Abstract
Purpose
To determine what physiological and biochemical factors predict development of bacteraemia and mortality in patients with acute liver failure (ALF).
Methods
Retrospective analysis of 206 ALF patients admitted to a specialist liver intensive therapy unit (LITU) from January 2003 to July 2005 (data collected prospectively).
Results
A total of 206 patients were defined with ALF: 72 (35%) suffered bacteraemia (BAClf) and 134 (65%) did not (NBAClf). Gram positive organisms were observed in 44% of isolates, gram negatives in 52% and fungaemia in 4%. Median time to first bacteraemia was 10 (7–16) days. On admission, BAClf patients had higher SIRS scores and degrees of hepatic encephalopathy (HE). During their LITU course, BAClf patients had significantly increased requirements for renal replacement therapy (RRT), mechanical ventilation, and longer median LITU stay. Multivariate analysis (logistical regression) demonstrated significant predictors of bacteraemia on admission were HE grade >2 (Odds Ratio 1.6) and SIRS score >1 (OR 2.7). In all patients, independent predictors of mortality (logistical) were age (OR 1.41), maximum HE grade pre-intubation (1.76), Lactate (1.14) and Acute Physiology and Chronic Health Evaluation II score (APACHEII) (1.09), but not bacteraemia. Transplantation was protective (OR 0.20).
Conclusion
In this study, severity of hepatic encephalopathy and SIRS score >1 were predictive of bacteraemia. APACHEII was independently predictive of mortality in all ALF patients but not bacteraemia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patients with acute liver failure (ALF) are abnormally susceptible to infection as a result of acquired immunological deficits [1, 2]. In previous studies, the main localisation of bacterial infection in ALF was in the respiratory tract with a median of 5 days to the diagnosis of pneumonia and 3 days to bacteraemia [3]. The rate of clinically significant positive cultures has been reported at between 10 and 80% with attributable mortality ranging from 10 to 37% [4–6].
In common with other immunocompromised patients, those with ALF may respond abnormally when infected. Previous studies have shown a third fail to develop a neutrophil response or a temperature greater than 38°C or less than 36°C [3]. Furthermore, ALF may frequently present almost indistinguishably from septic shock [7], related to the inflammatory focus of the necrotic liver.
Classically, sepsis and septic shock have been defined by the components of the systemic inflammatory response syndrome (SIRS) [8, 9]. Varied clinical and laboratory manifestations of SIRS in ALF are likely to be the result of variable response to increased cytokine production of monocytes and macrophages in response to infectious stimuli, including bacterial lipopolysaccharide [10–12]. Inflammatory responses have been associated with exacerbation of encephalopathy and increased risk of intracranial hypertension and infection may or may not be a contributor to the clinical picture seen at any time.
In this study, we reviewed 72 ALF patients admitted to a specialised liver intensive therapy unit (LITU) between January 2003 and July 2005 who developed bacteraemia, and compared their physiological and biochemical profile along with clinical outcomes with 134 patients with ALF admitted during the same time period who did not [13]. We were particularly interested in answering the following questions; (1) what physiological and biochemical factors predict development of bacteraemia in patients with ALF, and (2) what factors impact upon mortality?
Methods
Our study protocol was approved by an ethics committee (National Health Service, UK). We performed a retrospective analysis of 206 patients admitted with a diagnosis of ALF to LITU at King’s College Hospital between January 2003 and July 2005. Physiological and microbiological data had been previously collected and entered into a dedicated database. ALF was defined as the presence of hepatic encephalopathy (HE) in a patient who has developed synthetic dysfunction (jaundice, coagulopathy) in the absence of prior liver disease [14].
Standard aerobic and anaerobic (2 × 10 ml of blood) paired samples were taken on admission from new central lines inserted as well as one peripheral site. Blood cultures were also taken upon clinical suspicion of sepsis (temperature, SIRS, hypotension, abnormalities of central line site). Bacteraemia was defined as one culture yielding a recognised pathogen (e.g., Staphylococcus aureus, Enterococcus, member of Enterobacteriaciae, Pseudomonas spp., Candida spp.) from a single sample or an organism that forms part of the normal skin flora isolated from more than one culture occurring within a 48-h period. This is consistent with Nosocomial Infection National Surveillance Scheme, UK (NINSS) criterion one for the definition of bacteraemia [15]. Isolation of different species from different samples or isolates of the same species from samples taken more than 14 days apart were defined as separate episodes. Positive blood culture dates and organisms were collected from microbiology databases. Patients were excluded from this analysis if positive blood culture samples were taken prior to admission to LITU; either from the ward or a referring hospital.
Three SIRS components, white blood count, temperature and heart rate, were documented on admission. As the majority of patients were mechanically ventilated having reached grade III HE, respiratory rate and carbon dioxide levels values were not used in calculating a SIRS score [16]. The definitions of SIRS used were those of the Consensus Conference on Sepsis and multiorgan failure [8].
For both ALF patients with bacteraemia (BAClf) and without (NBAClf), lengths of stay (in hospital and LITU), requirement of renal replacement therapy (RRT) and ventilation (MV) along with other physiological, biochemical and outcome data were obtained from the Riyadh Intensive Care Physiology database (RIP), LITU patient charts and patient discharge summaries. Frequency of patients’ meeting King’s College Criteria listing criteria (acetaminophen and non-acetaminophen), maximum grade of HE prior to intubation, listing status on the super-urgent list and the number of patients transplanted was recorded [17]. Pathogen prediction in bacteraemia associated with pneumonia by tracheal surveillance cultures is associated with a higher rate of adequate empiric antibiotic therapy [18, 19]. Qualitative tracheal aspirates (TA) collected during the same time period were correlated with bloodstream isolates. Chest radiographic data were not captured in this study.
Patients with ALF (HE, coagulopathy ± acute renal failure) were started prophylactically on piperacillin-tazobactam upon admission to LITU. Amikacin and/or vancomycin were added if indicated by cutaneous swabs. If the patient was penicillin-allergic, meropenem was used as a second-line agent. Patients would receive a course of intravenous fluconazole if they fulfilled King’s College Listing criteria or had 2 of 3 criteria. Antibiotics were adjusted based on positive cultures. Normal antimicrobial courses lasted 5 days unless clinically indicated (e.g. Pseudomonas). Transplanted patients who became bacteraemic after LT were included in the BAClf group. The vast majority (97%) of these patients became bacteraemic after LT.
Statistics
Statistical analysis was performed using SPSS version 14 (Chicago, USA) and STATA 10 (College Station, Texas). Differences in categorical variables between groups were analysed using the χ 2-test. Measure of central tendency for continuous variables were compared using the Mann–Whitney U test (non-parametric variables) and the paired t test (parametric) following normality testing. Continuous non-parametric variables were reported with medians and inter-quartile ranges (IQR). Parametric variables were reported as means with standard deviations (SD). Statistical significance was required at the 95% level (P < 0.05).
The study endpoints were bacteraemia (all patients) and in-hospital mortality (total and non-transplanted). Following univariate logistic regression analysis variables found to be significant were used in multivariate modelling using “enter” and “backwards” mode (inclusion P < 0.10) for prediction of bacteraemia and mortality. Transplantation was not used with death as a composite outcome measure. Multivariate modelling is reported as odds ratios (OR) with 95% confidence intervals for significant predictors of endpoints.
Results
Etiology and microbiology information
Seventy-two ALF patients (acetaminophen = 36 (50%), drug = 4, hepatitis B = 3, budd-chiari = 3, ischemia = 2, fatty liver of pregnancy = 2, seronegative/undefined = 18) met NINSS criteria for bacteraemia (BAClf), while 134 patients [acetaminophen 69 (52%), fatty liver/HELLP 13, drug 8, hepatitis B7, budd-chiari 6, ischemia 2, seronegative/undefined = 29] did not (NBAClf), giving a total of 206 (Fig. 1
).
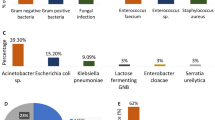
The 72 BAClf patients suffered 133 episodes of bacteraemia while in LITU. The distribution and frequency of isolates are shown in Fig. 2. Of 163 total isolates, 71 (44%) were gram positive and 84 (52%) were gram negative. Candidemia was observed in 8 isolates (4%). The three most frequent pathogens causing bacteraemia were: Enterococcus faecium (n = 21, 13%), Klebsiella spp. (21, 13%) and vancomycin-resistant Enterococcus spp. (18, 11%). The median time to first bacteraemia was 10 (7–16) days.
Analysis of all 206 ALF patients: admission data, requirement for organ support and transplantation
Univariate analysis
Results of univariate analysis of all 206 patients are presented in Table 1. Several factors showed significant differences. On the day of admission, BAClf patients had a significantly higher SIRS scores with 44 patients having a SIRS score ≥2. During LITU stay, more BAClf patients had a maximum HE grade >2, required RRT and were on it for a longer period. All 72 BAClf patients required MV for >24 h. Length of LITU stay was significantly longer in the BAClf group. Both groups had similar numbers who met King’s College criteria and who were subsequently listed for LT. There was no difference in survival to hospital discharge between the groups.
Multivariate analysis of all patients: bacteraemia and mortality
A multivariate logistical regression of all patients was performed to determine which factors predicted the development of bacteraemia. Significant predictors of bacteraemia on multivariate analysis were admission HE grade ≥3 (OR 1.6; 95% CI, 1.1–2.3; P = 0.01) and admission SIRS score ≥2 (OR 2.7; 95% CI, 1.4–5.4; P = 0.004). Two regression models were developed to predict mortality; SIRS (not using APACHEII) and APACHEII (not using SIRS). Using multivariate logistic regression analysis, SIRS was not predictive of mortality; however, illness severity as measured by APACHEII was. We subsequently performed a logistical regression for all ALF patients using APACHEII to adjust for illness severity (see Table 2). Transplantation was protective. Age, lactate, APACHEII and maximum grade of HE pre-intubation were independently predictive of mortality while bacteraemia was not.
Multivariate analysis of non-transplanted ALF patients
Two logistical regression models were developed to predict mortality on non-transplanted ALF patients (n = 147). Models were developed using SIRS and APACHEII. Using multivariate logistic regression analysis, SIRS was not predictive of mortality however illness severity as measured by APACHEII was. On multivariate logistical regression analysis (data not tabulated), APACHEII (OR 1.08; 95% CI, 1.02–1.15; P = 0.007), age (OR 1.04; 95% CI, 1.01–1.08; P = 0.01), maximum HE grade (OR 1.66; 95% CI, 1.01–2.74; P = 0.05) and lactate (OR 1.41; 95% CI, 1.18–1.69; P < 0.001) were significant.
Tracheal aspirate data: bacteraemic ALF patients and controls
Fifty-four patients (BAClf = 41, NBAClf = 13) had 81 positive qualitative TA samples (BAClf 58, NBAClf 23) during their LITU stay (111 isolates; BAClf = 78, NBAClf = 33). In the BAClf group, 33 (80%) patients grew the same bacterial species in both blood and chest samples and of those 17 (51%) had a positive TA sample prior to diagnosis of bacteraemia. Of all isolates (n = 111), 70 (63%) were gram negative species, 10 (9%) were gram positive species and 30 (27%) were fungal (Candida = 29, Aspergillus = 1). In the 17 patients who developed bacteraemia post positive TA, the median time to positive TA was 8 (6–13) days.
Discussion
In the intervening period since previous studies at King’s College Hospital by Rolando (1986–1987) [3] and Wade (1986–1996) [20], the epidemiology and timing of bacteraemia has changed. While Rolando found bacteraemia to occur at day 3 (median), the median time in this cohort was longer (day 10). In Wade’s study, 73% of bloodstream infections were gram positive with coagulase negative staphylococcus being the most common (34%). In our review, gram negatives are now more prevalent (52%) globally. Similarly, the aetiology of chest infections has also become predominantly gram negative (63%). Of note, Vaquero et al. showed that gram negative bacteria represented a higher fraction of causative organisms in 227 ALF patients who went on to develop Grade III/IV HE [16]. A possible explanation for this could be changes in empirical treatment for sepsis in LITU, particularly increased use of empirical piperacillin-tazobactam in patients who met King’s College Listing criteria. Between 1986 and 1996, the antimicrobial regimens most frequently commenced on admission to LITU were flucloxicillin and gentamicin, flucloxicillin and ceftazidime and piperacillin with gentamicin [20]. The relationship of aetiology with increased length of stay may also reflect the cumulative exposure to environmental reservoirs (e.g. Pseudomonas aeruginosa [21]). More importantly, studies have shown that critically ill patients are at increased risk of bloodstream infections with increasing APACHEII score, reflecting worse multiorgan failure [22, 23].
Previous studies have shown that critically ill patients are at an increased risk of antibiotic-resistant pathogens such as Pseudomonas, Acinetobacter and methicillin-resistant strains of S. aureus after prior antibiotic treatment, prolonged hospitalisation or MV, RRT or increasing multiorgan failure [23, 24]. Although our study was not geared to diagnose ventilator-associated pneumonia (VAP) (no radiographic information), given that most positive TA isolates were found at a median of 8 days and were largely gram negative etiologies (27% Acinetobacter, 18% Pseudomonas), it appears that a significant number of patients may have developed VAP. Risk factors for gram negative VAP in critically ill patients include >48 h on a ward prior to ICU admission, and >8 days in an ICU [25–27].
In this cohort, ALF patients with increased severity of liver disease were more predisposed to bacteraemia and required more organ support. This is supported by the fact that, on multivariate analyses, admission SIRS score ≥2 and maximum HE grade were independently associated with the development of bacteraemia. Of the 155 patients who required RRT following admission to LITU, 60% (94) were initiated within the first 48 h (median time to bacteraemia was 10 days). Leithead et al. showed similarly in 308 patients (multivariate analysis) with ALF, that while admission SIRS and paracetamol induced ALF were independent predictors of renal dysfunction (P < 0.05), infection was not [28].
Admission SIRS scores (out of three) were found to be higher in BAClf patients irrespective of whether or not they went on to be transplanted. Given the fact that patients developed bacteraemia 10 days later, this finding on admission may be a surrogate for increased hepatic necrosis contributing to severity of illness and predisposition to multiorgan failure and bacteraemia. Previous studies have shown that patients with higher SIRS scores early on after ICU admission exhibit increasingly severe acute hepatic failure later during their ICU stay, and that higher SIRS scores may reduce the probability of transplantation in patients with non-acetaminophen ALF [29]. Furthermore, an increased SIRS score has also been shown to be an independent predictor of outcome in patients with acetaminophen-induced ALF [30]. In contrast, Vaquero et al. showed that, while SIRS markers on admission in non-infected ALF patients were predictive of future progression of HE, neither SIRS nor infection were predictive of mortality [16]. Our results indicate that while SIRS score (≥2) was an independent predictor of future bacteraemia, APACHEII score (not SIRS) on admission was an independent predictor of mortality. APACHEII has been shown previously to be of similar predictive value as the King’s College Criteria in predicting mortality in patients suffering from acute liver failure [31, 32].
The lack of survival difference between the BAClf and NBAClf groups may be explained by the role of early SIRS-related mortality versus late mortality due to the compensatory anti-inflammatory response [33]. Bacteraemic patients who survived the acute systemic inflammatory response of ALF lived long enough to develop bacteraemia, possibly due to immunosuppression from monocyte exhaustion [34]. Potentially, the severity of ALF and not bacteraemia ultimately may have a greater impact on survival. This is supported by multivariate logistical regression analysis, which demonstrated that the maximum grade of HE, APACHEII score and lactate were independently predictive of mortality.
The main limitation of this study is the nature of the retrospective analysis, and hence it is difficult to attribute cause and effect between certain clinical outcomes (e.g. requirement for RRT and bacteraemia) that may emerge at differing points in time in the clinical course. We specifically focused on admission data to determine predictors of endpoints. Nevertheless, a prospective study specifically designed to ascertain the timing of need for RRT and emergence of bacteraemia would be of interest. Bacteraemia was used as a surrogate of sepsis given that sepsis and ALF are often indistinguishable [35]. We acknowledge that, while standard blood cultures may reflect current standards of care in LITU, we may have missed some bacteraemias that could have been detected by PCR methods and, furthermore, that some patients in the NBAClf group may have had sepsis without bacteraemia [36]. Although the length of accruement was short, we identified over 200 patients with ALF, which is of sufficient power to perform multivariate analysis. In terms of consistency in the provision of supportive medical therapy and anti-infective prescribing policies, a shorter recruitment time in a single centre is as potentially beneficial as many cohort studies in ALF recruited over many years where practice would be expected to change.
Collectively, these findings suggest that, once patients develop ALF, mortality is potentially more reflective of the underlying severity of the hepatic illness contributing to the development of multiorgan failure rather than the subsequent development of bacteraemia. Hence, while previous studies have shown attributable mortality to bacteraemia in ALF patients [4–6], we cannot draw the same conclusions from the present study.
Conclusion
In this study, severity of hepatic encephalopathy prior to intubation and admission SIRS score were independently predictive of bacteraemia in ALF. APACHEII was independently predictive of mortality but not bacteraemia in all ALF patients.
Abbreviations
- ALF:
-
Acute liver failure
- BAClf:
-
Bacteraemic acute liver failure patients
- 95% CI:
-
95% confidence interval
- HE:
-
Hepatic encephalopathy
- HR:
-
Heart rate
- ICU:
-
Intensive care unit
- LITU:
-
Liver Intensive therapy (care) unit
- LT:
-
Liver transplantation
- MAP:
-
Mean arterial pressure
- MV:
-
Mechanical ventilation
- NBAClf:
-
Non-bacteraemic acute liver failure
- NINSS:
-
Nosocomial infection national surveillance scheme
- OR:
-
Odds ratio
- RRT:
-
Renal replacement therapy
- SIRS:
-
Systemic inflammatory response syndrome
- TA:
-
Tracheal aspirates
- VAP:
-
Ventilator-associated pneumonia
References
Wyke RJ, Yousif-Kadaru AG, Rajkovic IA, Eddleston AL, Williams R (1982) Serum stimulatory activity and polymorphonuclear leucocyte movement in patients with fulminant hepatic failure. Clin Exp Immunol 50:442–449
Clapperton M, Rolando N, Sandoval L, Davies E, Williams R (1997) Neutrophil superoxide and hydrogen peroxide production in patients with acute liver failure. Eur J Clin Invest 27:164–168
Rolando N, Harvey F, Brahm J, Philpott-Howard J, Alexander G, Gimson A, Casewell M, Fagan E, Williams R (1990) Prospective study of bacterial infection in acute liver failure: an analysis of fifty patients. Hepatology 11:49–53
Rolando N, Wade JJ, Fagan E, Philpott-Howard J, Casewell MW, Williams R (1992) An open, comparative trial of aztreonam with vancomycin and gentamicin with piperacillin in patients with fulminant hepatic failure. J Antimicrob Chemother 30:215–220
Rolando N, Gimson A, Wade J, Philpott-Howard J, Casewell M, Williams R (1993) Prospective controlled trial of selective parenteral and enteral antimicrobial regimen in fulminant liver failure. Hepatology 17:196–201
Rolando N, Wade JJ, Stangou A, Gimson AE, Wendon J, Philpott-Howard J, Casewell MW, Williams R (1996) Prospective study comparing the efficacy of prophylactic parenteral antimicrobials, with or without enteral decontamination, in patients with acute liver failure. Liver Transpl Surg 2:8–13
Dirix LY, Polson RJ, Richardson A, Williams R (1989) Primary sepsis presenting as fulminant hepatic failure. Q J Med 73:1037–1043
American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference (1992) Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med 20:864–874
Pittet D, Rangel-Frausto S, Li N, Tarara D, Costigan M, Rempe L, Jebson P, Wenzel RP (1995) Systemic inflammatory response syndrome, sepsis, severe sepsis and septic shock: incidence, morbidities and outcomes in surgical ICU patients. Intensive Care Med 21:302–309
Billiau A, Vandekerckhove F (1991) Cytokines and their interactions with other inflammatory mediators in the pathogenesis of sepsis and septic shock. Eur J Clin Invest 21:559–573
Glauser MP, Zanetti G, Baumgartner JD, Cohen J (1991) Septic shock: pathogenesis. Lancet 338:732–736
Bone RC (1991) The pathogenesis of sepsis. Ann Intern Med 115:457–469
Karvellas CJ, Pink F, McPhail M, Cross T, Auzinger G, Bernal W, Sizer E, Kutsogiannis D, Eltringham I, Wendon J (2007) The incidence of bacteremia and outcomes in patients with acute liver failure. Abstract 847 in: Proceedings of the 58th Annual Meeting of the American Association for the Study of Liver Diseases. Hepatology 46(S1):612A
O’Grady JG, Schalm SW, Williams R (1993) Acute liver failure: redefining the syndromes. Lancet 342:273–275
Document N (1997) Nosocomial Infection National Surveillance Scheme (UK): protocol for the surveillance of hospital acquired bacteremia. PHLS Central Public Health Laboratory, London
Vaquero J, Polson J, Chung C, Helenowski I, Schiodt FV, Reisch J, Lee WM, Blei AT (2003) Infection and the progression of hepatic encephalopathy in acute liver failure. Gastroenterology 125:755–764
O’Grady JG, Alexander GJ, Hayllar KM, Williams R (1989) Early indicators of prognosis in fulminant hepatic failure. Gastroenterology 97:439–445
Depuydt P, Benoit D, Vogelaers D, Claeys G, Verschraegen G, Vandewoude K, Decruyenaere J, Blot S (2006) Outcome in bacteremia associated with nosocomial pneumonia and the impact of pathogen prediction by tracheal surveillance cultures. Intensive Care Med 32:1773–1781
Depuydt P, Benoit D, Vogelaers D, Decruyenaere J, Vandijck D, Claeys G, Verschraegen G, Blot S (2008) Systematic surveillance cultures as a toolto predict involvement of multidrug antibiotic resistant bacteria in ventilator-associated pneumonia. Intensive Care Med 34:675–682
Wade J, Rolando N, Philpott-Howard J, Wendon J (2003) Timing and aetiology of bacterial infections in a liver intensive care unit. J Hosp Infect 53:144–146
Agodi A, Barchitta M, Cipresso R, Giaquinta L, Romeo M, Denaro C (2007) Pseudomonas aeruginosa carriage, colonization, and infection in ICU patinets. Intensive Care Med; epub (TBA)
Laupland KB, Kirkpatrick AW, Church DL, Ross T, Gregson DB (2004) Intensive-care-unit-acquired bloodstream infections in a regional critically ill population. J Hosp Infect 58:137–145
Park DR (2005) The microbiology of ventilator-associated pneumonia. Respir Care 50:742–763; discussion 763–745
Garnacho-Montero J, Ortiz-Leyba C, Fernandez-Hinojosa E, Aldabo-Pallas T, Cayuela A, Marquez-Vacaro JA, Garcia-Curiel A, Jimenez-Jimenez FJ (2005) Acinetobacter baumannii ventilator-associated pneumonia: epidemiological and clinical findings. Intensive Care Med 31:649–655
Keenan SP, Heyland DK, Jacka MJ, Cook D, Dodek P (2002) Ventilator-associated pneumonia. Prevention, diagnosis, and therapy. Crit Care Clin 18:107–125
Heyland DK, Cook DJ, Griffith L, Keenan SP, Brun-Buisson C (1999) The attributable morbidity and mortality of ventilator-associated pneumonia in the critically ill patient. The Canadian Critical Trials Group. Am J Respir Crit Care Med 159:1249–1256
American Thoracic Society/Infectious Diseases Society of America (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388–416
Leithead JA, Ferguson JW, Bates CM, Davidson JS, Lee A, Bathgate AJ, Hayes PC, Simpson KJ (2008) The systemic inflammatory response syndrome is predictive of renal dysfunction in patients with non-paracetamol-induced acute liver failure. Gut; epub ahead of print
Miyake Y, Iwasaki Y, Makino Y, Kobashi H, Takaguchi K, Ando M, Sakaguchi K, Shiratori Y (2007) Prognostic factors for fatal outcomes prior to receiving liver transplantation in patients with non-acetaminophen-related fulminant liver failure. J Gastroenterol Hepatol 22:855–861
Schmidt LE, Larsen FS (2006) Prognostic implications of hyperlactatemia, multiple organ failure, and systemic inflammatory response syndrome in patients with acetaminophen-induced acute liver failure. Crit Care Med 34:337–343
Mitchell I, Bihari D, Chang R, Wendon J, Williams R (1998) Earlier identification of patients at risk from acetaminophen-induced acute liver failure. Crit Care Med 26:279–284
Bernal W, Wendon J, Rela M, Heaton N, Williams R (1998) Use and outcome of liver transplantation in acetaminophen-induced acute liver failure. Hepatology 27:1050–1055
Antoniades CG, Berry PA, Wendon JA, Vergani D (2008) The importance of immune dysfunction in determining outcome in acute liver failure. J Hepatol 49:845–861
Docke WD, Randow F, Syrbe U, Krausch D, Asadullah K, Reinke P, Volk HD, Kox W (1997) Monocyte deactivation in septic patients: restoration by IFN-gamma treatment. Nat Med 3:678–681
Trewby PN, Williams R (1977) Pathophysiology of hypotension in patients with fulminant hepatic failure. Gut 18:1021–1026
Peters RP, van Agtmael MA, Danner SA, Savelkoul PH, Vandenbroucke-Grauls CM (2004) New developments in the diagnosis of bloodstream infections. Lancet Infect Dis 4:751–760
Acknowledgments
Canadian Institute of Health Research and Canadian Association of Gastroenterology for Dr. Karvellas’ salary support.
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karvellas, C.J., Pink, F., McPhail, M. et al. Predictors of bacteraemia and mortality in patients with acute liver failure. Intensive Care Med 35, 1390–1396 (2009). https://doi.org/10.1007/s00134-009-1472-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-009-1472-x