Abstract
Acute kidney injury (AKI) is associated with persistent renal dysfunction, the receipt of dialysis, dialysis dependence, and mortality. Accordingly, the concept of major adverse kidney events (MAKE) has been adopted as an endpoint for assessing the impact of AKI. However, applied criteria or observation periods for operationalizing MAKE appear to vary across studies. To evaluate this heterogeneity for MAKE evaluation, we performed a systematic scoping review of studies that employed MAKE as an AKI endpoint. Four major academic databases were searched, and we identified 122 studies with increasing numbers over time. We found marked heterogeneity in applied criteria and observation periods for MAKE across these studies, with some even lacking a description of criteria. Moreover, 13 different observation periods were employed, with 30 days and 90 days as the most common. Persistent renal dysfunction was evaluated by estimated glomerular filtration rate (34%) or serum creatinine concentration (48%); however, 37 different definitions for this component were employed in terms of parameters, cut-off criteria, and assessment periods. The definition for the dialysis component also showed significant heterogeneity regarding assessment periods and duration of dialysis requirement (chronic vs temporary). Finally, MAKE rates could vary by 7% [interquartile range: 1.7-16.7%] with different observation periods or by 36.4% with different dialysis component definitions. Our findings revealed marked heterogeneity in MAKE definitions, particularly regarding component assessment and observation periods. Dedicated discussion is needed to establish uniform and acceptable standards to operationalize MAKE in terms of selection and applied criteria of components, observation period, and reporting criteria for future trials on AKI and related conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This scoping review revealed substantial heterogeneity in applied criteria for each component and observation periods to evaluate major adverse kidney events (MAKE) across existing articles. This heterogeneity potentially leads to inconsistent results as well as difficulties in comparing or integrating renal outcome data among studies, underscoring the need for dedicated discussion for establishing uniform and acceptable standards to operationalize MAKE for future trials. |
Introduction
Acute kidney injury (AKI) is a common complication among critically ill patients and is associated with high morbidity and mortality [1,2,3]. The diagnosis and staging of AKI have been standardized through the consensus criteria of the Kidney Disease Improving Global Outcomes (KDIGO) 2012 guidelines [4]. Such AKI diagnosis and staging criteria have helped clinicians understand and compare AKI epidemiology. Similarly, long-term outcomes in AKI patients regarding progressive renal functional decline or new onset of chronic kidney disease (CKD) have been investigated by many studies using consensus definitions of both AKI and CKD. Finally, to create a bridge from the definition of AKI to the definition of CKD, the Acute Disease Quality Initiative developed a consensus definition of acute kidney disease (AKD) [5]. Thus, several epidemiological studies have now reported the impact of AKD on outcomes [6, 7]. These studies have demonstrated that AKI, AKD, and CKD are closely connected and are part of a continuum of renal injury across a patient’s illness [8]
Linked with the above developments, the concept of major adverse kidney events (MAKE) has been proposed as analogous to that of major adverse cardiac events (MACE) [9, 10], a concept that has been successfully used in cardiovascular research [11, 12]. MAKE has been defined as a composite outcome comprising death, dialysis dependence, and persistent renal dysfunction, which are all patient-centered and related to AKI, AKD, subsequent CKD development, and mortality [13,14,15]. Importantly, MAKE components are relatively easily measured, subject to limited confounding, indicative of major morbidity, and potentially applicable in a consistent manner across all patient populations [16].
However, despite the above potential advantages, uniform and acceptable criteria for MAKE operationalization have yet to be established. Thus, different definitions of MAKE appear to have been used across studies, suggesting the need for a systematic assessment of current practice and evidence. The aim of such assessment is to quantify such heterogeneity, identify which MAKE elements are affected, and help develop a common language in relation to this outcome measure.
Accordingly, we conducted a systematic scoping review to investigate the heterogeneity in applied criteria in MAKE components and observation periods used to evaluate those components across existing studies in acute critical settings and to assess the impact of this heterogeneity on MAKE rates.
Methods
Study design
We conducted a systematic scoping review with adherence to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline and its extension for scoping reviews [17]. The study protocol was developed following the Joanna Briggs Institute (JBI) Manual for Evidence Synthesis [18] and registered with the Open Science Framework (https://osf.io/thqp8).
Eligibility criteria
We included all randomized controlled trials (RCTs), non-RCTs, and prospective or retrospective cohort studies that employed MAKE as endpoints in acute critical care settings to systematically evaluate current heterogeneity in MAKE definitions. We included studies reported as full text in peer-reviewed journals. Reviews, editorials, conference articles, comments, standalone abstracts, and nonhuman studies were excluded.
Search strategy
The databases of Medical Literature Analysis and Retrieval System Online (MEDLINE), Excerpta Medica Database (Embase), Web of Science, and Cochrane Central Register of Controlled Trials were searched to retrieve relevant articles from the database inception to November 3, 2023. Supplementary Appendix I summarizes the search strategies used for each database. In addition, the reference lists of all included articles and recent relevant reports or reviews were manually searched.
Study selection and data extraction
Two authors (AM and RI) independently screened the titles and abstracts of all articles retrieved using Covidence. In the case of disagreements between the two authors, a consensus was reached via discussion with a third reviewer (KD). With eligible or potentially eligible/unclear articles after this initial screening, we retrieved full texts and assessed them for eligibility. We determined study eligibility and recorded reasons for exclusion of the studies. We extracted the title, year of publication, study design, the observation periods for MAKE evaluation, and the applied criteria for each MAKE component. Applied criteria for each MAKE component were determined through a consensus between the abovementioned two authors with any disagreements referred to the third reviewer for further discussion and resolution.
Description of the subsets of articles
We utilized several subsets of articles for further description. The first subset included articles that reported MAKE rates for multiple observation periods under consistent MAKE criteria within each article. This subset was used to evaluate the impact of observation periods on MAKE rates. The second subset included articles that only included patients treated with renal replacement therapy (RRT) and that employed chronic dialysis or dialysis dependence as the criteria for the dialysis component of MAKE and was used to evaluate the impact of the variation in the MAKE dialysis component on MAKE rates. Additionally, we utilized subsets of articles stratified by i) study type, ii) patient population, iii) single or multicentre, and iv) AKI for inclusion or as an outcome to describe the variation in MAKE operationalization based on study characteristics.
Statistical analysis
Descriptive statistics were used to characterize included studies and to summarize key findings. Frequencies and percentages were calculated to report the distribution of study characteristics. Visualization of trends, including histogram, bar chart, and alluvial plot, was also employed to illustrate the ranges and distributions of publication years and applied criteria for each MAKE component. All statistical analyses were performed using R version 4.2.1 (R Core Team. 2022 R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.) with the packages 'tidyverse', 'ggpubr', 'easyalluvial', 'ggstatplot [19]', and 'tableone'.
Results
Characteristics of included articles
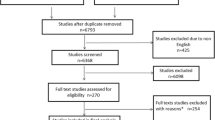
The database search retrieved 692 citations, and 204 full-text articles were assessed for eligibility. Among these, 82 articles were excluded for a variety of reasons and 122 articles met our eligibility criteria (Fig. 1).
The characteristics of the included articles are summarized in Table 1. The most common study design was that of retrospective cohort studies (74 articles, 60.7%), [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92] followed by prospective cohort studies (35 articles, 28.7%) [93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127] and RCTs (13 articles, 10.7%) [128,129,130,131,132,133,134,135,136,137,138,139,140]. Regarding each MAKE component, all three components of MAKE (death, dialysis, and persistent renal dysfunction) were included in the majority of articles (119 articles, 97.5%). However, death was not included in two studies (1.6%), [47, 86] and information on its inclusion was not reported in another study (0.8%). [76] Dialysis was not included in one MAKE study (0.8%), [81] and information on its inclusion was not reported in another study (0.8%). [76] Moreover, information on the inclusion of persistent renal dysfunction was not reported in five studies (4.1%). [28, 76, 94, 96, 112] Additionally, three articles incorporated hospitalization as a MAKE component. Two of these articles [131, 140] employed rehospitalizations for AKI and the remaining article [137] employed rehospitalization with cause unspecified. Finally, as shown in Fig. 2, the annual number of articles using MAKE as endpoints has increased annually, surpassing 20 articles/year as of 2021.
Number of articles using MAKE as endpoints. This bar histogram shows the annual number of articles using MAKE as an endpoint from each year, with bars representing the number of articles and colored by study design. Data are presented from full calendar years except 2023, which includes articles through November 3rd. Cluster randomized controlled trials are included under Randomized controlled trials and crossover trials are included under Prospective cohort studies
Three articles employed rehospitalization as a MAKE component: two articles with AKI-related rehospitalization and one with rehospitalization (with cause unspecified)
Observation period for MAKE evaluation
Table 2 describes the observation periods for MAKE evaluation. Most articles (104 articles) evaluated MAKE at a single moment in time, while 18 articles [22, 27, 43, 46, 48, 92, 94, 97, 103, 108, 109, 112, 117, 129, 133, 135, 138, 140] evaluated MAKE at multiple time points. Overall, thirteen different observation periods were employed across articles, with 30 days (27.9% in single period articles, 55.6% in multiple period articles) and 90 days (22.1% in single period articles, 77.8% in multiple period articles) as the most frequently used periods for evaluation.
Time-to-MAKE analysis over a given observation period was also utilized in a proportion of single (18 articles, 17.3%) [20, 24,25,26, 38, 39, 47, 56, 58, 66, 68, 83, 86, 96, 107, 113, 119, 123] and multiple (2 articles, 11.1%) [46, 103] period articles. Observation periods were not reported in five articles (4.8%) that used a single period evaluation. [35, 76, 80, 90, 104] Fig. 3 described observation periods in articles that used a single period evaluation based on the study type. RCTs were more likely to employ 90 days or longer (7 articles, 87.5%) compared to prospective studies (9 articles, 32.1%) or retrospective studies (21 articles, 30.9%). The observation periods employed in articles with single or multiple period evaluations were described based on study characteristics, as presented in supplementary Tables S1 and S2.
Additionally, we assessed another subset of articles from this scoping review cohort that reported MAKE rates for multiple observation periods (n = 13). [22, 27, 43, 48, 92, 94, 108, 109, 117, 129, 133, 135, 138] This analysis revealed that the median of the largest difference in reported MAKE rates resulting from different observation periods within each article was 7% [interquartile range (IQR): 1.7–16.7%] (supplementary Table S3).
Applied criteria for death component
Among 119 articles that included death as a MAKE component, 116 articles employed death from any cause or death with cause unspecified, leaving three articles [24, 38, 119] specifically defined death as “death from renal failure”, “death due to kidney disease”, or “death while receiving dialysis”, respectively.
Applied criteria for persistent renal dysfunction component
Figure 4 and supplementary Table S4 show the parameters used in applied criteria for persistent renal dysfunction, one of the MAKE components, in the included articles. Most commonly used parameters for defining persistent renal dysfunction were serum creatinine concentration (59 articles, 48%) [21, 23, 27, 31,32,33,34, 36, 37, 39, 40, 42, 43, 45, 49, 50, 52, 53, 55, 59,60,61, 63, 65, 70,71,72,73,74,75, 77, 79, 81, 84, 88, 89, 93, 95, 98, 99, 102, 105, 110, 111, 113,114,115,116,117,118, 122, 123, 126,127,128,129, 132, 136, 138] and estimated glomerular filtration rate (eGFR) (41 articles, 34%) [20, 22, 24,25,26, 29, 30, 35, 38, 44, 46, 47, 51, 56, 57, 62, 64, 67, 68, 78, 82, 83, 85, 87, 100, 101, 106,107,108,109, 119,120,121, 125, 133,134,135, 137, 139,140,141]. The criteria for persistent renal dysfunction were not described in 7 studies (6%). [28, 66, 76, 94, 96, 112, 131] In addition, as shown in supplementary Table S5, we identified 37 different definitions based on variations in the parameters used, cut-off values, and assessment periods, even when the same parameters were used in the criteria for this component. Furthermore, when defining baseline creatinine for the application of MAKE criteria, most of the included articles (95 articles, 77.9%) used pre-study creatinine values, but also estimated creatinine and/or study-derived creatinine as shown in supplementary Fig. S1. Finally, in one study [139] in cardiac surgery patients with risk factors for AKI, the MAKE90 rate using eGFR based on creatinine was 20.1%, whereas employing eGFR based on cystatin C yielded a higher rate of 40.5%.
Applied criteria for dialysis component
Figure 5 and supplementary Table S6 show the applied criteria for the dialysis component of MAKE across 122 articles with 150 different observation periods. While 43 articles (35.2%) applied criteria for this component using the performance of chronic dialysis or RRT dependence assessed at the specific study endpoint, 51 articles (41.8%) applied it as the receipt of any RRT at any time until the end of the observation period [20,21,22,23,24,25,26, 28,29,30,31,32,33,34, 37, 39,40,41,42, 45, 46, 48,49,50,51,52,53,54,55, 57,58,59,60,61,62, 64,65,66,67, 69,70,71,72,73,74,75, 77,78,79, 81,82,83,84,85,86,87,88, 92, 93, 95,96,97, 99,100,101,102,103, 105, 107,108,109,110, 115,116,117,118, 120,121,122,123, 125,126,127,128,129,130,131,132, 134,135,136, 138, 139, 141] and two articles (1.6%) employed combined criteria of both for this component. [137, 140] Moreover, the criteria for the dialysis component varied according to the duration of the observation period [20,21,22, 24,25,26,27,28, 31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80, 83,84,85,86,87, 89,90,91,92,93,94,95,96,97,98, 100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120, 122,123,124,125,126,127,128,129,130,131,132,133,134,135,136, 138, 139, 141] such that among the 48 articles using an observation period of ≤ 30 days, 32 (66.7%) applied it as any RRT at any time until the study endpoint, while seven (14.6%) applied it as chronic dialysis or RRT dependence at the specific study endpoint. In contrast, in the 54 articles with an observation period of > 30 days, 18 (33.3%) applied the dialysis component as any RRT at any time until the endpoint, and 24 (44.4%) applied it as chronic dialysis or RRT dependence at the specific study endpoint. Similarly, in the 20 articles employing Time-to-MAKE analysis, only two (10%) applied the former criteria of any RRT at any time, while 12 (60%) applied the latter of dialysis dependence at specific endpoint. Finally, 13 articles with 20 observation periods did not describe whether RRT was limited to chronic dialysis or not and 23 articles with 30 observation periods did not report the assessment window for RRT.
Variation in definitions for dialysis component. This alluvial plot illustrates the heterogeneity in the criteria and assessment window applied to evaluate the dialysis component of MAKE across study publications. Each flow represents publications applying a criterion for dialysis component in terms of RRT modality and assessment window, with flow color indicating the observation period for MAKE evaluation and flow width representing the number of publications. Publications evaluating MAKE at multiple observation periods are represented repeatedly in the plot, comprising 122 publications with 146 observation periods. MAKE definitions varied substantially across studies. Articles with observation periods of ≤ 30 days more often applied a criterion for this component as any use of RRT at any time until the study endpoint (66.7%) rather than chronic dialysis or RRT dependence at the endpoint (14.6%). In contrast, for articles with > 30 days observation periods or employing Time-to-MAKE analyses, a higher proportion used criteria of chronic dialysis or RRT dependence at the final assessment endpoint (44.4% and 60.0%, respectively) rather than any RRT until that endpoint (33.3% and 10.0%). Additionally, 10.7% of articles did not describe the RRT modality and 18.9% did not describe the assessment window. One article employed combined criteria of chronic and temporary dialysis that could not be distinctly classified within the categories shown, and was thus excluded from this visualization. ICU intensive care unit, MAKE major adverse kidney events, RRT renal replacement therapy
Finally, we assessed a subset of articles that only included patients treated with RRT and reported the rate of patients with chronic dialysis or RRT dependence at day 90 as the dialysis component criteria (n = 6). [33, 37, 51, 87, 100, 109] By definition, if the “any RRT at any time” criterion was used, 100% would have been MAKE positive. However, using the actual pre-set study MAKE criteria only 63.6% were reported as MAKE positive, and dialysis at day 90 was seen in only 8% (supplementary Table S7).
Discussion
Key findings
In this systematic scoping review, we found marked heterogeneity in applied criteria for MAKE across 122 eligible studies. While the number of published articles using MAKE as an endpoint continued to rise, heterogeneity remained, both in terms of the observation periods selected for MAKE evaluation and the criteria applied to each MAKE component. In particular, we identified 37 different criteria for persistent renal dysfunction, which can be further influenced by baseline creatinine definitions and/or eGFR calculations. Moreover, in some studies, descriptions of applied criteria for MAKE components were lacking and criteria and observation periods for MAKE evaluation vary according to study characteristics. Furthermore, MAKE rates varied by as much as 7% with different observation periods and by 36.4% with different dialysis component definitions.
Relationship to previous studies
To the best of our knowledge, this is the first systematic scoping review to describe the heterogeneity in the applied criteria and observation periods used to detect MAKE. Observed heterogeneity and its impact on MAKE rates, with some studies even lacking a description of applied criteria, are noteworthy and even problematic in study interpretation or comparison and planning RCTs. [142, 143]
The concept of MAKE was originally proposed by Shaw in 2011 [144] as a patient-centered renal outcome including death, new dialysis, and incident or progressive chronic kidney disease. Subsequently, it was operationalized to include death, dialysis, and persistent renal dysfunction in most articles. Since the introduction of MAKE, death has been a key component. However, it remains unclear if death is a kidney injury related endpoint or a competing endpoint for renal dysfunction. This is because mortality mostly reflects the underlying acute illness. Although death from kidney failure has been employed as a component for proposed Major Adverse Renal Events for CKD patients [145], attributing mortality to kidney injury in intensive care unit patients, however, is particularly challenging and possibly misleading. Yet, death is a fundamental patient-centered outcome. As a consequence, despite such concerns, death from any cause continues to be used in studies of MAKE.
The original concept of MAKE was to serve as the renal analog of MACE. In this regard, since 2008, MACE has been adopted by the United States of America Food and Drug Administration (FDA) to assess the cardiovascular risks of therapies for diabetes. [9] Subsequently, the European Medicines Agency (EMA) adopted it as the preferred cardiovascular safety endpoint in trials of new medicinal products. [146] Additionally, the Academic Research Consortium-2 Consensus Document recommended such composite outcomes in coronary intervention trials. [10] These guidelines have accelerated the adoption of MACE as a primary outcome measure in cardiovascular research. Despite early heterogeneity, [147,148,149] several statements describing uniform definitions of MACE components [150, 151] have now been published as collaborative consensus statements of relevant associations including the Standardized Data Collection for Cardiovascular Trials Initiative (SCTI) and FDA. Our observations suggest that MAKE may be in a position similar to that of MACE approximately 15 years ago.
Our findings suggest that, despite the lack of consensus, the use of MAKE as an outcome measure is increasing as it may help provide more statistical power in RCTs. This was suggested by the findings of the REVIVAL trial (Recombinant human alkaline phosphatase SA-AKI survival), [137] which reported a significant difference in MAKE rates between treatment groups despite non-significant differences in 28-day mortality in sepsis-associated AKI. In response, the need for standardized MAKE definitions and the risk of applying different definitions in RCTs were highlighted in the accompanying editorial. [142]
Implications of study findings
Our findings imply that the use of MAKE as an endpoint is increasing and, as such, its clinical relevance is also increasing. At the same time, our observations demonstrate that there is substantial heterogeneity in applied criteria for each MAKE component and observation period. This heterogeneity impacts on reported MAKE rates and makes its current use as an endpoint more challenging. Additionally, considering the difficulties in powering RCTs of AKI and AKD treatment, MAKE’s potential advantage of increasing event numbers and thus statistical power is likely to lead to more studies using it as the preferred primary outcome. In this regard, our observations suggest that establishing uniform and acceptable standards for operationalizing each MAKE component with the assistance of independent adjudication committees, a similar path to MACE, may well be fundamental for the advancement of trial medicine in critical care nephrology. Moreover, as MAKE faces additional specific challenges due to a lack of consensus on indications for initiating dialysis or on minimal clinically important differences for persistent renal dysfunction, dedicated discussion for unified principles and acceptable standards in operationalizing MAKE is warranted.
Finally, MAKE can be seen as part of the continuum of AKI, AKD, and CKD definitions, aligning with the original intent of MAKE. Toward this end, the evolution of MAKE may incorporate elements from proposed AKD or CKD definitions, such as structural markers of kidney damage [152]. We acknowledge that until more consensus is achieved, a degree of flexibility in the MAKE definition, accompanied by clear descriptions, will remain. This is particularly true for exploratory retrospective studies that utilize established databases or in studies with specific aims requiring a tailored selection of MAKE components.
Strength and limitations
To our knowledge, the heterogeneity in applied criteria for each MAKE component employed in the literature has not been previously systematically evaluated. Thus, this study provides the first systematic assessment of such heterogeneity for component choice, observation periods, and component identification, as reported in the literature. Our findings offer a comprehensive overview of variability in the use of MAKE as study endpoint and provide perspective on the potential effect of different MAKE definitions on MAKE rates. This characterization generates fundamental insights into the current landscape surrounding MAKE and is important to future standardization of criteria for trials.
We acknowledge some limitations. First, the varied design and reporting of studies posed some difficulties in extracting fully comparable information. However, our formal methodology utilizing multiple independent reviewers helped minimize misinterpretation risks. Moreover, such heterogeneity was sufficiently large to minimize the impact of minor differences in the interpretation of individual articles. On the other hand, such heterogeneity made it necessary to conduct a scoping review rather than a formal meta-analysis and systematic review. This scoping design allowed us to comprehensively describe and quantify such heterogeneity. Moreover, given the wide variability demonstrated, our findings reinforce the challenges of performing a robust meta-analysis on this topic. Second, we did not describe the analytical challenges associated with employing composite endpoints like MAKE. We acknowledge that individual MAKE components may differently impact patient outcomes and any interpretability depends on how components are weighted or analyzed statistically. However, emerging methods like win-ratio modeling may help address this challenge. [153] Furthermore, MAKE emphasizes clinically relevant elements beyond simple creatinine/eGFR and aims to capture competing risks, supporting its usefulness despite these challenges. Third, we did not capture studies employing alternative composite outcomes with similar underlying endpoints but different naming conventions than MAKE. However, no competing widely used system exists to challenge MAKE’s pre-eminence in this field.
Conclusion
In this systemic scoping review, we found an annual increase in the number of articles employing MAKE as an endpoint. We also found substantial heterogeneity in the applied criteria and observation periods for MAKE evaluation across published articles, particularly regarding the components of persistent renal dysfunction and dialysis dependence. This heterogeneity leads to inconsistent results as well as difficulties in comparing or integrating renal outcome data among studies. Since employing composite outcomes like MAKE that incorporate patient-centered perspectives appears logical and clinically valid, our findings indicate the need for dedicated discussion on how to develop more uniform and acceptable standards for the operationalization of MAKE. Such discussion is important for the application of MAKE in future RCTs of patients with AKI or AKD.
Data availability
Data sharing is not applicable to this scoping review as no new data were created or analyzed in this review.
References
Hoste EA, Bagshaw SM, Bellomo R, Cely CM, Colman R, Cruz DN, Edipidis K, Forni LG, Gomersall CD, Govil D, Honore PM, Joannes-Boyau O, Joannidis M, Korhonen AM, Lavrentieva A, Mehta RL, Palevsky P, Roessler E, Ronco C, Uchino S, Vazquez JA, Vidal Andrade E, Webb S, Kellum JA (2015) Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med 41:1411–1423. https://doi.org/10.1007/s00134-015-3934-7
Hoste EAJ, Kellum JA, Selby NM, Zarbock A, Palevsky PM, Bagshaw SM, Goldstein SL, Cerda J, Chawla LS (2018) Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol 14:607–625. https://doi.org/10.1038/s41581-018-0052-0
See EJ, Jayasinghe K, Glassford N, Bailey M, Johnson DW, Polkinghorne KR, Toussaint ND, Bellomo R (2019) Long-term risk of adverse outcomes after acute kidney injury: a systematic review and meta-analysis of cohort studies using consensus definitions of exposure. Kidney Int 95:160–172. https://doi.org/10.1016/j.kint.2018.08.036
Kellum J, Lameire N, Aspelin P (2012) KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl 2:1–138. https://doi.org/10.1038/kisup.2012.1
Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM, Bittleman D, Cruz D, Endre Z, Fitzgerald RL, Forni L, Kane-Gill SL, Hoste E, Koyner J, Liu KD, Macedo E, Mehta R, Murray P, Nadim M, Ostermann M, Palevsky PM, Pannu N, Rosner M, Wald R, Zarbock A, Ronco C, Kellum JA, Acute Disease Quality Initiative W (2017) Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol 13:241–257. https://doi.org/10.1038/nrneph.2017.2
James MT, Levey AS, Tonelli M, Tan Z, Barry R, Pannu N, Ravani P, Klarenbach SW, Manns BJ, Hemmelgarn BR (2019) Incidence and prognosis of acute kidney diseases and disorders using an integrated approach to laboratory measurements in a universal health care system. JAMA Netw Open 2:e191795. https://doi.org/10.1001/jamanetworkopen.2019.1795
Sawhney S, Bell S, Black C, Christiansen CF, Heide-Jorgensen U, Jensen SK, Ronksley PE, Tan Z, Tonelli M, Walker H, James MT (2022) Harmonization of epidemiology of acute kidney injury and acute kidney disease produces comparable findings across four geographic populations. Kidney Int 101:1271–1281. https://doi.org/10.1016/j.kint.2022.02.033
Chawla LS, Eggers PW, Star RA, Kimmel PL (2014) Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med 371:58–66. https://doi.org/10.1056/NEJMra1214243
FDA Center for Drug Evaluation and Research (CDER) (2008) Guidance for Industry Diabetes Mellitus -Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes. https://downloads.regulations.gov/FDA-2008-D-0118-0029/content.pdf, Accessed 6 Feb 2024.
Garcia-Garcia HM, McFadden EP, Farb A, Mehran R, Stone GW, Spertus J, Onuma Y, Morel MA, van Es GA, Zuckerman B, Fearon WF, Taggart D, Kappetein AP, Krucoff MW, Vranckx P, Windecker S, Cutlip D, Serruys PW, Academic Research C (2018) Standardized End point definitions for coronary intervention trials: the Academic Research Consortium-2 Consensus Document. Circulation 137:2635–2650. https://doi.org/10.1161/CIRCULATIONAHA.117.029289
Sharma A, Pagidipati NJ, Califf RM, McGuire DK, Green JB, Demets D, George JT, Gerstein HC, Hobbs T, Holman RR, Lawson FC, Leiter LA, Pfeffer MA, Reusch J, Riesmeyer JS, Roe MT, Rosenberg Y, Temple R, Wiviott S, McMurray J, Granger C (2020) Impact of regulatory guidance on evaluating cardiovascular risk of new glucose-lowering therapies to treat type 2 diabetes mellitus: lessons learned and future directions. Circulation 141:843–862. https://doi.org/10.1161/CIRCULATIONAHA.119.041022
Holm NR, Andreasen LN, Neghabat O, Laanmets P, Kumsars I, Bennett J, Olsen NT, Odenstedt J, Hoffmann P, Dens J, Chowdhary S, O’Kane P, Bulow Rasmussen SH, Heigert M, Havndrup O, Van Kuijk JP, Biscaglia S, Mogensen LJH, Henareh L, Burzotta FCHE, Mylotte D, Llinas MS, Koltowski L, Knaapen P, Calic S, Witt N, Santos-Pardo I, Watkins S, Lonborg J, Kristensen AT, Jensen LO, Calais F, Cockburn J, McNeice A, Kajander OA, Heestermans T, Kische S, Eftekhari A, Spratt JC, Christiansen EH, Group OT (2023) OCT or angiography guidance for PCI in complex bifurcation lesions. N Engl J Med 389:1477–1487. https://doi.org/10.1056/NEJMoa2307770
Palevsky PM, Molitoris BA, Okusa MD, Levin A, Waikar SS, Wald R, Chertow GM, Murray PT, Parikh CR, Shaw AD, Go AS, Faubel SG, Kellum JA, Chinchilli VM, Liu KD, Cheung AK, Weisbord SD, Chawla LS, Kaufman JS, Devarajan P, Toto RM, Hsu CY, Greene T, Mehta RL, Stokes JB, Thompson AM, Thompson BT, Westenfelder CS, Tumlin JA, Warnock DG, Shah SV, Xie Y, Duggan EG, Kimmel PL, Star RA (2012) Design of clinical trials in acute kidney injury: report from an NIDDK workshop on trial methodology. Clin J Am Soc Nephrol 7:844–850. https://doi.org/10.2215/CJN.12791211
Kellum JA, Zarbock A, Nadim MK (2017) What endpoints should be used for clinical studies in acute kidney injury? Intensive Care Med 43:901–903. https://doi.org/10.1007/s00134-017-4732-1
Palevsky PM, Liu KD (2018) What endpoints should not be used for clinical studies of acute kidney injury? Intensive Care Med 44:363–365. https://doi.org/10.1007/s00134-017-4841-x
Billings FTt, Shaw AD (2014) Clinical trial endpoints in acute kidney injury. Nephron Clin Pract 127:89–93. https://doi.org/10.1159/000363725
Zorzela L, Loke YK, Ioannidis JP, Golder S, Santaguida P, Altman DG, Moher D, Vohra S, Group PR (2016) PRISMA harms checklist: improving harms reporting in systematic reviews. BMJ 352:i157. https://doi.org/10.1136/bmj.i157
Peters MDJ GC, McInerney P, Munn Z, Tricco AC, Khalil, H. (2020) Chapter 11: Scoping Reviews (2020 version). Aromataris E, Munn Z, editors. JBI Manual for Evidence Synthesis. https://synthesismanual.jbi.global, Accessed 18 Dec 2023.
Patil I (2021) Visualizations with statistical details: The “ggstatsplot” approach. J Open Source Softw. https://doi.org/10.21105/joss.03167
Chen JJ, Lee TH, Kuo G, Yen CL, Chen SW, Chu PH, Fan PC, Chien-Chia WuV, Chang CH (2022) Acute kidney disease after acute decompensated heart failure. Kidney Int Rep 7:526–536. https://doi.org/10.1016/j.ekir.2021.12.033
Yan P, Duan XJ, Liu Y, Wu X, Zhang NY, Yuan F, Tang H, Liu Q, Deng YH, Wang HS, Wang M, Duan SB (2021) Acute kidney disease in hospitalized acute kidney injury patients. PeerJ 9:e11400. https://doi.org/10.7717/peerj.11400
Musajee M, Katsogridakis E, Kiberu Y, Banerjee C, George R, Modarai B, Saratzis A, Sandford B (2023) Acute kidney injury in patients with acute type B aortic dissection. Eur J Vasc Endovasc Surg 65:256–262. https://doi.org/10.1016/j.ejvs.2022.10.032
Thongprayoon C, Cheungpasitporn W, Srivali N, Harrison AM, Gunderson TM, Kittanamongkolchai W, Greason KL, Kashani KB (2016) AKI after transcatheter or surgical aortic valve replacement. J Am Soc Nephrol 27:1854–1860. https://doi.org/10.1681/ASN.2015050577
Menez S, Wen Y, Xu L, Moledina DG, Thiessen-Philbrook H, Hu D, Obeid W, Bhatraju PK, Ikizler TA, Siew ED, Chinchilli VM, Garg AX, Go AS, Liu KD, Kaufman JS, Kimmel PL, Himmelfarb J, Coca SG, Cantley LG, Parikh CR (2023) The ASSESS-AKI Study found urinary epidermal growth factor is associated with reduced risk of major adverse kidney events. Kidney Int 104:1194–1205. https://doi.org/10.1016/j.kint.2023.08.007
Chawla LS, Amdur RL, Shaw AD, Faselis C, Palant CE, Kimmel PL (2014) Association between AKI and long-term renal and cardiovascular outcomes in United States veterans. Clin J Am Soc Nephrol 9:448–456. https://doi.org/10.2215/CJN.02440213
Silver SA, Adhikari NK, Jeyakumar N, Luo B, Harel Z, Dixon SN, Brimble KS, Clark EG, Neyra JA, Vijayaraghavan BKT, Garg AX, Bell CM, Wald R (2023) Association of an acute kidney injury follow-up clinic with patient outcomes and care processes: a cohort study. Am J Kidney Dis 81(554–563):e551. https://doi.org/10.1053/j.ajkd.2022.10.011
Gomez H, Del Rio-Pertuz G, Priyanka P, Manrique-Caballero CL, Chang CH, Wang S, Liu Q, Zuckerbraun BS, Murugan R, Angus DC, Kellum JA (2022) Association of metformin use during hospitalization and mortality in critically ill adults with type 2 diabetes mellitus and sepsis. Crit Care Med 50:935–944. https://doi.org/10.1097/CCM.0000000000005468
Kang YX, Luo XQ, Zhang NY, Wu SJ, Song GB, Yan P, Duan SB (2023) Association of underweight and obesity with adverse postoperative renal outcomes in infants and young children undergoing congenital heart surgery. Eur J Pediatr 182:3691–3700. https://doi.org/10.1007/s00431-023-05041-w
Isshiki R, Asada T, Sato D, Sumida M, Hamasaki Y, Inokuchi R, Matsubara T, Ishii T, Yahagi N, Nangaku M, Noiri E, Doi K (2016) Association of urinary neutrophil gelatinase-associated lipocalin with long-term renal outcomes in ICU survivors: a retrospective observational cohort study. Shock 46:44–51. https://doi.org/10.1097/SHK.0000000000000580
Behal ML, Nguyen JL, Li X, Feola DJ, Neyra JA, Flannery AH (2022) Azithromycin and Major adverse kidney events in critically ill patients with sepsis-associated acute kidney injury. Shock 57:479–485. https://doi.org/10.1097/SHK.0000000000001883
Brems JH, Casey JD, Wang L, Self WH, Rice TW, Semler MW, Investigators S, the Pragmatic Critical Care Research G (2021) Balanced crystalloids versus saline in critically ill adults with low plasma bicarbonate: a secondary analysis of a clinical trial. J Crit Care 63:250–253. https://doi.org/10.1016/j.jcrc.2020.12.016
Brown RM, Wang L, Coston TD, Krishnan NI, Casey JD, Wanderer JP, Ehrenfeld JM, Byrne DW, Stollings JL, Siew ED, Bernard GR, Self WH, Rice TW, Semler MW (2019) Balanced crystalloids versus saline in sepsis. A secondary analysis of the SMART clinical trial. Am J Respir Crit Care Med 200:1487–1495. https://doi.org/10.1164/rccm.201903-0557OC
Shawwa K, Kompotiatis P, Wiley BM, Jentzer JC, Kashani KB (2021) Change in right ventricular systolic function after continuous renal replacement therapy initiation and renal recovery. J Crit Care 62:82–87. https://doi.org/10.1016/j.jcrc.2020.11.022
Forni LG, Joannidis M, Artigas A, Bell M, Hoste E, Joannes-Boyau O, Kashani K, Koyner J, Rimmele T, Shi J, Ostermann M, Chawla LS, Kellum JA, Sapphire I (2022) Characterising acute kidney injury: the complementary roles of biomarkers of renal stress and renal function. J Crit Care 71:154066. https://doi.org/10.1016/j.jcrc.2022.154066
Hochstadt A, Avivi I, Ingbir M, Shacham Y, Merdler I, Granot Y, Viskin S, Rosso R, Banai S, Konigstein M (2021) Clinically significant high-grade AV block as a reversible cause for acute kidney injury in hospitalized patients-a propensity score matched cohort. J Clin Med. https://doi.org/10.3390/jcm10112424
Xin Q, Xie T, Chen R, Wang H, Zhang X, Wang S, Liu C, Zhang J (2022) Construction and validation of an early warning model for predicting the acute kidney injury in elderly patients with sepsis. Aging Clin Exp Res 34:2993–3004. https://doi.org/10.1007/s40520-022-02236-3
Liu C, Peng Z, Dong Y, Li Z, Song X, Liu X, Andrijasevic NM, Gajic O, Albright RC Jr, Kashani KB (2021) Continuous renal replacement therapy liberation and outcomes of critically ill patients with acute kidney injury. Mayo Clin Proc 96:2757–2767. https://doi.org/10.1016/j.mayocp.2021.05.031
Zhang H, Yu M, Wang R, Fan R, Zhang K, Chen W, Chen X (2022) Derivation and validation a risk model for acute kidney injury and subsequent adverse events after cardiac surgery: a multicenter cohort study. Int J Gen Med 15:7751–7760. https://doi.org/10.2147/IJGM.S354821
Bobba A, Costanian C, Bahous SA, Tohme FA (2022) Determinants of major adverse kidney events in extracorporeal membrane oxygenation survivors. Crit Care Explor 4:e0636. https://doi.org/10.1097/CCE.0000000000000636
Venugopal H, Jacob KA, Dieleman JM, Leaf DE (2020) Dexamethasone for preventing major adverse kidney events following cardiac surgery: post-hoc analysis to identify subgroups. Kidney360 1:530–533. https://doi.org/10.34067/KID.0000812019
Ruth A, Basu RK, Gillespie S, Morgan C, Zaritsky J, Selewski DT, Arikan AA (2022) Early and late acute kidney injury: temporal profile in the critically ill pediatric patient. Clin Kidney J 15:311–319. https://doi.org/10.1093/ckj/sfab199
Funke BE, Jackson KE, Self WH, Collins SP, Saunders CT, Wang L, Blume JD, Wickersham N, Brown RM, Casey JD, Bernard GR, Rice TW, Siew ED, Semler MW, Investigators S, Pragmatic Critical Care Research G (2021) Effect of balanced crystalloids versus saline on urinary biomarkers of acute kidney injury in critically ill adults. BMC Nephrol 22:54. https://doi.org/10.1186/s12882-021-02236-x
Gomez H, Priyanka P, Bataineh A, Keener CM, Clermont G, Kellum JA (2021) Effects of 5% albumin plus saline versus saline alone on outcomes from large-volume resuscitation in critically ill patients. Crit Care Med 49:79–90. https://doi.org/10.1097/CCM.0000000000004706
Pandirajan K, Katsogridakis E, Sidloff D, Sayers RD, Bown MJ, Saratzis A (2020) Effects of left renal vein ligation during open abdominal aortic aneurysm repair on renal function. Eur J Vasc Endovasc Surg 60:829–835. https://doi.org/10.1016/j.ejvs.2020.08.003
Wang M, Wang X, Zhu B, Li W, Jiang Q, Zuo Y, Wen J, He Y, Xi X, Jiang L (2023) The effects of timing onset and progression of AKI on the clinical outcomes in AKI patients with sepsis: a prospective multicenter cohort study. Ren Fail 45:1–10. https://doi.org/10.1080/0886022X.2022.2138433
See EJ, Polkinghorne KR, Toussaint ND, Bailey M, Johnson DW, Bellomo R (2021) Epidemiology and outcomes of acute kidney diseases: a comparative analysis. Am J Nephrol 52:342–350. https://doi.org/10.1159/000515231
Chan YC, Yeh CH, Li LC, Chen CL, Wang CC, Lin CC, Ong AD, Chiou TY, Yong CC (2022) Excess risk of major adverse cardiovascular and kidney events after acute kidney injury following living donor liver transplantation. J Clin Med. https://doi.org/10.3390/jcm11113100
Vinclair C, De Montmollin E, Sonneville R, Reuter J, Lebut J, Cally R, Mourvillier B, Neuville M, Ruckly S, Timsit JF, Bouadma L (2020) Factors associated with major adverse kidney events in patients who underwent veno-arterial extracorporeal membrane oxygenation. Ann Intensive Care 10:44. https://doi.org/10.1186/s13613-020-00656-w
Mele A, Cerminara E, Habel H, Rodriguez-Galvez B, Oldner A, Nelson D, Gardh J, Thobaben R, Jonmarker S, Cronhjort M, Hollenberg J, Martensson J (2022) Fluid accumulation and major adverse kidney events in sepsis: a multicenter observational study. Ann Intensive Care 12:62. https://doi.org/10.1186/s13613-022-01040-6
Alobaidi R, Basu RK, DeCaen A, Joffe AR, Lequier L, Pannu N, Bagshaw SM (2020) Fluid accumulation in critically ill children. Crit Care Med 48:1034–1041. https://doi.org/10.1097/CCM.0000000000004376
Woodward CW, Lambert J, Ortiz-Soriano V, Li Y, Ruiz-Conejo M, Bissell BD, Kelly A, Adams P, Yessayan L, Morris PE, Neyra JA (2019) Fluid overload associates with major adverse kidney events in critically ill patients with acute kidney injury requiring continuous renal replacement therapy. Crit Care Med 47:e753–e760. https://doi.org/10.1097/CCM.0000000000003862
Shawwa K, Kompotiatis P, Jentzer JC, Wiley BM, Williams AW, Dillon JJ, Albright RC, Kashani KB (2019) Hypotension within one-hour from starting CRRT is associated with in-hospital mortality. J Crit Care 54:7–13. https://doi.org/10.1016/j.jcrc.2019.07.004
Semler MW, Rice TW, Shaw AD, Siew ED, Self WH, Kumar AB, Byrne DW, Ehrenfeld JM, Wanderer JP (2016) Identification of major adverse kidney events within the electronic health record. J Med Syst 40:167. https://doi.org/10.1007/s10916-016-0528-z
Haase M, Bellomo R, Albert C, Vanpoucke G, Thomas G, Laroy W, Verleysen K, Kropf S, Kuppe H, Hetzer R, Haase-Fielitz A (2014) The identification of three novel biomarkers of major adverse kidney events. Biomark Med 8:1207–1217. https://doi.org/10.2217/bmm.14.90
Priyanka P, Zarbock A, Izawa J, Gleason TG, Renfurm RW, Kellum JA (2021) The impact of acute kidney injury by serum creatinine or urine output criteria on major adverse kidney events in cardiac surgery patients. J Thorac Cardiovasc Surg 162(143–151):e147. https://doi.org/10.1016/j.jtcvs.2019.11.137
Chawla LS, Amdur RL, Faselis C, Li P, Kimmel PL, Palant CE (2017) Impact of acute kidney injury in patients hospitalized with pneumonia. Crit Care Med 45:600–606. https://doi.org/10.1097/CCM.0000000000002245
Molina Andujar A, Escudero VJ, Pineiro GJ, Lucas A, Rovira I, Matute P, Ibanez C, Blasco M, Quintana LF, Sandoval E, Sanchez MC, Quintana E, Poch E (2023) Impact of cardiac surgery associated acute kidney injury on 1-year major adverse kidney events. Front Nephrol 3:1059668. https://doi.org/10.3389/fneph.2023.1059668
Chang CH, Chen SW, Chen JJ, Chan YH, Yen CL, Lee TH, Cheng YT (2021) Incidence and transition of acute kidney injury, acute kidney disease to chronic kidney disease after acute type a aortic dissection surgery. J Clin Med. https://doi.org/10.3390/jcm10204769
Zhou D, Jiang J, Zhang J, Cao F, Peng Z (2023) Increase in chloride is associated with major adverse kidney events in critically ill patients. Shock 59:338–343. https://doi.org/10.1097/SHK.0000000000002062
Parikh CR, Liu C, Mor MK, Palevsky PM, Kaufman JS, Thiessen Philbrook H, Weisbord SD (2020) Kidney biomarkers of injury and repair as predictors of contrast-associated AKI: a substudy of the PRESERVE Trial. Am J Kidney Dis 75:187–194. https://doi.org/10.1053/j.ajkd.2019.06.011
Murugan R, Boudreaux-Kelly MY, Kellum JA, Palevsky PM, Weisbord S, Biomarker Effectiveness Analysis in Contrast Nephropathy Study I (2023) Kidney cell cycle arrest and cardiac biomarkers and acute kidney injury following angiography: the prevention of serious adverse events following angiography (PRESERVE) study. Kidney Med 5:100592. https://doi.org/10.1016/j.xkme.2022.100592
Bowe B, Xie Y, Xu E, Al-Aly Z (2021) Kidney outcomes in long COVID. J Am Soc Nephrol 32:2851–2862. https://doi.org/10.1681/ASN.2021060734
Chisavu F, Gafencu M, Chisavu L, Stroescu R, Schiller A (2023) Kinetic estimated glomerular filtration rate in predicting paediatric acute kidney disease. J Clin Med. https://doi.org/10.3390/jcm12196314
Meersch M, Kullmar M, Schmidt C, Gerss J, Weinhage T, Margraf A, Ermert T, Kellum JA, Zarbock A (2018) Long-term clinical outcomes after early initiation of RRT in critically ill patients with AKI. J Am Soc Nephrol 29:1011–1019. https://doi.org/10.1681/ASN.2017060694
Zarbock A, Kellum JA, Van Aken H, Schmidt C, Kullmar M, Rosenberger P, Martens S, Gorlich D, Meersch M (2017) Long-term effects of remote ischemic preconditioning on kidney function in high-risk cardiac surgery patients: follow-up results from the RenalRIP Trial. Anesthesiology 126:787–798. https://doi.org/10.1097/ALN.0000000000001598
Chen SW, Lu YA, Lee CC, Chou AH, Wu VC, Chang SW, Fan PC, Tian YC, Tsai FC, Chang CH (2019) Long-term outcomes after extracorporeal membrane oxygenation in patients with dialysis-requiring acute kidney injury: a cohort study. PLoS ONE 14:e0212352. https://doi.org/10.1371/journal.pone.0212352
Lu JY, Boparai MS, Shi C, Henninger EM, Rangareddy M, Veeraraghavan S, Mirhaji P, Fisher MC, Duong TQ (2023) Long-term outcomes of COVID-19 survivors with hospital AKI: association with time to recovery from AKI. Nephrol Dial Transplant 38:2160–2169. https://doi.org/10.1093/ndt/gfad020
Patidar KR, Naved MA, Kabir S, Grama A, Allegretti AS, Cullaro G, Asrani SK, Worden A, Desai AP, Ghabril MS, Nephew LD, Orman ES (2023) Longer time to recovery from acute kidney injury is associated with major adverse kidney events in patients with cirrhosis. Aliment Pharmacol Ther 57:1397–1406. https://doi.org/10.1111/apt.17457
Kula AJ, Qu P, Strub B, Smith JM, Menon S (2022) Major adverse kidney events after acute kidney injury in the pediatric intensive care unit: a propensity score-matched cohort study. Pediatr Nephrol 37:2099–2107. https://doi.org/10.1007/s00467-021-05348-6
Weiss SL, Balamuth F, Thurm CW, Downes KJ, Fitzgerald JC, Laskin BL (2019) Major adverse kidney events in pediatric sepsis. Clin J Am Soc Nephrol 14:664–672. https://doi.org/10.2215/CJN.12201018
Zhang X, Ye B, Mao W, Liu L, Li G, Zhou J, Zhang J, Guo J, Ke L, Tong Z, Li W (2022) Major adverse kidney events within 30 days in patients with acute pancreatitis: a tertiary-center cohort study. HPB (Oxford) 24:169–175. https://doi.org/10.1016/j.hpb.2021.05.012
Deng YH, Luo XQ, Yan P, Zhang NY, Liu Y, Duan SB (2022) Outcome prediction for acute kidney injury among hospitalized children via eXtreme Gradient Boosting algorithm. Sci Rep 12:8956. https://doi.org/10.1038/s41598-022-13152-x
Qian ET, Wang L, Stollings JL, Casey JD, Rice TW, Semler MW (2023) Piperacillin-tazobactam versus anti-pseudomonal cephalosporins and renal and neurologic outcomes in critically ill adults: a secondary analysis of the SMART trial. J Intensive Care Med 38:1127–1135. https://doi.org/10.1177/08850666231184177
Cheng W, Wu X, Liu Q, Wang HS, Zhang NY, Xiao YQ, Yan P, Li XW, Duan XJ, Peng JC, Feng S, Duan SB (2020) Post-contrast acute kidney injury in a hospitalized population: short-, mid-, and long-term outcome and risk factors for adverse events. Eur Radiol 30:3516–3527. https://doi.org/10.1007/s00330-020-06690-3
Liu C, Mor MK, Palevsky PM, Kaufman JS, Thiessen Philbrook H, Weisbord SD, Parikh CR (2020) Postangiography increases in serum creatinine and biomarkers of injury and repair. Clin J Am Soc Nephrol 15:1240–1250. https://doi.org/10.2215/CJN.15931219
See EJ, Ransley DG, Polkinghorne KR, Toussaint ND, Bailey M, Johnson DW, Robbins R, Bellomo R (2022) Practice patterns and predictors of outpatient care following acute kidney injury in an Australian healthcare setting. Intern Med J 52:79–88. https://doi.org/10.1111/imj.15138
McKown AC, Wang L, Wanderer JP, Ehrenfeld J, Rice TW, Bernard GR, Semler MW (2017) Predicting major adverse kidney events among critically ill adults using the electronic health record. J Med Syst 41:156. https://doi.org/10.1007/s10916-017-0806-4
Neyra JA, Ortiz-Soriano V, Liu LJ, Smith TD, Li X, Xie D, Adams-Huet B, Moe OW, Toto RD, Chen J (2023) Prediction of mortality and major adverse kidney events in critically ill patients with acute kidney injury. Am J Kidney Dis 81:36–47. https://doi.org/10.1053/j.ajkd.2022.06.004
Xin Q, Xie T, Chen R, Zhang X, Tong Y, Wang H, Wang S, Liu C, Zhang J (2022) A predictive model based on inflammatory and coagulation indicators for sepsis-induced acute kidney injury. J Inflamm Res 15:4561–4571. https://doi.org/10.2147/JIR.S372246
Albert C, Haase M, Albert A, Ernst M, Kropf S, Bellomo R, Westphal S, Braun-Dullaeus RC, Haase-Fielitz A, Elitok S (2021) Predictive value of plasma NGAL:Hepcidin-25 for major adverse kidney events after cardiac surgery with cardiopulmonary bypass: a pilot study. Ann Lab Med 41:357–365. https://doi.org/10.3343/alm.2021.41.4.357
Chauhan K, Pattharanitima P, Piani F, Johnson RJ, Uribarri J, Chan L, Coca SG (2022) Prevalence and outcomes associated with hyperuricemia in hospitalized patients with COVID-19. Am J Nephrol 53:78–86. https://doi.org/10.1159/000520355
Whitenack K, Behal ML, Thompson Bastin ML, Aycinena JC, Adams PM, Flannery AH (2022) Progression of kidney injury with the combination of vancomycin and piperacillin-tazobactam or cefepime in sepsis-associated acute kidney Injury. Front Nephrol. https://doi.org/10.3389/fneph.2022.995358
See EJ, Toussaint ND, Bailey M, Johnson DW, Polkinghorne KR, Robbins R, Bellomo R (2021) Risk factors for major adverse kidney events in the first year after acute kidney injury. Clin Kidney J 14:556–563. https://doi.org/10.1093/ckj/sfz169
Kusirisin P, Thanapongsatorn P, Chaikomon K, Lumlertgul N, Yimsangyad K, Leewongworasingh A, Sirivongrangson P, Peerapornratana S, Chaijamorn W, Avihingsanon Y, Srisawat N (2023) The role of erythropoietin levels in predicting long-term outcomes following severe acute kidney injury. Blood Purif 52:793–801. https://doi.org/10.1159/000531954
Chen YW, Wu MY, Mao CH, Yeh YT, Chen TT, Liao CT, Zheng CM, Hsu YH, Cherng YG, Wu MS (2022) Severe acute kidney disease is associated with worse kidney outcome among acute kidney injury patients. Sci Rep 12:6492. https://doi.org/10.1038/s41598-022-09599-7
Andonovic M, Traynor JP, Shaw M, Sim MAB, Mark PB, Puxty KA (2022) Short- and long-term outcomes of intensive care patients with acute kidney disease. EClinicalMedicine 44:101291. https://doi.org/10.1016/j.eclinm.2022.101291
Fuhrman DY, Thadani S, Hanson C, Carcillo JA, Kellum JA, Park HJ, Lu L, Kim-Campbell N, Horvat CM, Arikan AA (2023) Therapeutic plasma exchange is associated with improved major adverse kidney events in children and young adults with thrombocytopenia at the time of continuous kidney replacement therapy initiation. Crit Care Explor 5:e0891. https://doi.org/10.1097/CCE.0000000000000891
Thongprayoon C, Cheungpasitporn W, Srivali N, Harrison AM, Kittanamongkolchai W, Greason KL, Kashani KB (2017) Transapical versus transfemoral approach and risk of acute kidney injury following transcatheter aortic valve replacement: a propensity-adjusted analysis. Ren Fail 39:13–18. https://doi.org/10.1080/0886022X.2016.1244072
Depret F, Dunyach C, De Tymowski C, Chaussard M, Bataille A, Ferry A, Moreno N, Cupaciu A, Soussi S, Benyamina M, Mebazaa A, Serror K, Chaouat M, Garnier JP, Pirracchio R, Legrand M, group P, (2017) Undetectable haptoglobin is associated with major adverse kidney events in critically ill burn patients. Crit Care 21:245. https://doi.org/10.1186/s13054-017-1837-4
Albert C, Haase M, Albert A, Kropf S, Bellomo R, Westphal S, Westerman M, Braun-Dullaeus RC, Haase-Fielitz A (2020) Urinary biomarkers may complement the Cleveland score for prediction of adverse kidney events after cardiac surgery: a pilot study. Ann Lab Med 40:131–141. https://doi.org/10.3343/alm.2020.40.2.131
Albert C, Albert A, Bellomo R, Kropf S, Devarajan P, Westphal S, Baraki H, Kutschka I, Butter C, Haase M, Haase-Fielitz A (2018) Urinary neutrophil gelatinase-associated lipocalin-guided risk assessment for major adverse kidney events after open-heart surgery. Biomark Med 12:975–985. https://doi.org/10.2217/bmm-2018-0071
Lumlertgul N, Amprai M, Tachaboon S, Dinhuzen J, Peerapornratana S, Kerr SJ, Srisawat N (2020) Urine neutrophil gelatinase-associated lipocalin (NGAL) for prediction of persistent AKI and major adverse kidney events. Sci Rep 10:8718. https://doi.org/10.1038/s41598-020-65764-w
Chen Y, Wang G, Zhou H, Yang L, Zhang C, Yang X, Lei G (2020) 90 days impacts of remote ischemic preconditioning on patients undergoing open total aortic arch replacement: a post-hoc analysis of previous trial. BMC Anesthesiol 20:169. https://doi.org/10.1186/s12871-020-01085-9
Katsogridakis E, Lea T, Yap T, Batchelder A, Saha P, Diamantopoulos A, Saratzis N, Davies R, Zayed H, Bown MJ, Saratzis A (2021) Acute kidney injury following endovascular intervention for peripheral artery disease. Br J Surg 108:152–159. https://doi.org/10.1093/bjs/znaa057
Bhatraju PK, Chai XY, Sathe NA, Ruzinski J, Siew ED, Himmelfarb J, Hoofnagle AN, Wurfel MM, Kestenbaum BR (2021) Assessment of kidney proximal tubular secretion in critical illness. JCI Insight. https://doi.org/10.1172/jci.insight.145514
Bhatraju PK, Zelnick LR, Chinchilli VM, Moledina DG, Coca SG, Parikh CR, Garg AX, Hsu CY, Go AS, Liu KD, Ikizler TA, Siew ED, Kaufman JS, Kimmel PL, Himmelfarb J, Wurfel MM (2020) Association between early recovery of kidney function after acute kidney injury and long-term clinical outcomes. JAMA Netw Open 3:e202682. https://doi.org/10.1001/jamanetworkopen.2020.2682
Massoth C, Kullmar M, Enders D, Kellum JA, Forni LG, Meersch M, Zarbock A, Progressive AKIG (2023) Comparison of C-C motif chemokine ligand 14 with other biomarkers for adverse kidney events after cardiac surgery. J Thorac Cardiovasc Surg 165(199–207):e192. https://doi.org/10.1016/j.jtcvs.2021.03.016
Gorga SM, Carlton EF, Kohne JG, Barbaro RP, Basu RK (2021) Consensus acute kidney injury criteria integration identifies children at risk for long-term kidney dysfunction after multiple organ dysfunction syndrome. Pediatr Nephrol 36:1637–1646. https://doi.org/10.1007/s00467-020-04865-0
Chua HR, Low S, Murali TM, Wong ET, He HD, Teo BW, Thian YL, Akalya K, Vathsala A (2021) Cumulative iodinated contrast exposure for computed tomography during acute kidney injury and major adverse kidney events. Eur Radiol 31:3258–3266. https://doi.org/10.1007/s00330-020-07428-x
Yoshida T, Matsuura R, Komaru Y, Miyamoto Y, Yoshimoto K, Hamasaki Y, Noiri E, Nangaku M, Doi K (2023) Different roles of functional and structural renal markers measured at discontinuation of renal replacement therapy for acute kidney injury. Blood Purif 52:786–792. https://doi.org/10.1159/000532034
Saratzis A, Joshi S, Benson RA, Bosanquet D, Dattani N, Batchelder A, Fisher O, Ioannidou E, Bown MJ, Imray CH, Collaborators V (2020) Editor’s Choice-Acute Kidney Injury (AKI) in aortic intervention: findings from the midlands aortic renal injury (MARI) Cohort Study. Eur J Vasc Endovasc Surg 59:899–909. https://doi.org/10.1016/j.ejvs.2019.09.508
Boutin L, Legrand M, Sadoune M, Mebazaa A, Gayat E, Chadjichristos CE, Depret F (2022) Elevated plasma Galectin-3 is associated with major adverse kidney events and death after ICU admission. Crit Care 26:13. https://doi.org/10.1186/s13054-021-03878-x
Menez S, Coca SG, Moledina DG, Wen Y, Chan L, Thiessen-Philbrook H, Obeid W, Garibaldi BT, Azeloglu EU, Ugwuowo U, Sperati CJ, Arend LJ, Rosenberg AZ, Kaushal M, Jain S, Wilson FP, Parikh CR, Consortium T (2023) Evaluation of plasma biomarkers to predict major adverse kidney events in hospitalized patients with COVID-19. Am J Kidney Dis 82:322-332 e321. https://doi.org/10.1053/j.ajkd.2023.03.010
Shin J, Yoon Y, Oh DJ (2020) Evaluation of the Wnt signaling pathway as a prognostic marker in patients with urosepsis. Mol Cell Biochem 473:15–23. https://doi.org/10.1007/s11010-020-03804-9
Vauchel T, Pirracchio R, Chaussard M, Lafaurie M, Rouveau M, Rousseau C, Benyamina M, Soussi S, Depret F, Ferry A, Oueslati H, Chaouat M, Mimoun M, Jarlier V, Moreno N, Mebazaa A, Legrand M (2019) Impact of an Acinetobacter baumannii outbreak on kidney events in a burn unit: a targeted machine learning analysis. Am J Infect Control 47:435–438. https://doi.org/10.1016/j.ajic.2018.09.010
Collaborative ST (2021) Impact of postoperative acute kidney injury in patients undergoing major gastrointestinal surgery on 1-year survival and renal outcomes: a national multicentre cohort study. BJS Open. https://doi.org/10.1093/bjsopen/zrab134
Bhatraju PK, Prince DK, Mansour S, Ikizler TA, Siew ED, Chinchilli VM, Garg AX, Go AS, Kaufman JS, Kimmel PL, Coca SG, Parikh CR, Wurfel MM, Himmelfarb J (2023) Integrated analysis of blood and urine biomarkers to identify acute kidney injury subphenotypes and associations with long-term outcomes. Am J Kidney Dis 82(311–321):e311. https://doi.org/10.1053/j.ajkd.2023.01.449
Flannery AH, Bosler K, Ortiz-Soriano VM, Gianella F, Prado V, Lambert J, Toto RD, Moe OW, Neyra JA (2021) Kidney biomarkers and major adverse kidney events in critically ill patients. Kidney 360 2:26–32. https://doi.org/10.34067/KID.0003552020
De Corte W, Dhondt A, Vanholder R, De Waele J, Decruyenaere J, Sergoyne V, Vanhalst J, Claus S, Hoste EA (2016) Long-term outcome in ICU patients with acute kidney injury treated with renal replacement therapy: a prospective cohort study. Crit Care 20:256. https://doi.org/10.1186/s13054-016-1409-z
Bergmann L, Nowak H, Siffert W, Peters J, Adamzik M, Koos B, Rahmel T (2020) Major adverse kidney events are associated with the aquaporin 5–1364A/C promoter polymorphism in sepsis: a prospective validation study. Cells. https://doi.org/10.3390/cells9040904
See EJ, Clapham C, Liu J, Khasin M, Liskaser G, Chan JW, Serpa Neto A, Costa Pinto R, Bellomo R (2023) A pilot study of angiotensin ii as primary vasopressor in critically ill adults with vasodilatory hypotension: the Aramis study. Shock 59:691–696. https://doi.org/10.1097/SHK.0000000000002109
van der Slikke EC, Boekhoud L, Bourgonje AR, Olgers TJ, Ter Maaten JC, Henning RH, van Goor H, Bouma HR (2022) Plasma free thiol levels during early sepsis predict future renal function decline. Antioxidants (Basel). https://doi.org/10.3390/antiox11050800
Reichetzeder C, Heunisch F, Einem GV, Tsuprykov O, Kellner KH, Dschietzig T, Kretschmer A, Hocher B (2017) Pre-interventional kynurenine predicts medium-term outcome after contrast media exposure due to coronary angiography. Kidney Blood Press Res 42:244–256. https://doi.org/10.1159/000477222
Depret F, Boutin L, Jarkovsky J, Chaussard M, Soussi S, Bataille A, Oueslati H, Moreno N, de Tymowski C, Parenica J, Benesova K, Vauchel T, Ferry A, Benyamina M, Cupaciu A, Coutrot M, Garnier JP, Serror K, Chaouat M, Mebazaa A, Legrand M, Group P (2018) Prediction of major adverse kidney events in critically ill burn patients. Burns 44:1887–1894. https://doi.org/10.1016/j.burns.2018.08.007
Chen L, Wang X, Wang Q, Ding D, Jiang W, Ruan Z, Zhang W (2022) Predictive value of two different definitions of contrast-associated acute kidney injury for long-term major adverse kidney events in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Cardiol J. https://doi.org/10.5603/CJ.a2022.0034
Hollinger A, Wittebole X, Francois B, Pickkers P, Antonelli M, Gayat E, Chousterman BG, Lascarrou JB, Dugernier T, Di Somma S, Struck J, Bergmann A, Beishuizen A, Constantin JM, Damoisel C, Deye N, Gaudry S, Huberlant V, Marx G, Mercier E, Oueslati H, Hartmann O, Sonneville R, Laterre PF, Mebazaa A, Legrand M (2018) Proenkephalin A 119–159 (Penkid) Is an early biomarker of septic acute kidney injury: the kidney in sepsis and septic shock (Kid-SSS) study. Kidney Int Rep 3:1424–1433. https://doi.org/10.1016/j.ekir.2018.08.006
Panwar R, Tarvade S, Lanyon N, Saxena M, Bush D, Hardie M, Attia J, Bellomo R, Van Haren F, Investigators RSS, Research C (2020) Relative hypotension and adverse kidney-related outcomes among critically ill patients with shock. a multicenter, prospective cohort study. Am J Respir Crit Care Med 202:1407–1418. https://doi.org/10.1164/rccm.201912-2316OC
Sukmark T, Lumlertgul N, Praditpornsilpa K, Tungsanga K, Eiam-Ong S, Srisawat N (2020) SEA-MAKE score as a tool for predicting major adverse kidney events in critically ill patients with acute kidney injury: results from the SEA-AKI study. Ann Intensive Care 10:42. https://doi.org/10.1186/s13613-020-00657-9
Yadav AK, Ghosh A, Divyaveer S, Mukhopadhyay B, Kundu M, Kumar V, Lele SS, Rajapurkar MM, Jha V, Indian Chronic Kidney Disease Study G (2021) Serum catalytic iron and progression of chronic kidney disease: findings from the ICKD study. Nephrol Dial Transplant. https://doi.org/10.1093/ndt/gfab271
Collett JA, Ortiz-Soriano V, Li X, Flannery AH, Toto RD, Moe OW, Basile DP, Neyra JA (2022) Serum IL-17 levels are higher in critically ill patients with AKI and associated with worse outcomes. Crit Care 26:107. https://doi.org/10.1186/s13054-022-03976-4
Flannery AH, Ortiz-Soriano V, Li X, Gianella FG, Toto RD, Moe OW, Devarajan P, Goldstein SL, Neyra JA (2021) Serum renin and major adverse kidney events in critically ill patients: a multicenter prospective study. Crit Care 25:294. https://doi.org/10.1186/s13054-021-03725-z
Panwar R, Van Haren F, Cazzola F, Nourse M, Brinkerhoff G, Quail A (2022) Standard care versus individualized blood pressure targets among critically ill patients with shock: a multicenter feasibility and preliminary efficacy study. J Crit Care 70:154052. https://doi.org/10.1016/j.jcrc.2022.154052
Du J, Li Y, Sun Q, Wang Z, Wang F, Chen F, Wang H, Liu Y, Zhou H, Shang G, Chen X, Ding S, Li C, Wu D, Zhang W, Zhong M (2021) Urinary exosomal CD26 is associated with recovery from acute kidney injury in intensive care units: a prospective cohort study. Clin Chem Lab Med 59:1535–1546. https://doi.org/10.1515/cclm-2021-0040
Garcia-Alvarez M, Glassford NJ, Betbese AJ, Ordonez J, Banos V, Argilaga M, Martinez A, Suzuki S, Schneider AG, Eastwood GM, Victoria Moral M, Bellomo R (2015) Urinary neutrophil gelatinase-associated lipocalin as predictor of short- or long-term outcomes in cardiac surgery patients. J Cardiothorac Vasc Anesth 29:1480–1488. https://doi.org/10.1053/j.jvca.2015.05.060
Neyra JA, Li X, Mescia F, Ortiz-Soriano V, Adams-Huet B, Pastor J, Hu MC, Toto RD, Moe OW, Klotho, Acute Kidney Injury Study G (2019) Urine klotho is lower in critically ill patients with versus without acute kidney injury and associates with major adverse kidney events. Crit Care Explor. https://doi.org/10.1097/cce.0000000000000016
Semler MW, Wanderer JP, Ehrenfeld JM, Stollings JL, Self WH, Siew ED, Wang L, Byrne DW, Shaw AD, Bernard GR, Rice TW, * SI, the Pragmatic Critical Care Research G, Investigators S (2017) Balanced crystalloids versus saline in the intensive care unit. The SALT Randomized Trial. Am J Respir Crit Care Med 195:1362–1372. https://doi.org/10.1164/rccm.201607-1345OC
Self WH, Semler MW, Wanderer JP, Wang L, Byrne DW, Collins SP, Slovis CM, Lindsell CJ, Ehrenfeld JM, Siew ED, Shaw AD, Bernard GR, Rice TW, Investigators S-E (2018) Balanced crystalloids versus saline in noncritically ill adults. N Engl J Med 378:819–828. https://doi.org/10.1056/NEJMoa1711586
Semler MW, Self WH, Wanderer JP, Ehrenfeld JM, Wang L, Byrne DW, Stollings JL, Kumar AB, Hughes CG, Hernandez A, Guillamondegui OD, May AK, Weavind L, Casey JD, Siew ED, Shaw AD, Bernard GR, Rice TW, Investigators S, the Pragmatic Critical Care Research G (2018) Balanced crystalloids versus saline in critically ill adults. N Engl J Med 378:829–839. https://doi.org/10.1056/NEJMoa1711584
Qian ET, Casey JD, Wright A, Wang L, Shotwell MS, Siemann JK, Dear ML, Stollings JL, Lloyd BD, Marvi TK, Seitz KP, Nelson GE, Wright PW, Siew ED, Dennis BM, Wrenn JO, Andereck JW, Han JH, Self WH, Semler MW, Rice TW, Vanderbilt Center for Learning H, the Pragmatic Critical Care Research G (2023) Cefepime vs piperacillin-tazobactam in adults hospitalized with acute infection: the ACORN Randomized Clinical Trial. JAMA 330:1557–1567. https://doi.org/10.1001/jama.2023.20583
Thanapongsatorn P, Chaikomon K, Lumlertgul N, Yimsangyad K, Leewongworasingh A, Kulvichit W, Sirivongrangson P, Peerapornratana S, Chaijamorn W, Avihingsanon Y, Srisawat N (2021) Comprehensive versus standard care in post-severe acute kidney injury survivors, a randomized controlled trial. Crit Care 25:322. https://doi.org/10.1186/s13054-021-03747-7
James MT, Har BJ, Tyrrell BD, Faris PD, Tan Z, Spertus JA, Wilton SB, Ghali WA, Knudtson ML, Sajobi TT, Pannu NI, Klarenbach SW, Graham MM (2022) Effect of clinical decision support with audit and feedback on prevention of acute kidney injury in patients undergoing coronary angiography: a randomized clinical trial. JAMA 328:839–849. https://doi.org/10.1001/jama.2022.13382
Weiss R, Meersch M, Gerke M, Wempe C, Schafers M, Kellum JA, Zarbock A (2023) Effect of glutamine administration after cardiac surgery on kidney damage in patients at high risk for acute kidney injury: a randomized controlled trial. Anesth Analg 137:1029–1038. https://doi.org/10.1213/ANE.0000000000006288
van Till JWO, Nojima H, Kameoka C, Hayashi C, Sakatani T, Washburn TB, Molitoris BA, Shaw AD, Engelman DT, Kellum JA (2023) The effects of peroxisome proliferator-activated receptor-delta modulator ASP1128 in patients at risk for acute kidney injury following cardiac surgery. Kidney Int Rep 8:1407–1416. https://doi.org/10.1016/j.ekir.2023.04.004
Silver SA, Adhikari NK, Bell CM, Chan CT, Harel Z, Kitchlu A, Meraz-Munoz A, Norman PA, Perez A, Zahirieh A, Wald R (2021) Nephrologist follow-up versus usual care after an acute kidney injury hospitalization (FUSION): a randomized controlled trial. Clin J Am Soc Nephrol 16:1005–1014. https://doi.org/10.2215/CJN.17331120
Lei C, Berra L, Rezoagli E, Yu B, Dong H, Yu S, Hou L, Chen M, Chen W, Wang H, Zheng Q, Shen J, Jin Z, Chen T, Zhao R, Christie E, Sabbisetti VS, Nordio F, Bonventre JV, Xiong L, Zapol WM (2018) Nitric oxide decreases acute kidney injury and stage 3 chronic kidney disease after cardiac surgery. Am J Respir Crit Care Med 198:1279–1287. https://doi.org/10.1164/rccm.201710-2150OC
Weisbord SD, Gallagher M, Jneid H, Garcia S, Cass A, Thwin SS, Conner TA, Chertow GM, Bhatt DL, Shunk K, Parikh CR, McFalls EO, Brophy M, Ferguson R, Wu H, Androsenko M, Myles J, Kaufman J, Palevsky PM, Group PT (2018) Outcomes after angiography with sodium bicarbonate and acetylcysteine. N Engl J Med 378:603–614. https://doi.org/10.1056/NEJMoa1710933
Pickkers P, Angus DC, Bass K, Bellomo R, van den Berg E, Bernholz J, Bestle MH, Doi K, Doig CJ, Ferrer R, Francois B, Gammelager H, Pedersen UG, Hoste E, Iversen S, Joannidis M, Kellum JA, Liu K, Meersch M, Mehta R, Millington S, Murray PT, Nichol A, Ostermann M, Pettila V, Solling C, Winkel M, Young PJ, Zarbock A, Investigators R (2024) Phase-3 trial of recombinant human alkaline phosphatase for patients with sepsis-associated acute kidney injury (REVIVAL). Intensive Care Med 50:68–78. https://doi.org/10.1007/s00134-023-07271-w
Meersch M, Schmidt C, Hoffmeier A, Van Aken H, Wempe C, Gerss J, Zarbock A (2017) Prevention of cardiac surgery-associated AKI by implementing the KDIGO guidelines in high risk patients identified by biomarkers: the PrevAKI randomized controlled trial. Intensive Care Med 43:1551–1561. https://doi.org/10.1007/s00134-016-4670-3
Thielmann M, Corteville D, Szabo G, Swaminathan M, Lamy A, Lehner LJ, Brown CD, Noiseux N, Atta MG, Squiers EC, Erlich S, Rothenstein D, Molitoris B, Mazer CD (2021) Teprasiran, a small interfering RNA, for the prevention of acute kidney injury in high-risk patients undergoing cardiac surgery: a randomized clinical study. Circulation 144:1133–1144. https://doi.org/10.1161/CIRCULATIONAHA.120.053029
Pickkers P, Mehta RL, Murray PT, Joannidis M, Molitoris BA, Kellum JA, Bachler M, Hoste EAJ, Hoiting O, Krell K, Ostermann M, Rozendaal W, Valkonen M, Brealey D, Beishuizen A, Meziani F, Murugan R, de Geus H, Payen D, van den Berg E, Arend J, Investigators S-A (2018) Effect of human recombinant alkaline phosphatase on 7-day creatinine clearance in patients with sepsis-associated acute kidney injury: a randomized clinical trial. JAMA 320:1998–2009. https://doi.org/10.1001/jama.2018.14283
Bhaskhar N, Ip W, Chen JH, Rubin DL (2023) Clinical outcome prediction using observational supervision with electronic health records and audit logs. J Biomed Inform 147:104522. https://doi.org/10.1016/j.jbi.2023.104522
Prowle JR, Bagshaw SM, Forni LG (2024) Tackling sepsis-associated AKI: are there any chances of REVIVAL with new approaches? Intensive Care Med 50:131–133. https://doi.org/10.1007/s00134-023-07294-3
van Raalte DH, Bjornstad P, Heerspink HJL, Persson F, Cherney DZI (2021) Importance of standardizing renal outcomes in clinical trials: illustration by recent sodium glucose cotransporter 2 inhibitor studies. Kidney Int 99:768–770. https://doi.org/10.1016/j.kint.2020.12.006
Shaw A (2011) Models of preventable disease: contrast-induced nephropathy and cardiac surgery-associated acute kidney injury. Contrib Nephrol 174:156–162. https://doi.org/10.1159/000329387
Prischl FC, Rossing P, Bakris G, Mayer G, Wanner C (2021) Major adverse renal events (MARE): a proposal to unify renal endpoints. Nephrol Dial Transplant 36:491–497. https://doi.org/10.1093/ndt/gfz212
EMA Committee for Medicinal Products for Human Use (CHMP) (2016) Reflection paper on assessment of cardiovascular safety profile of medicinal products. https://www.ema.europa.eu/en/assessment-cardiovascular-safety-profile-medicinal-products-scientific-guideline, Accessed 6 Feb 2024
Ferreira-Gonzalez I, Busse JW, Heels-Ansdell D, Montori VM, Akl EA, Bryant DM, Alonso-Coello P, Alonso J, Worster A, Upadhye S, Jaeschke R, Schunemann HJ, Permanyer-Miralda G, Pacheco-Huergo V, Domingo-Salvany A, Wu P, Mills EJ, Guyatt GH (2007) Problems with use of composite end points in cardiovascular trials: systematic review of randomised controlled trials. BMJ 334:786. https://doi.org/10.1136/bmj.39136.682083.AE
Kip KE, Hollabaugh K, Marroquin OC, Williams DO (2008) The problem with composite end points in cardiovascular studies: the story of major adverse cardiac events and percutaneous coronary intervention. J Am Coll Cardiol 51:701–707. https://doi.org/10.1016/j.jacc.2007.10.034
Bosco E, Hsueh L, McConeghy KW, Gravenstein S, Saade E (2021) Major adverse cardiovascular event definitions used in observational analysis of administrative databases: a systematic review. BMC Med Res Methodol 21:241. https://doi.org/10.1186/s12874-021-01440-5
Hicks KA, Tcheng JE, Bozkurt B, Chaitman BR, Cutlip DE, Farb A, Fonarow GC, Jacobs JP, Jaff MR, Lichtman JH, Limacher MC, Mahaffey KW, Mehran R, Nissen SE, Smith EE, Targum SL, American College of C, American Heart A (2015) 2014 ACC/AHA Key Data Elements and Definitions for Cardiovascular Endpoint Events in Clinical Trials: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Data Standards (Writing Committee to Develop Cardiovascular Endpoints Data Standards). Circulation 132:302–361. https://doi.org/10.1161/CIR.0000000000000156
Hicks KA, Mahaffey KW, Mehran R, Nissen SE, Wiviott SD, Dunn B, Solomon SD, Marler JR, Teerlink JR, Farb A, Morrow DA, Targum SL, Sila CA, Hai MTT, Jaff MR, Joffe HV, Cutlip DE, Desai AS, Lewis EF, Gibson CM, Landray MJ, Lincoff AM, White CJ, Brooks SS, Rosenfield K, Domanski MJ, Lansky AJ, McMurray JJV, Tcheng JE, Steinhubl SR, Burton P, Mauri L, O’Connor CM, Pfeffer MA, Hung HMJ, Stockbridge NL, Chaitman BR, Temple RJ, Standardized Data Collection Standardized Data Collection for Cardiovascular Trials I (2018) 2017 Cardiovascular and stroke endpoint definitions for clinical trials. Circulation 137:961–972. https://doi.org/10.1161/CIRCULATIONAHA.117.033502
Lameire NH, Levin A, Kellum JA, Cheung M, Jadoul M, Winkelmayer WC, Stevens PE, Conference P (2021) Harmonizing acute and chronic kidney disease definition and classification: report of a Kidney Disease: Improving Global Outcomes (KDIGO) Consensus Conference. Kidney Int 100:516–526. https://doi.org/10.1016/j.kint.2021.06.028
Zarbock A, Forni LG, Ostermann M, Ronco C, Bagshaw SM, Mehta RL, Bellomo R, Kellum JA (2023) Designing acute kidney injury clinical trials. Nat Rev Nephrol. https://doi.org/10.1038/s41581-023-00758-1
Acknowledgements
The authors would like to thank Fernando Godinho Zampieri and John A. Kellum for critically reviewing our paper and providing comments and suggestions.
Funding
Open Access funding provided by The University of Tokyo.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by AM, RI, and KD. Analysis was performed by AM. The first draft of the manuscript was written by AM and RB. All authors critically revised the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
There are no financial or any other conflicts of interest, or competing interests associated with this work, writing, and the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Maeda, A., Inokuchi, R., Bellomo, R. et al. Heterogeneity in the definition of major adverse kidney events: a scoping review. Intensive Care Med 50, 1049–1063 (2024). https://doi.org/10.1007/s00134-024-07480-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-024-07480-x