Abstract
Mechanical ventilation may have adverse effects on both the lung and the diaphragm. Injury to the lung is mediated by excessive mechanical stress and strain, whereas the diaphragm develops atrophy as a consequence of low respiratory effort and injury in case of excessive effort. The lung and diaphragm-protective mechanical ventilation approach aims to protect both organs simultaneously whenever possible. This review summarizes practical strategies for achieving lung and diaphragm-protective targets at the bedside, focusing on inspiratory and expiratory ventilator settings, monitoring of inspiratory effort or respiratory drive, management of dyssynchrony, and sedation considerations. A number of potential future adjunctive strategies including extracorporeal CO2 removal, partial neuromuscular blockade, and neuromuscular stimulation are also discussed. While clinical trials to confirm the benefit of these approaches are awaited, clinicians should become familiar with assessing and managing patients’ respiratory effort, based on existing physiological principles. To protect the lung and the diaphragm, ventilation and sedation might be applied to avoid excessively weak or very strong respiratory efforts and patient-ventilator dysynchrony.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This review explains the principles of lung and diaphragm-protective mechanical ventilation. The overall aim of this approach is to limit the adverse effects of mechanical ventilation on the lung and the diaphragm at the same time. This requires understanding of the pathophysiology of ventilator-induced lung injury, critical illness-associated diaphragm weakness and especially respiratory drive. We discuss clinical applicable techniques to monitor lung and diaphragm function, and how to use these techniques to optimize ventilator settings and sedation. Future techniques that allow to control respiratory drive are discussed. |
Introduction
Lung and diaphragm-protective mechanical ventilation is a novel approach that aims to limit side effects of mechanical ventilation in critically ill patients. This approach integrates the principles of lung-protective ventilation with the new concept of diaphragm-protective ventilation in an effort to simultaneously protect both organs. The approach centers on optimizing patient respiratory effort to avoid lung and diaphragm injury while maintaining acceptable respiratory homeostasis. Ultimately, the goal of the approach is to reduce the duration of mechanical ventilation, enhance survival, accelerate recovery, and prevent long-term disability in patients with acute respiratory failure.
Principles and rationale
Principles of lung-protective ventilation
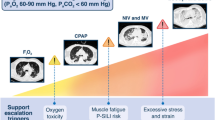
Lung-protective ventilation can best be understood in terms of limiting global and regional mechanical stress (pressure applied to the lung) and strain (deformation from resting shape) (Fig. 1). Lung injury may occur from overdistension (volutrauma/barotrauma), repetitive tidal recruitment and collapse (atelectrauma), both resulting from heterogeneous insufflation of patchy alveolar flooding or collapsed alveoli [1]. Importantly, lung injury may occur irrespective of whether the ventilator (ventilator-induced lung injury, VILI), patient breathing effort (patient self-inflicted lung injury, P-SILI), or both together are generating the forces applied to the lung [2].
Bedside measures of stress are available (changes in transpulmonary pressure, driving pressure), but not for measuring the resulting strain, making it challenging to appropriately individualize mechanical ventilation settings to maximize lung protection. Furthermore, even if global stress can be measured quite precisely using transpulmonary pressure calculated from airway and esophageal pressure, the effect of gravity on the edematous lung makes the distribution of collapse and aeration very uneven between the dependent and non-dependent lung regions; therefore, global indices do not reflect regional stress or strain. To minimize total stress and strain, dependent regions (usually prone to atelectasis) often require recruitment while non-dependent regions (usually well ventilated) require relief of overdistension.
During invasive ventilation, tidal volume (VT) is routinely scaled to predicted body weight (PBW), which correlates with lung volume in healthy subjects. This correlation is much less accurate in patients with acute respiratory distress syndrome (ARDS) because of alveolar flooding and atelectasis, resulting in a “baby lung” much smaller than the predicted lung volume [3, 4]. Using the driving pressure to scale tidal volume to respiratory system compliance (Crs, VT/Crs = airway driving pressure, ΔPaw) is particularly attractive because Crs is affected by the aerated lung size and could, therefore, better reflect the global strain (VT/baby lung). Driving pressure correlates with ARDS outcomes among patients with the same VT/PBW [5] and may be useful to guide tidal ventilation, although its role remains to be tested in a prospective trial. It should be acknowledged that static airway pressure is not a very reliable marker of lung stress (both at end-inspiration and end-expiration), because it reflects contributions from both the lung and chest wall (two pressures acting in series). Obese patients are an example where high intrathoracic pressure (and therefore higher plateau pressure) exist because of the weight imposed by the chest wall [6]. Lung stress is preferably measured as transpulmonary pressure (PL), which allows to quantify the contribution of the lung and chest wall to changes in airway pressure.
Principles of diaphragm-protective ventilation
The respiratory muscle pump drives alveolar ventilation and is composed of a number of skeletal muscles acting in a highly organized fashion. The diaphragm is the primary muscle of inspiration and the lateral abdominal wall muscles are the most prominent expiratory muscles [7]. Mechanical ventilation is employed to unload the respiratory muscle pump and limit the consequences of high breathing effort (e.g., dyspnea sensation, respiratory failure, possible respiratory muscle injury). However, mechanical ventilation delivered as the predominant breathing source can also lead to diaphragm atrophy and injury with a substantial deleterious impact on patient outcome [8]. Clinical studies demonstrate that after 24 h of mechanical ventilation, 64% of patients exhibit diaphragm weakness [9] and at the time of weaning, diaphragm weakness is present in up to 80% of patients with weaning difficulties [10]. While many factors contribute to diaphragm weakness in the critically ill [11], both excessive and insufficient respiratory muscle unloading rapidly result in deleterious changes in diaphragm structure and function [11]. Low respiratory muscle effort, due to ventilator over-assist or sedation, may result in muscle atrophy, while high effort has been associated with load-induced injury (Fig. 1). In a landmark study, Levine et al. demonstrated the development of diaphragm disuse atrophy in brain dead patients on controlled mechanical ventilation [12] and subsequent studies confirmed the presence of time-dependent fiber atrophy in the diaphragm of ventilated patients [13, 14]. In line with these findings, ultrasound studies demonstrated that low diaphragm effort during mechanical ventilation is associated with time-dependent development of atrophy [15] and that atrophy is associated with poor outcomes [8]. It may be hypothesized that patients are at risk of developing load-induced diaphragm injury, as suggested by the presence of fiber injury, sarcomeric disruption, inflammation and contractile dysfunction in biopsies [13] and acute increases in diaphragm thickness on ultrasound [15]—this hypothesis requires further confirmation.
Taken together, these considerations suggest that the diaphragm might be protected by titrating ventilation and sedation to restore early diaphragm activity while avoiding excess respiratory effort. The various lines of physiological and clinical evidence suggesting that a respiratory effort level similar to that of resting quiet breathing is probably optimal for both lung and diaphragm protection were recently summarized elsewhere [16].
Monitoring strategies
To implement lung and diaphragm-protective mechanical ventilation, the variables that mediate injury, principally lung stress and respiratory effort, should be monitored. The available monitoring techniques, their advantages and disadvantages, and proposed specific targets are summarized in Table 1.
Airway driving pressure, ΔPaw (i.e., plateau pressure—PEEPtot), is a measure that aims to estimate global tidal lung stress [5]. ΔPaw can be measured either during controlled or assisted ventilation by manual or automated short end-inspiratory and end-expiratory occlusions [17,18,19] Importantly, ΔPaw is determined by transpulmonary driving pressure (ΔPL) and driving pressure across the chest wall (ΔPcw); thus changes in chest wall elastance affect ΔPaw, without affecting lung stress [20]. Because pendelluft and regional variations in lung stress are “dynamic” phenomena that cannot be detected under static conditions, the risk of excess regional lung stress during assisted breathing may be more accurately estimated by dynamic ΔPL (ΔPL,dyn, peak PL—end-expiratory PL) rather than by static measures like ΔPaw [21, 22]. Esophageal pressure (Pes) monitoring, as an estimate of pleural pressure, can provide information about both the predisposition to end-expiratory collapse and atelectasis (end-expiratory PL) and alveolar overdistension within the baby lung (elastance-derived plateau PL) [23].
Monitoring and controlling respiratory muscle effort are major challenges in implementing lung and diaphragm-protective mechanical ventilation. The gold standard to quantify global respiratory muscle effort is the esophageal pressure–time product (PTP), while the PTP of the transdiaphragmatic pressure (Pdi, i.e., difference between gastric pressure (Pga) and Pes) during inspiration provides a measure of diaphragmatic effort [24]. The amplitude of Pes or Pdi during tidal breathing provides a simple estimate of the pressure generated by all respiratory muscles (Pes), or the diaphragm (Pdi), whereas the expiratory increase in Pga reflects expiratory muscle activity. The diaphragm electrical activity (EAdi) is the most precise surrogate of respiratory drive and correlates with indices of effort [25] but with considerable variability between patients. Also, values for peak EAdi in young healthy subjects during tidal breathing may vary between 4 and 29 μV [26]. Nevertheless, changes in EAdi are useful to monitor changes in patient’s respiratory drive and effort, especially to identify patients at risk for ventilator over-assistance. Finally, Pes or EAdi can complement ventilator waveform analysis to facilitate the identification of patient-ventilator dyssynchronies.
Other less invasive techniques are available to monitor patient breathing efforts during mechanical ventilation at the bedside. Airway occlusion pressure (P0.1), the deflection in Paw during the first 0.1 s of an inspiratory effort against an occluded airway, is an estimate of the respiratory drive and can be used to detect both very low and high effort [27]. The maximum deflection of Paw during a whole breath occlusion (ΔPocc) has been recently shown to accurately detect excessive respiratory muscle pressure (Pmus) or ΔPL,dyn; this maneuver can also be used to assess different forms of patient-ventilator dyssynchrony [28, 29]. Ultrasound can be used to visualize and quantify the thickening of the diaphragm during inspiration in the zone of apposition (thickening fraction, TFdi) [30]. TFdi provides an index of diaphragmatic contractility and correlates reasonably well with inspiratory effort (ΔPes) and EAdi [31].
In conclusion, although estimation of pleural pressure using an esophageal balloon appears to be the preferred technique to quantify lung stress and respiratory effort, the technique is currently not widely implemented; moreover, the potential impact on patient outcome remains to be determined in clinical studies. We suggest routine monitoring of tidal volume, inspiratory plateau pressures and airway driving pressure to limit lung injury, and P0.1 to monitor respiratory drive and prevent inadequate effort (Table 1).
Clinical strategies to facilitate lung and diaphragm-protective ventilation
Several strategies can be used to facilitate lung and diaphragm protective ventilation, including modulation of ventilator inspiratory and expiratory assist, drugs that modify respiratory drive and/or effort, extracorporeal CO2 removal (ECCO2R) and electrical stimulation of the respiratory muscles, as shown in Fig. 2. Here, we will briefly discuss these different strategies.
Inspiratory ventilator settings
A lung and diaphragm-protective ventilation approach aims to minimize lung stress and strain while limiting diaphragm atrophy and injury. To achieve these goals, inspiratory ventilator settings can be adjusted to (1) modulate the patient’s inspiratory effort, (2) minimize the dynamic lung stress, and (3) prevent or correct patient-ventilator dyssynchrony or any form of mismatch between needs and support.
Titrating the inspiratory ventilator settings to optimize respiratory effort requires a thorough understanding of the control of breathing under mechanical ventilation [32, 33], acknowledging that the control of breathing system responds to changes in ventilatory demands by modifying inspiratory effort (and thus tidal volume) to a greater extent than respiratory rate [34]. Therefore, the inspiratory ventilator settings will affect the inspiratory effort by modifying the delivered tidal volume, and thus, in spontaneously breathing patients, increasing pressure or volume assist will increase the delivered tidal volume and reduce the inspiratory effort (as respiratory drive depends mainly on the chemoreflex control of arterial pH). Excessive assist, resulting in a tidal volume that is higher than the patient’s demands, may almost abolish the patient’s the inspiratory effort, and as such promote diaphragmatic atrophy. However, increasing inspiratory support may not attenuate inspiratory effort in the presence of high respiratory drive due to stimuli other than arterial pH/PaCO2, such as pain, anxiety, or stimulation of peripheral lung receptors by lung edema or inflammation [32]. In such case, transpulmonary pressure (and hence dynamic lung stress) may progressively increase with increasing inspiratory support. Increasing FiO2 to increase PaO2 and reduce the hypoxic stimulus to breathe may alleviate increased respiratory drive in some patients (hyperoxemia is not required to achieve this effect) [35].
In a volume-targeted mode, the patient’s effort will be modified mainly by the set tidal volume and the flow delivery profile (flow pattern and peak flow). In pressure-targeted modes, the delivered tidal volume, and thus the patient’s inspiratory effort, is influenced by the set inspiratory pressure, rise time and cycling-off criterion, and of course the mechanical properties of the respiratory system [36]. Irrespective of the mode of assist, the delivered tidal volume and respiratory effort will together determine global and regional lung stress, depending on the mechanical properties of the respiratory system [37].
Neurally adjusted ventilatory assist (NAVA) delivers inspiratory assist proportional to the electrical activity of the diaphragm [38]. Increasing inspiratory assist will reduce diaphragm electrical activity (and vice versa) over a wide range of respiratory demand, and consequently tidal volume remains relatively stable over a wide range of assist [39]. In theory, pulmonary reflex mechanisms prevent patients from spontaneously inspiring large tidal volumes and NAVA may therefore facilitate lung-protective ventilation. Also, diaphragm inactivity due to over-assistance is unlikely in NAVA, as low diaphragm activity will immediately reduce inspiratory assist. Future studies should confirm the role of NAVA in lung and diaphragm-protective ventilation, but recent randomized trials suggest clinical benefit of NAVA (reduced time on the ventilator) compared to pressure support mode [40, 41].
Expiratory ventilator settings
The expiratory ventilator setting (i.e., positive end-expiratory pressure, PEEP) has been traditionally adjusted to optimize oxygenation and/or lung mechanics [42, 43]. A higher PEEP ventilation strategy (of which there are several, generally resulting in 15 ± 4 cmH2O) is currently recommended over lower PEEP (approximately 9 ± 3 cmH2O) in moderate and severe ARDS [44]. In the presence of spontaneous breathing during mechanical ventilation, a higher PEEP strategy offers several additional potential advantages to facilitate lung and diaphragm-protective ventilation (Fig. 1). First, in patients with significant lung recruitability, PEEP reduces the amount of atelectatic ‘solid-like’ lung and, therefore, can achieve a more homogeneous distribution of the tidal pleural pressure swing (∆Ppl) over the whole lung surface following a diaphragmatic contraction. The even distribution of inspiratory dynamic stress can diminish injurious asymmetric inflation associated with spontaneous effort (i.e., pendelluft), reducing regional lung stress in dependent lung regions [45]. Second, by increasing end-expiratory lung volume, forcing the diaphragm to operate at a shorter length and thereby impairing diaphragm neuromuscular coupling [46, 47], increased PEEP can attenuate the force generated by diaphragmatic contraction [48]. Indeed, several clinical studies provide indirect evidence to suggest that higher PEEP may render spontaneous effort less injurious in patients with acute respiratory failure before intubation [49], in patients with ARDS [45, 50], and in pediatric patients with lung injury [51].
On the other hand, preliminary experimental evidence suggests that if the diaphragm is maintained at a shorter length during acute mechanical ventilation, the diaphragm muscle fibers could adapt to the reduced length by absorbing sarcomeres in series (i.e., longitudinal atrophy) [52]. This may result in fibers overstretching with the release of PEEP during a T-tube weaning trial or after extubation. The possibility of diaphragm weakness resulting from excess PEEP should therefore be borne in mind.
Resolving dyssynchrony
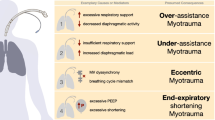
Patient-ventilator dyssynchronies may cause lung and/or diaphragm injury by increasing dynamic lung stress and/or injurious diaphragmatic contractions, respectively. Dyssynchronies may occur during inspiration (flow starvation, short cycles, prolonged insufflation and reverse triggering), during expiration (auto-triggering, ineffective effort) or both during inspiration and expiration (reverse triggering and double triggering). We will briefly discuss dyssynchronies most relevant for lung and diaphragm-protective ventilation; for more extensive discussion of dyssynchronies we refer to other reviews [53].
Reverse triggering, a diaphragmatic contraction triggered by mechanical inflation, is common in fully sedated patients (in whom drive is abolished) [54]. Reverse triggering can induce breath stacking resulting in excessive tidal volumes and high dynamic lung stress [55], and it may create eccentric diaphragm loading conditions with resultant muscle injury [56]. When necessary to avoid breath stacking, reverse triggering can be abolished by neuromuscular blocking agents. Alternatively, the development of reverse triggering may indicate that sedation should be stopped to allow the patient to take control of ventilation.
In patients with relatively high respiratory drive and a low respiratory system time constant, the neural inspiration time may exceed the mechanical inflation (premature cycling). In such cases, the contraction of the inspiratory muscles continues during mechanical expiration and the diaphragm is forced to contract while lengthening (eccentric contraction). In volume-targeted modes, unmet high demands appear as ‘flow-starvation’, a downward curvature of inspiratory Paw, and the patient may experience dyspnea and distress, which can be resolved by increasing inspiratory flow rate using a decelerating flow pattern. Strong inspiratory efforts may result in double-triggering, breath stacking and, therefore, delivery of high tidal volumes. A better match of mechanical and neural inspiratory time can be achieved by increasing ventilator inspiratory time and using a decelerating flow pattern in volume-assist control mode, by decreasing the cycling-off criterion in pressure support mode, or using proportional modes of assist. Importantly, in patients with high respiratory drive, modification of inspiratory time may not suffice to resolve dyssynchrony. Increasing the level of assist to match the patient’s demands should be considered, but, if that results in an injurious high ventilation, other means to decrease the patient's respiratory drive, such as sedation, may be required.
Another dyssynchrony occurring in patients with absent or low respiratory drive is auto-triggering, i.e., the delivery of a ventilator-assisted breath in the absence of patient effort. Auto-triggering due to strong cardiac oscillations transmitted to the Paw or airflow signal is more likely to occur when the respiratory system time constant is low, such as in ARDS. Air leaks and moisture in the ventilator circuit are also common causes of auto-triggering.
Ineffective triggering (or ineffective efforts) develops when a patient's effort fails to trigger a ventilator-delivered breath. Ineffective triggering is generally the consequence of weak inspiratory efforts, either from low respiratory drive due to sedation, metabolic alkalosis or excessive ventilatory assist, or because of diaphragm weakness. When the respiratory system time constant is high, (i.e., obstructive lung disease), ventilator over-assistance results in delayed cycling, dynamic hyperinflation, and increased intrinsic PEEP, predisposing to ineffective triggering. Decreasing the level of assist can therefore alleviate ineffective efforts [57]. Over-assistance in assisted ventilation can also induce apneas during sleep.
Interestingly, several studies have demonstrated that NAVA improves patient-ventilator interaction, especially reducing the risks of ineffective efforts and over-assist [39, 58]. Whether the reduced duration of mechanical ventilation reported in some NAVA trials [40, 41] results from improved patient-ventilator interaction remains to be investigated.
Sedation strategies
Sedation can facilitate lung and diaphragm-protective ventilation by ameliorating, when present, excessive respiratory effort. Complete suppression of respiratory drive and effort with sedation can also contribute to diaphragm disuse atrophy. A judicious approach to sedation is key and monitoring of respiratory drive and effort may be helpful in selecting the sedation strategy that facilitates lung and diaphragm-protective ventilation. Before administering sedation to address excessive respiratory drive or ventilator dyssynchrony, ventilator settings should be adjusted and other factors increasing respiratory drive such as metabolic acidosis or pain should be addressed. Relying on sedation alone to enhance patient–ventilator interaction without addressing these issues can paradoxically exacerbate dyssynchrony, prolong mechanical ventilation, and exacerbate diaphragm dysfunction [59]. Recent clinical practice guidelines have recommended an “analgesia-first approach” to minimize the risk of excessive sedation as opioids during mechanical ventilation were associated with less dyssynchrony and depressed consciousness in comparison to sedative-based approaches [60].
Nevertheless, when elevated respiratory drive cannot otherwise be resolved, sedatives can attenuate the ventilatory response to hypoxemia and hypercapnia and cortical input to the respiratory centres [33] (Table 2). Propofol and benzodiazepines are gamma-aminobutyric acid (GABA) agonists known to cause respiratory depression, primarily by reducing the amplitude of respiratory effort [61,62,63]. Because benzodiazepines are associated with a high risk of delirium and prolonged mechanical ventilation [64], propofol is the preferred sedative of choice for controlling high respiratory drive. Because propofol or benzodiazepines reduce the amplitude of inspiratory effort, ineffective triggering may develop as sedation depth increases [61]. Inhalational sedation offers a potential alternative for controlling respiratory effort though clinical experience is limited to date [65]. To avoid excessive sedation, strategies aimed at active titration of sedatives or daily interruption of sedation should be employed and respiratory drive and effort should be monitored closely.
For patients without excessive breathing effort (Table 2), a multimodal analgesia approach that minimizes opiate use is recommended to avoid diaphragm inactivity. Dexmedetomidine is a selective alpha-2 agonist which, in contrast to propofol and benzodiazepines, provides sedation, anxiolysis, and analgesia without respiratory depression [66]. This property makes it an interesting drug of choice to preserve awareness and diaphragm contractility and at the same time limiting excess delirium risk in agitated patients without elevated respiratory drive.
Prone positioning
The prone position has been used for decades in early ARDS to improve oxygenation and over time an appreciation for the lung-protective benefit of prone positioning has emerged [67]. As the amount of lung tissue is larger in dorsal lung regions, gravitational forces generate more dependent atelectasis in the supine position compared to prone position. Therefore, ventilation-perfusion matching is improved in the prone position and, more importantly, the energy applied to the lung by mechanical ventilation is distributed among more (non-atelectatic) alveoli, reducing lung stress. This is the putative basis for the observed mortality benefit of prone positioning in patients with ARDS [68]. The mechanistic benefits of prone positioning may also apply under assisted ventilation with spontaneous breathing, because the lung recruitment accrued by prone positioning may attenuate ‘solid-like’ lung behaviour and reduce effort-dependent regional lung stress. Prone positioning improves oxygenation in spontaneously breathing patients with COVID-19 pneumonia [69]; it is possible that prone positioning could also reduce the risk of patient self-inflicted lung injury [70]. Thus, prone positioning might facilitate safe spontaneous breathing and diaphragm-protective ventilation as well as lung protection.
Future approaches to lung and respiratory muscle-protective ventilation
Extracorporeal carbon dioxide removal
Eliminating CO2 is the primary purpose of alveolar ventilation. ECCO2R reduces the ventilatory demands, decreasing the respiratory effort, and thus may ameliorate dynamic lung stress. ECCO2R is feasible and effective in reducing tidal volume, driving pressure, and mechanical power in patients with ARDS [71]. In spontaneously breathing patients, ECCO2R can dampen respiratory drive and effort [72], theoretically reducing the requirement for ventilatory support or sedation to control respiratory effort. Karagiannidis et al. showed that increasing sweep gas flow, increasing CO2 elimination, in ARDS patients undergoing extracorporeal membrane oxygenation (ECMO) reduced respiratory drive, estimated by EAdi [73]. Mauri et al. [7] also showed that higher ECCO2R support reduced P0.1, respiratory muscle effort, and transpulmonary pressure in spontaneously breathing patients recovering from severe ARDS [74]. Pilot clinical studies have explored the extreme possibility of extubating severe ARDS patients early after intubation by means of ECCO2R: preliminary results were encouraging but they also recognized the need to identify the subgroup of patients with a high probability of success [75,76,77].
Despite the appeal and physiological rationale of this strategy, there are relevant limitations. First, in some patients, non-chemoreceptive stimuli (pain, agitation, discomfort, metabolic acidosis, lung mechanical stimuli) may predominate and high respiratory drive may persist despite ECCO2R [78]. Second, ECCO2R requires full anticoagulation and the risk of bleeding is not insubstantial [79]. Third, the application of ECCO2R may exacerbate hypoxemia by various mechanisms [80].
Partial neuromuscular blockade
Complete neuromuscular blockade may increase the risk for diaphragm disuse atrophy and increases sedation requirements. Low-dose neuromuscular blockers (“partial neuromuscular blockade”) is an interesting compromise between total paralysis and strenuous breathing efforts, particularly when respiratory effort does inadequately respond to titration of ventilatory support or sedation. The feasibility of partial neuromuscular blockade has been evaluated in a proof of concept study in patients with moderate ARDS and high respiratory drive on partially supported modes [81]. Titration of rocuronium decreased tidal volume from approximately 9 mL/kg to approximately 6 mL/kg while maintaining Pdi at approximately 5 cmH2O (within the physiological range for diaphragm activity in healthy subjects). These preliminary findings suggest that partial neuromuscular blockade could be a feasible approach to achieving lung and diaphragm-protective ventilation targets in patients with high respiratory effort. Importantly, partial neuromuscular blockade does not reduce respiratory drive, but only the mechanical consequences of high drive. This dissociation between central drive and respiratory muscle mechanical output may result in dyspnea [33]; adequate relief of dyspnea and distress must be ensured by judicious application of sedatives and opioids. Future clinical studies should confirm the safety and efficacy of prolonged partial neuromuscular blockade in ventilated patients.
Neuromuscular stimulation
Neuromuscular stimulation (“pacing”) uses electrical currents to generate muscle contraction in the absence of volitional efforts, making it an attractive intervention in incapacitated critically ill patients. There is growing interest in neuromuscular stimulation as a novel strategy to preserve or restore respiratory muscle activity and, in turn, to prevent or treat ICU-acquired diaphragm weakness. In addition, by inducing diaphragm contractions, neuromuscular stimulation may improve lung aeration of dependent lung regions [82]. Pacing must be synchronized with the ventilator and potentially injurious inspiratory efforts must be avoided.
There is as yet no clinical evidence of benefit from diaphragm pacing in ICU patients. Direct stimulation of the phrenic nerves by surgically implanted electrodes has been employed to restore spontaneous ventilation in patients with high-level spinal cord injury and central hypoventilation syndrome [83]. The feasibility of direct pacing using temporary implanted electrodes for the prevention of diaphragm dysfunction is currently under investigation in cardiac surgery patients identified to be at risk for prolonged mechanical ventilation (NCT04309123). Preclinical work showed that this technique could reduce the development of diaphragm type II fiber atrophy [84, 85]. Recently, Reynolds et al. presented a first-in-human series of temporary transvenous phrenic nerve pacing in surgical patients and showed that this technology delivered safe and effective diaphragm contractions [86]. This strategy is currently being studied as potential intervention for improving diaphragm strength in difficult-to-wean patients (NCT03096639). The role of transvenous phrenic nerve pacing for the prevention of diaphragm disuse atrophy remains to be investigated.
Neuromuscular stimulation strategies targeting the expiratory muscles of ICU patients are less well studied. This is surprising, as stimulation of the expiratory abdominal wall muscles can be employed noninvasively via surface electrodes placed over the abdominal wall. Feasibility of a breath-synchronized expiratory muscle stimulation technique during the early phase of mechanical ventilation was recently demonstrated with promising results [87] and its efficacy is under investigation (NCT03453944).
Summary and future directions
Clinicians caring for mechanically ventilated patients are generally well aware of the risk of causing barotrauma, volutrauma, and atelectrauma. Given the mounting evidence of clinically important diaphragm atrophy and injury, consideration must also be given to protecting the diaphragm. Based on the foregoing discussion about ventilation and sedation, a basic algorithm and approach to lung and diaphragm-protective ventilation is presented in Fig. 3. Clinical trials testing new ventilation algorithms and sedation strategies targeted at optimizing respiratory effort are required to confirm the benefit of the lung and diaphragm-protective approach outlined in this paper. The benefit of adjunctive strategies such as ECCO2R, partial neuromuscular blockade and phrenic nerve stimulation requires further evaluation, in particular to identify the subpopulations of patients most likely to benefit from these more costly and invasive interventions. For the present, we encourage clinicians to incorporate routine monitoring of respiratory drive and effort in their clinical practice and to adjust the ventilator to achieve a physiological level of effort where possible while carefully attending to the effect on lung stress.
Clinical-physiological pathway for achieving lung and diaphragm-protective ventilation targets. It should be stressed that at each step clinical evaluation of the patient, including signs of high breathing effort, agitation, and over-sedation is of major importance and should be interpreted together with clinical-physiological measurements as outlined in this pathway. ΔP: change in airway pressure during inspiration; P0.1: decrease in airway pressure during the first 100 ms of inspiratory effort against an occluded airway; PaCO2: arterial carbon dioxide tension; PEEP: positive end-expiratory pressure; Pes: esophageal pressure; PL: transpulmonary pressure; Pocc: airway pressure deflection during a whole breath occlusion; RR: respiratory rate; VT: tidal volume
References
Slutsky AS, Ranieri VM (2013) Ventilator-induced lung injury. N Engl J Med 369:2126–2136. https://doi.org/10.1056/NEJMra1208707
Brochard L, Slutsky A, Pesenti A (2017) Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am J Respir Crit Care Med 195:438–442. https://doi.org/10.1164/rccm.201605-1081CP
Chiumello D, Carlesso E, Cadringher P et al (2008) Lung stress and strain during mechanical ventilation for acute respiratory distress syndrome. Am J Respir Crit Care Med 178:346–355. https://doi.org/10.1164/rccm.200710-1589OC
Beitler JR, Majumdar R, Hubmayr RD et al (2016) Volume delivered during recruitment maneuver predicts lung stress in acute respiratory distress syndrome. Crit Care Med 44:91–99. https://doi.org/10.1097/CCM.0000000000001355
Amato MB, Meade MO, Slutsky AS et al (2015) Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med 372:747–755. https://doi.org/10.1056/NEJMsa1410639
Coudroy R, Vimpere D, Aissaoui N et al (2020) Prevalence of complete airway closure according to body mass index in acute respiratory distress syndrome: pooled cohort analysis. Anesthesiology (Philadelphia) 133:867–878. https://doi.org/10.1097/ALN.0000000000003444
Shi ZH, Jonkman A, de Vries H et al (2019) Expiratory muscle dysfunction in critically ill patients: towards improved understanding. Intensive Care Med 45:1061–1071. https://doi.org/10.1007/s00134-019-05664-4
Goligher EC, Dres M, Fan E et al (2018) Mechanical ventilation-induced diaphragm atrophy strongly impacts clinical outcomes. Am J Respir Crit Care Med 197:204–213. https://doi.org/10.1164/rccm.201703-0536OC
Demoule A, Jung B, Prodanovic H et al (2013) Diaphragm dysfunction on admission to the intensive care unit. Prevalence, risk factors, and prognostic impact-a prospective study. Am J Respir Crit Care Med 188:213–219. https://doi.org/10.1164/rccm.201209-1668OC
Dres M, Dube BP, Mayaux J et al (2017) Coexistence and impact of limb muscle and diaphragm weakness at time of liberation from mechanical ventilation in medical intensive care unit patients. Am J Respir Crit Care Med 195:57–66. https://doi.org/10.1164/rccm.201602-0367OC
Dres M, Goligher EC, Heunks LMA, Brochard LJ (2017) Critical illness-associated diaphragm weakness. Intensive Care Med 43:1441–1452. https://doi.org/10.1007/s00134-017-4928-4
Levine S, Nguyen T, Taylor N et al (2008) Rapid disuse atrophy of diaphragm fibers in mechanically ventilated humans. N Engl J Med 358:1327–1335. https://doi.org/10.1056/NEJMoa070447
Hooijman PE, Beishuizen A, Witt CC et al (2015) Diaphragm muscle fiber weakness and ubiquitin-proteasome activation in critically ill patients. Am J Respir Crit Care Med 191:1126–1138. https://doi.org/10.1164/rccm.201412-2214OC
Jaber S, Petrof BJ, Jung B et al (2011) Rapidly progressive diaphragmatic weakness and injury during mechanical ventilation in humans. Am J Respir Crit Care Med 183:364–371. https://doi.org/10.1164/rccm.201004-0670OC
Goligher EC, Fan E, Herridge MS et al (2015) Evolution of diaphragm thickness during mechanical ventilation. Impact of inspiratory effort. Am J Respir Crit Care Med 192:1080–1088. https://doi.org/10.1164/rccm.201503-0620OC
Goligher EC, Dres M, Patel BK et al (2020) Lung and diaphragm-protective ventilation. Am J Respir Crit Care Med. https://doi.org/10.1164/rccm.202003-0655CP
Bellani G, Grassi A, Sosio S, Gatti S, Kavanagh BP, Pesenti A, Foti G (2019) Driving pressure is associated with outcome during assisted ventilation in acute respiratory distress syndrome. Anesthesiology 131:594–604. https://doi.org/10.1097/ALN.0000000000002846
Vaporidi K, Psarologakis C, Proklou A et al (2019) Driving pressure during proportional assist ventilation: an observational study. Ann Intensive Care 9:1–4. https://doi.org/10.1186/s13613-018-0477-4
Soundoulounaki S, Akoumianaki E, Kondili E, Pediaditis E, Prinianakis G, Vaporidi K, Georgopoulos D (2020) Airway pressure morphology and respiratory muscle activity during end-inspiratory occlusions in pressure support ventilation. Crit Care 24:467–x. https://doi.org/10.1186/s13054-020-03169-x
Mauri T, Yoshida T, Bellani G et al (2016) Esophageal and transpulmonary pressure in the clinical setting: meaning, usefulness and perspectives. Intensive Care Med 42:1360–1373. https://doi.org/10.1007/s00134-016-4400-x
Yoshida T, Torsani V, Gomes S et al (2013) Spontaneous effort causes occult pendelluft during mechanical ventilation. Am J Respir Crit Care Med 188:1420–1427. https://doi.org/10.1164/rccm.201303-0539OC
Yoshida T, Amato MBP, Kavanagh BP (2018) Understanding spontaneous vs. ventilator breaths: impact and monitoring. Intensive Care Med 44:2235–2238. https://doi.org/10.1007/s00134-018-5145-5
Yoshida T, Amato MBP, Grieco DL et al (2018) Esophageal manometry and regional transpulmonary pressure in lung injury. Am J Respir Crit Care Med 197:1018–1026. https://doi.org/10.1164/rccm.201709-1806OC
Doorduin J, van Hees HW, van der Hoeven JG, Heunks LM (2013) Monitoring of the respiratory muscles in the critically ill. Am J Respir Crit Care Med 187:20–27. https://doi.org/10.1164/rccm.201206-1117CP
Bellani G, Mauri T, Coppadoro A et al (2013) Estimation of patient’s inspiratory effort from the electrical activity of the diaphragm. Crit Care Med 41:1483–1491. https://doi.org/10.1097/CCM.0b013e31827caba0
Piquilloud L, Beloncle F, Richard JM, Mancebo J, Mercat A, Brochard L (2019) Information conveyed by electrical diaphragmatic activity during unstressed, stressed and assisted spontaneous breathing: a physiological study. Ann Intensive Care 9:89–1. https://doi.org/10.1186/s13613-019-0564-1
Telias I, Junhasavasdikul D, Rittayamai N et al (2020) Airway occlusion pressure as an estimate of respiratory drive and inspiratory effort during assisted ventilation. Am J Respir Crit Care Med. https://doi.org/10.1164/rccm.201907-1425OC
Dianti J, Bertoni M, Goligher EC (2020) Monitoring patient-ventilator interaction by an end-expiratory occlusion maneuver. Intensive Care Med. https://doi.org/10.1007/s00134-020-06167-3
Bertoni M, Telias I, Urner M et al (2019) A novel non-invasive method to detect excessively high respiratory effort and dynamic transpulmonary driving pressure during mechanical ventilation. Crit Care 23:346. https://doi.org/10.1186/s13054-019-2617-0
Tuinman PR, Jonkman AH, Dres M et al (2020) Respiratory muscle ultrasonography: methodology, basic and advanced principles and clinical applications in ICU and ED patients-a narrative review. Intensive Care Med. doi: https://doi.org/10.1007/s00134-019-05892-8
Goligher EC, Laghi F, Detsky ME et al (2015) Measuring diaphragm thickness with ultrasound in mechanically ventilated patients: feasibility, reproducibility and validity. Intensive Care Med 41:734–2. https://doi.org/10.1007/s00134-015-3724-2
Jonkman AH, de Vries HJ, Heunks LMA (2020) Physiology of the respiratory drive in icu patients: implications for diagnosis and treatment. Crit Care 24:104-z. doi: https://doi.org/10.1186/s13054-020-2776-z
Vaporidi K, Akoumianaki E, Telias I, Goligher EC, Brochard L, Georgopoulos D (2020) Respiratory drive in critically ill patients. pathophysiology and clinical implications. Am J Respir Crit Care Med 201:20–32. https://doi.org/10.1164/rccm.201903-0596SO
Akoumianaki E, Vaporidi K, Georgopoulos D (2019) The injurious effects of elevated or nonelevated respiratory rate during mechanical ventilation. Am J Respir Crit Care Med 199:149–157. https://doi.org/10.1164/rccm.201804-0726CI
Pesenti A, Rossi N, Calori A, Foti G, Rossi GP (1993) Effects of short-term oxygenation changes on acute lung injury patients undergoing pressure support ventilation. Chest 103:1185–1189. https://doi.org/10.1378/chest.103.4.1185
Bonmarchand G, Chevron V, Menard JF, Girault C, Moritz-Berthelot F, Pasquis P, Leroy J (1999) Effects of pressure ramp slope values on the work of breathing during pressure support ventilation in restrictive patients. Crit Care Med 27:715–722. https://doi.org/10.1097/00003246-199904000-00023
Yoshida T, Nakahashi S, Nakamura MAM et al (2017) Volume-controlled ventilation does not prevent injurious inflation during spontaneous effort. Am J Respir Crit Care Med 196:590–601. https://doi.org/10.1164/rccm.201610-1972OC
Sinderby C, Navalesi P, Beck J et al (1999) Neural control of mechanical ventilation in respiratory failure. Nat Med 5:1433–1436. https://doi.org/10.1038/71012
Carteaux G, Córdoba-Izquierdo A, Lyazidi A, Heunks L, Thille AW, Brochard L (2016) Comparison between neurally adjusted ventilatory assist and pressure support ventilation levels in terms of respiratory effort. Crit Care Med 44:503–511. https://doi.org/10.1097/ccm.0000000000001418
Liu L, Xu X, Sun Q et al (2020) Neurally adjusted ventilatory assist versus pressure support ventilation in difficult weaning: a randomized trial. Anesthesiology (Philadelphia) 132:1482–1493. https://doi.org/10.1097/ALN.0000000000003207
Kacmarek RM, Villar J, Parrilla D et al (2020) Neurally adjusted ventilatory assist in acute respiratory failure: a randomized controlled trial. Intensive Care Med pp 1–11. doi: https://doi.org/10.1007/s00134-020-06181-5
Sahetya SK, Goligher EC, Brower RG (2017) Fifty years of research in ARDS. Setting positive end-expiratory pressure in acute respiratory distress syndrome. Am J Respir Crit Care Med 195:1429–1438. https://doi.org/10.1164/rccm.201610-2035CI
Suter PM, Fairley B, Isenberg MD (1975) Optimum end-expiratory airway pressure in patients with acute pulmonary failure. N Engl J Med 292:284–289. https://doi.org/10.1056/NEJM197502062920604
Fan E, Del Sorbo L, Goligher EC et al (2017) An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: Mechanical Ventilation in Adult Patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med 195:1253–1263. https://doi.org/10.1164/rccm.201703-0548ST
Morais CCA, Koyama Y, Yoshida T et al (2018) High positive end-expiratory pressure renders spontaneous effort noninjurious. Am J Respir Crit Care Med 197:1285–1296. https://doi.org/10.1164/rccm.201706-1244OC
Grassino A, Goldman MD, Mead J, Sears TA (1978) Mechanics of the human diaphragm during voluntary contraction: statics. J Appl Physiol Respir Environ Exerc Physiol 44:829–839. https://doi.org/10.1152/jappl.1978.44.6.829
Laghi F, Shaikh HS, Morales D, Sinderby C, Jubran A, Tobin MJ (2014) Diaphragmatic neuromechanical coupling and mechanisms of hypercapnia during inspiratory loading. Respir Physiol Neurobiol 198:32–41. https://doi.org/10.1016/j.resp.2014.03.004
Pengelly LD, Alderson AM, Milic-Emili J (1971) Mechanics of the diaphragm. J Appl Physiol 30:797–805. https://doi.org/10.1152/jappl.1971.30.6.797
Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP (2016) Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA 315:2435–2441. https://doi.org/10.1001/jama.2016.6338
Teggia Droghi M, De Santis Santiago RR, Pinciroli R et al (2018) High positive end-expiratory pressure allows extubation of an obese patient. Am J Respir Crit Care Med 198:524–525. https://doi.org/10.1164/rccm.201712-2411IM
Rossi FS, Costa ELV, Iope DDM et al (2019) Pendelluft detection using electrical impedance tomography in an infant. keep those images in mind. Am J Respir Crit Care Med 200:1427–1429. https://doi.org/10.1164/rccm.201902-0461IM
Lindqvist J, van den Berg M, van der Pijl R et al (2018) Positive end-expiratory pressure ventilation induces longitudinal atrophy in diaphragm fibers. Am J Respir Crit Care Med 198:472–485. https://doi.org/10.1164/rccm.201709-1917OC
Pham T, Brochard LJ, Slutsky AS (2017) Mechanical Ventilation: State of the Art. Mayo Clin Proc 92:1382–1400. https://doi.org/10.1016/j.mayocp.2017.05.004
Akoumianaki E, Lyazidi A, Rey N et al (2013) Mechanical ventilation-induced reverse-triggered breaths: a frequently unrecognized form of neuromechanical coupling. Chest 143:927–938. https://doi.org/10.1378/chest.12-1817
Beitler JR, Sands SA, Loring SH et al (2016) Quantifying unintended exposure to high tidal volumes from breath stacking dyssynchrony in ARDS: the BREATHE criteria. Intensive Care Med 42:1427–1436. https://doi.org/10.1007/s00134-016-4423-3
Goligher EC, Brochard LJ, Reid WD et al (2019) Diaphragmatic myotrauma: a mediator of prolonged ventilation and poor patient outcomes in acute respiratory failure. Lancet Respir Med 7:90–98. https://doi.org/10.1016/S2213-2600(18)30366-7
Pham T, Telias I, Piraino T, Yoshida T, Brochard LJ (2018) Asynchrony consequences and management. Crit Care Clin 34:325–341
Demoule A, Clavel M, Rolland-Debord C et al (2016) Neurally adjusted ventilatory assist as an alternative to pressure support ventilation in adults: a French multicentre randomized trial. Intensive Care Med 42:1723–1732. https://doi.org/10.1007/s00134-016-4447-8
de Wit M, Pedram S, Best AM, Epstein SK (2009) Observational study of patient-ventilator asynchrony and relationship to sedation level. J Crit Care 24:74–80. https://doi.org/10.1016/j.jcrc.2008.08.011
Devlin JW, Skrobik Y, Gelinas C et al (2018) Clinical practice guidelines for the prevention and management of pain, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med 46:e825–e873. https://doi.org/10.1097/CCM.0000000000003299
Vaschetto R, Cammarota G, Colombo D et al (2014) Effects of propofol on patient-ventilator synchrony and interaction during pressure support ventilation and neurally adjusted ventilatory assist. Crit Care Med 42:74–82. https://doi.org/10.1097/CCM.0b013e31829e53dc
Liu L, Wu AP, Yang Y et al (2017) Effects of propofol on respiratory drive and patient-ventilator synchrony during pressure support ventilation in postoperative patients: a prospective study. Chin Med J (Engl) 130:1155–1160. https://doi.org/10.4103/0366-6999.205864
Forster A, Gardaz JP, Suter PM, Gemperle M (1980) Respiratory depression by midazolam and diazepam. Anesthesiology 53:494–497. https://doi.org/10.1097/00000542-198012000-00010
Pandharipande P, Shintani A, Peterson J et al (2006) Lorazepam is an independent risk factor for transitioning to delirium in intensive care unit patients. Anesthesiology 104:21–26
Jerath A, Ferguson ND, Cuthbertson B (2020) Inhalational volatile-based sedation for COVID-19 pneumonia and ARDS. Intensive Care Med. https://doi.org/10.1007/s00134-020-06154-8
Belleville JP, Ward DS, Bloor BC, Maze M (1992) Effects of intravenous dexmedetomidine in humans. I. Sedation, ventilation, and metabolic rate. Anesthesiology 77:1125–1133. https://doi.org/10.1097/00000542-199212000-00013
Gattinoni L, Busana M, Giosa L, Macri MM, Quintel M (2019) Prone positioning in acute respiratory distress syndrome. Semin Respir Crit Care Med 40:94–100. https://doi.org/10.1055/s-0039-1685180
Guerin C, Reignier J, Richard JC et al (2013) Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 368:2159–2168. https://doi.org/10.1056/NEJMoa1214103
Elharrar X, Trigui Y, Dols AM, Touchon F, Martinez S, Prud’homme E, Papazian L (2020) Use of prone positioning in nonintubated patients with COVID-19 and hypoxemic acute respiratory failure. JAMA. https://doi.org/10.1001/jama.2020.8255
Telias I, Katira BH, Brochard L (2020) Is the prone position helpful during spontaneous breathing in patients with COVID-19? JAMA. https://doi.org/10.1001/jama.2020.8539
Goligher EC, Combes A, Brodie D et al (2019) Determinants of the effect of extracorporeal carbon dioxide removal in the SUPERNOVA trial: implications for trial design. Intensive Care Med 45:1219–1230. https://doi.org/10.1007/s00134-019-05708-9
Kolobow T, Gattinoni L, Tomlinson TA, Pierce JE (1977) Control of breathing using an extracorporeal membrane lung. Anesthesiology 46:138–141. https://doi.org/10.1097/00000542-197702000-00012
Karagiannidis C, Lubnow M, Philipp A, Riegger GA, Schmid C, Pfeifer M, Mueller T (2010) Autoregulation of ventilation with neurally adjusted ventilatory assist on extracorporeal lung support. Intensive Care Med 36:2038–2044. https://doi.org/10.1007/s00134-010-1982-6
Mauri T, Grasselli G, Suriano G et al (2016) Control of respiratory drive and effort in extracorporeal membrane oxygenation patients recovering from severe acute respiratory distress syndrome. Anesthesiology 125:159–167. https://doi.org/10.1097/ALN.0000000000001103
Crotti S, Bottino N, Ruggeri GM et al (2017) Spontaneous breathing during extracorporeal membrane oxygenation in acute respiratory failure. Anesthesiology 126:678–687. https://doi.org/10.1097/ALN.0000000000001546
Hoeper MM, Wiesner O, Hadem J et al (2013) Extracorporeal membrane oxygenation instead of invasive mechanical ventilation in patients with acute respiratory distress syndrome. Intensive Care Med 39:2056–2057. https://doi.org/10.1007/s00134-013-3052-3
Kurihara C, Walter JM, Singer BD et al (2018) Extracorporeal membrane oxygenation can successfully support patients with severe acute respiratory distress syndrome in lieu of mechanical ventilation. Crit Care Med 46:e1070–e1073. https://doi.org/10.1097/CCM.0000000000003354
Spinelli E, Mauri T, Lissoni A et al (2020) Spontaneous breathing patterns during maximum extracorporeal CO2 removal in subjects with early severe ARDS. Respir Care 65:911–919. https://doi.org/10.4187/respcare.07391
Combes A, Fanelli V, Pham T, Ranieri VM, European Society of Intensive Care Medicine Trials Group and the “Strategy of Ultra-Protective lung ventilation with Extracorporeal CO2 Removal for New-Onset moderate to severe ARDS” (SUPERNOVA) investigators (2019) Feasibility and safety of extracorporeal CO2 removal to enhance protective ventilation in acute respiratory distress syndrome: the SUPERNOVA study. Intensive Care Med 45:592–600. https://doi.org/10.1007/s00134-019-05567-4
Diehl JL, Mercat A, Pesenti A (2019) Understanding hypoxemia on ECCO2R: back to the alveolar gas equation. Intensive Care Med 45:255–256. https://doi.org/10.1007/s00134-018-5409-0
Doorduin J, Nollet JL, Roesthuis LH et al (2017) Partial neuromuscular blockade during partial ventilatory support in sedated patients with high tidal volumes. Am J Respir Crit Care Med 195:1033–1042. https://doi.org/10.1164/rccm.201605-1016OC
Froese AB, Bryan AC (1974) Effects of anesthesia and paralysis on diaphragmatic mechanics in man. Anesthesiology 41:242–255. https://doi.org/10.1097/00000542-197409000-00006
Le Pimpec-Barthes F, Legras A, Arame A, Pricopi C, Boucherie JC, Badia A, Panzini CM (2016) Diaphragm pacing: the state of the art. J Thorac Dis 8:376. https://doi.org/10.21037/jtd.2016.03.97
Masmoudi H, Coirault C, Demoule A et al (2013) Can phrenic stimulation protect the diaphragm from mechanical ventilation-induced damage? Eur Respir J 42:280–283. https://doi.org/10.1183/09031936.00045613
Reynolds SC, Meyyappan R, Thakkar V et al (2017) Mitigation of ventilator-induced diaphragm atrophy by transvenous phrenic nerve stimulation. Am J Respir Crit Care Med 195:339–348. https://doi.org/10.1164/rccm.201502-0363OC
Reynolds S, Ebner A, Meffen T et al (2017) Diaphragm activation in ventilated patients using a novel transvenous phrenic nerve pacing catheter. Crit Care Med 45:e691–e694. https://doi.org/10.1097/CCM.0000000000002366
McCaughey EJ, Jonkman AH, Boswell-Ruys CL et al (2019) Abdominal functional electrical stimulation to assist ventilator weaning in critical illness: a double-blinded, randomised, sham-controlled pilot study. Crit Care 23:261. https://doi.org/10.1186/s13054-019-2544-0
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
EG is supported by an Early Career Investigator award from the Canadian Institutes of Health Research (AR7-162822). He has received research support in the form of equipment from Getinge and Timpel and personal fees from Getinge. SJ reports receiving consulting fees from Drager, Medtronic, Baxter, Fresenius Medical and Fisher & Paykel. TM received personal fees from Fisher and Paykel, Drager, Mindray, Braun outside of the submitted work. LB’s laboratory reports grants from Medtronic Covidien, grants and non-financial support from Fisher Paykel, non-financial support from Air Liquide, non-financial support from Sentec, non-financial support from Philips, a patent with General Electric, outside the submitted work. LH received research grants from Liberate Medical and Orion Pharma, and travel and speakers fee from Getinge and Orion Pharma.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Goligher, E.C., Jonkman, A.H., Dianti, J. et al. Clinical strategies for implementing lung and diaphragm-protective ventilation: avoiding insufficient and excessive effort. Intensive Care Med 46, 2314–2326 (2020). https://doi.org/10.1007/s00134-020-06288-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-020-06288-9