; a consultant for Amgen, AstraZeneca, GlaxoSmithKline, Medtronics, Nastech, Nestle, Fonterra Brands, Novartis, Ono Pharma, Osteologix, Pfizer, Eli Lilly, Sanofi Aventis, Tethys, Unilever, Unipath, Inverness Medical, Johnson & Johnson, SPD and MSD and IDS; received grants from AstraZeneca, Unilever, Amgen, Department of Health, National Institute for Health Research, Sheffield Teaching Hospitals Trust, Queen Mary University of London, Procter & Gamble, Unipath Limited, Novartis, Medical Research Council UK, Arthritis Research UK, Nestle Foundation and Sanofi Aventis; received payment for lectures including service on speakers bureaus from Takeda, Eli Lilly, Amgen, National Osteoporosis Foundation, Procter & Gamble, GlaxoSmithKline Nutrition and Roche; received travel, accommodations or meeting expenses from Novartis, Procter & Gamble, GlaxoSmithKline Nutrition, Fonterra Brands, Amgen, AstraZeneca, Medtronics, Unilever, Ono Pharma, Servier, Tethys, Eli Lilly, SPD and International Osteoporosis Foundation.
Kanis J.A. receives consulting fees, paid advisory boards, lecture fees and/or grant support from the majority of companies concerned with skeletal metabolism.
Langdahl B. is a member of the advisory board of Amgen, Lilly, Nycomed and MSD and the speakers bureau of Lilly, Amgen and MSD; and received research funding from Lilly, Novartis, MSD and Amgen.
McCloskey E. receives research funding and/or advisory board and/or speaker's fees from the following: Amgen, AstraZeneca, GSK, Hologic, Innovus, Lilly, MSD, Novartis, Pfizer, Roche, Servier, Tethys and Warner Chilcott.
Napoli N. receives consultancy and lecture fees from MSD.
Ralston S. received research grants from Amgen and his employer received consultancy income from Amgen, Merck and Novartis.
Silverman S. is a member of the speaker's bureau for Amgen, Lilly, Pfizer and Roche Pharmaceuticals; a consultant for Amgen, Lilly, Novartis, Pfizer, Roche Pharmaceuticals, Roche Diagnostics and Warner Chilcott; and received research support from Alliance for Better Bone Health, Lilly and Pfizer.
Compston J.E. receives consultancy fees and lecture fees, and/or received grant support from the Alliance for Better Bone Health, Amgen, Eli Lilly, GlaxoSmithKline, Medtronic, MSD, Novartis, Nycomed, Procter & Gamble, Sanofi Aventis, Servier and Warner Chilcott.
Borgstrom F., Hofbauer L., Lekamwasam S., Lesnyak O., Lorenc R., Messina O.D., Obermayer-Pietsch B., Stepan J., Sambrook P., Sosa M., Suppan G. and Wahl D.A. declare no conflict of interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
These guidelines have been endorsed by the Committee of Scientific Advisors of the IOF and the ECTS Board and Professional Practice Committee. An appendix to these guidelines can be found in Archives of Osteoporosis (DOI 10.1007/s11657-012-0070-7).
Rights and permissions
About this article
Cite this article
Lekamwasam, S., Adachi, J.D., Agnusdei, D. et al. A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos Int 23, 2257–2276 (2012). https://doi.org/10.1007/s00198-012-1958-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-012-1958-1