Abstract
Aims/hypothesis
Tirzepatide is a novel dual glucose-dependent insulinotropic peptide (GIP) and glucagon-like peptide-1 receptor agonist (GLP-1 RA) currently under review for marketing approval. Individual trials have assessed the clinical profile of tirzepatide vs different comparators. We conducted a systematic review and meta-analysis to assess the efficacy and safety of tirzepatide for type 2 diabetes.
Methods
We searched PubMed, Embase, Cochrane and ClinicalTrials.gov up until 27 October 2021 for randomised controlled trials with a duration of at least 12 weeks that compared once-weekly tirzepatide 5, 10 or 15 mg with placebo or other glucose-lowering drugs in adults with type 2 diabetes irrespective of their background glucose-lowering treatment. The primary outcome was change in HbA1c from baseline. Secondary efficacy outcomes included change in body weight, proportion of individuals reaching the HbA1c target of <53 mmol/mol (<7.0%), ≤48 mmol/mol (≤6.5%) or <39 mmol/mol (<5.7%), and proportion of individuals with body weight loss of at least 5%, 10% or 15%. Safety outcomes included hypoglycaemia, gastrointestinal adverse events, treatment discontinuation due to adverse events, serious adverse events, and mortality. We used version 2 of the Cochrane risk-of-bias tool for randomised trials to assess risk of bias for the primary outcome.
Results
Seven trials (6609 participants) were included. A dose-dependent superiority in lowering HbA1c was evident with all three tirzepatide doses vs all comparators, with mean differences ranging from −17.71 mmol/mol (−1.62%) to −22.35 mmol/mol (−2.06%) vs placebo, −3.22 mmol/mol (−0.29%) to −10.06 mmol/mol (−0.92%) vs GLP-1 RAs, and −7.66 mmol/mol (−0.70%) to −12.02 mmol/mol (−1.09%) vs basal insulin regimens. Tirzepatide was more efficacious in reducing body weight; reductions vs GLP-1 RAs ranged from 1.68 kg with tirzepatide 5 mg to 7.16 kg with tirzepatide 15 mg. Incidence of hypoglycaemia with tirzepatide was similar vs placebo and lower vs basal insulin. Nausea was more frequent with tirzepatide vs placebo, especially with tirzepatide 15 mg (OR 5.60 [95% CI 3.12, 10.06]), associated with higher incidence of vomiting (OR 5.50 [95% CI 2.40, 12.59]) and diarrhoea (OR 3.31 [95% CI 1.40, 7.85]). Odds of gastrointestinal events were similar between tirzepatide and GLP-1 RAs, except for diarrhoea with tirzepatide 10 mg (OR 1.51 [95% CI 1.07, 2.15]). Tirzepatide 15 mg led to higher discontinuation rate of study medication due to adverse events regardless of comparator, while all tirzepatide doses were safe in terms of serious adverse events and mortality.
Conclusions/interpretation
A dose-dependent superiority on glycaemic efficacy and body weight reduction was evident with tirzepatide vs placebo, GLP-1 RAs and basal insulin. Tirzepatide did not increase the odds of hypoglycaemia but was associated with increased incidence of gastrointestinal adverse events. Study limitations include presence of statistical heterogeneity in the meta-analyses for change in HbA1c and body weight, assessment of risk of bias solely for the primary outcome, and generalisation of findings mainly to individuals who are overweight or obese and already on metformin-based background therapy.
PROSPERO registration no. CRD42021283449.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.

Introduction
Glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP) are among the main incretin hormones [1]. GLP-1 is released from L cells in the distal ileum and colon, while GIP is secreted from K cells in the duodenum and jejunum and is responsible for most of the insulinotropic incretin effect [2]. In people with type 2 diabetes the incretin effect is considerably diminished [3]. This has led to the development of GLP-1 receptor agonists (GLP-1 RAs), which have demonstrated favourable effects not only on metabolic variables but also on cardiovascular endpoints [4,5,6]. The combined GLP-1 and GIP receptor activation has been examined recently as a promising therapeutic concept, given that the two incretins can act on pancreatic beta cells both synergistically and complementarily through distinct metabolic effects [7]. Moreover, GIP can exert therapeutic benefits beyond its primary incretin role, by improving insulin sensitivity and lipid homeostasis in adipose tissue [8].
Tirzepatide (LY3298176) is a dual GIP and GLP-1 RA recently developed for the treatment of type 2 diabetes [9]. It has greater affinity to GIP receptors, rather than GLP-1 receptors, while its t½ of approximately 5 days allows once-weekly subcutaneous administration [9]. Early proof-of-concept and phase 2 studies suggested that tirzepatide can improve both markers of beta cell function and insulin sensitivity compared with selective GLP-1 RA therapy [9, 10]. On the basis of these findings, the overall efficacy and safety of tirzepatide has been investigated in the SURPASS clinical trial programme in comparison with placebo and other glucose-lowering medications including GLP-1 RAs and basal insulin [11]. In a recent news release, the manufacturer announced the submission of a new drug application to the US Food and Drug Administration (FDA) and a marketing authorisation application to the European Medicines Agency (EMA) for tirzepatide for the treatment of adults with type 2 diabetes [12]. We aimed to systematically retrieve all currently available RCTs of tirzepatide in individuals with type 2 diabetes and synthesise the evidence by means of clinically relevant meta-analyses for outcomes of efficacy and safety.
Methods
The protocol of this systematic review and meta-analysis has been registered in PROSPERO (registration no. CRD42021283449). We report our methods and results in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement [13].
Eligibility criteria
We included RCTs with a duration of intervention of at least 12 weeks that compared tirzepatide at a maintenance dose of 5, 10 or 15 mg once-weekly, administered subcutaneously, with placebo or any other glucose-lowering medication. Eligible participants were adults with type 2 diabetes irrespective of background glucose-lowering treatment.
Information sources
We searched PubMed, Embase and Cochrane databases on 27 October 2021 for English-language studies. The search strategy included the keywords ‘tirzepatide’ and ‘ly3298176’ as free-text and MeSH (Medical Subject Headings) terms. Search records that were identified as abstract publications in journals’ supplementary issues of the EASD or the ADA scientific meetings were also considered eligible. We also manually searched the websites of the EASD and ADA scientific meetings, and ClinicalTrials.gov to retrieve either additional eligible trials or any additional information for trials already identified through the database searches.
Selection process
Results from the databases’ search were imported in a reference management software and, after deduplication, were juxtaposed with the results from the additional search sources. Records were initially screened at title and abstract level, and potentially eligible records were examined in full text with reasons for exclusion being recorded. Two independent reviewers performed the study selection process, and any disagreements were resolved by a third reviewer.
Data collection process
For each included trial, we used predesigned forms to extract study characteristics, participants’ demographics and baseline characteristics, and outcome data. Our primary outcome was change in HbA1c from baseline. Secondary efficacy outcomes included change in body weight from baseline, proportion of individuals reaching the HbA1c target of <53 mmol/mol (<7.0%), ≤48 mmol/mol (≤6.5%) or <39 mmol/mol (<5.7%) and proportion of patients with at least 5%, 10% or 15% body weight loss. Safety and tolerability outcomes comprised discontinuation of treatment due to adverse events, incidence of serious adverse events, all-cause mortality, hypoglycaemia (plasma glucose ≤3.9 mmol/l), severe hypoglycaemia (a hypoglycaemic event requiring assistance), nausea, vomiting and diarrhoea. The unit of measurement for all dichotomous outcomes was the number of individuals experiencing at least one event of interest. For all outcomes, we extracted data for the modified intention-to-treat population, defined as all randomly assigned participants who received a least one dose of the study drug. For efficacy outcomes, in case trials reported results for different estimand analyses, we preferably extracted data for the efficacy estimand, which represents on-treatment efficacy without the influence of rescue therapy [14]. Data extraction was done by two independent reviewers and arbitrated by a third reviewer.
Risk-of-bias assessment
We used the version 2 of the Cochrane risk-of-bias tool for randomised trials to assess risk of bias for the primary outcome (change in HbA1c) [15]. Overall risk of bias for each trial was considered low if all domains were at low risk of bias, and high if at least one domain was at high risk of bias. In any other case, the risk of bias was deemed as being of some concern. Risk-of-bias assessment was done independently by two reviewers and any disagreements were resolved through consensus. We did not evaluate small-study effect bias with a funnel plot due to the small number of included trials [16].
Data synthesis
We conducted meta-analyses when at least two studies reported relevant outcome data. For continuous outcomes, we calculated mean differences and 95% CIs using an inverse variance random-effects model. For dichotomous outcomes, we calculated ORs and 95% CIs using the random-effects Mantel–Haenszel approach. In all analyses, we used the Paule–Mandel method to estimate between-study variance [17], and the I2 statistic to assess statistical heterogeneity. We performed separate analyses based on type of comparator (placebo, GLP-1 RA or basal insulin) and subgroup analyses based on tirzepatide maintenance dose (5, 10 or 15 mg once-weekly). In the placebo-controlled analyses for change in HbA1c and change in body weight, we conducted a post hoc sensitivity analysis excluding one trial with a short duration (12 weeks) [18] and one trial that recruited participants on background insulin therapy [19]. All analyses were done using R version 4.0.5 (R Core Team, Vienna, Austria) and the statistical package ‘meta’.
Results
Search results
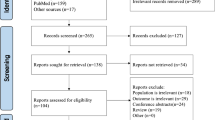
The initial search identified 210 results. After screening these records, eight reports of seven RCTs [18,19,20,21,22,23,24,25] with a total of 6609 participants were included in the systematic review and meta-analysis (Fig. 1).
Study characteristics
The main characteristics of the included studies are presented in Table 1. One study was published in 2022, four in 2021, one in 2020 and one in 2018. Of note, the 2022 study [19] was initially identified in our literature search as a 2021 conference abstract and was subsequently published in a journal during preparation of a revision of our manuscript. Six studies assessed all three eligible tirzepatide maintenance doses (5, 10 and 15 mg once-weekly), while one study included two arms with a maintenance dose of 15 mg but with two different dose-escalation regimens [18]. In our analyses, we merged data for these two arms into a single treatment arm. For one study (SURPASS-2), we used data both from the journal publication [25] and from a conference abstract [24]. The comparator arm was placebo, a GLP-1 RA (subcutaneous administration of semaglutide 1 mg once-weekly) and basal insulin in three [18, 19, 22], one [24, 25] and two trials [20, 21], respectively, while one trial included both a placebo arm and a GLP-1 RA arm (dulaglutide 1.5 mg once-weekly) [23]. All studies had a parallel-group design, and three were open-label. Duration of intervention was 12, 26, 40 and 52 weeks in one, one, three and two studies, respectively. Overall risk of bias for the primary outcome was low in all studies.
Participant characteristics
Across all trials, participants’ mean HbA1c at baseline was 66.47 mmol/mol (8.2%), mean body weight was 91.5 kg and mean age was 58 years (Table 1). In five studies, background glucose-lowering treatment comprised metformin either as monotherapy or in combination with another oral medication. In one study, more than half the participants (54%) had no previous use of oral glucose-lowering medication [22], while in another study, all participants were on a stable dose of insulin glargine with (83%) or without (17%) metformin [19]. Information regarding the percentage of participants requiring rescue glucose-lowering therapy was retrieved for five studies [20,21,22,23, 25]. Based on these data, across all studies, few participants received rescue therapy (ranging approximately between 0.3% and 3.5% of participants), except for one study in which 25% of participants in the placebo arm required rescue therapy [22]. Study medications, GLP-1 RAs, dipeptidyl peptidase 4 inhibitors and pramlintide were not allowed as rescue medications.
Glycaemic efficacy
Compared with placebo, reductions in HbA1c levels ranged between 17.71 mmol/mol (1.62%) with tirzepatide 5 mg and 22.35 mmol/mol (2.06%) with tirzepatide 15 mg (Fig. 2a). Results were consistent and statistical heterogeneity was reduced in a sensitivity analysis excluding one trial with a short duration and one trial which recruited participants on background insulin therapy. All tirzepatide doses were superior to placebo in terms of achieving the HbA1c target of <53 mmol/mol (<7.0%), ≤48 mmol/mol (≤6.5%) or <39 mmol/mol (<5.7%) (electronic supplementary material [ESM] Table 1). Compared with GLP-1 RAs, tirzepatide 5, 10 and 15 mg reduced HbA1c levels by 3.22 mmol/mol (0.29%), 7.11 mmol/mol (0.65%) and 10.06 mmol/mol (0.92%), respectively (Fig. 2b). Similarly, more participants receiving any tirzepatide dose achieved the three HbA1c targets, except for the target of <53 mmol/mol (<7.0%) with tirzepatide 10 mg, as compared with participants who received a GLP-1 RA (ESM Table 1). All three tirzepatide doses were more effective than basal insulin both in reducing HbA1c (mean differences ranging between 7.66 mmol/mol [0.70%] with tirzepatide 5 mg and 12.02 mmol/mol [1.09%] with tirzepatide 15 mg [ESM Fig. 1]) and in achieving the three HbA1c targets (ESM Table 1). Of note, the mean basal insulin dose at the study endpoint (week 52) was 48.8 U with insulin degludec in the SURPASS-3 trial and 43.5 U with insulin glargine in the SURPASS-4 trial [20, 21].
Body weight
Dose-dependent reductions in body weight were evident vs placebo with tirzepatide 5 mg (6.31 kg [95% CI 4.38, 8.25], I2 70%), 10 mg (8.43 kg [95% CI 6.77, 10.09], I2 68%) and 15 mg (9.36 kg [95% CI 6.20, 12.53], I2 91%) (Fig. 3a). The sensitivity analysis excluding one trial with a short duration and one trial that recruited participants on background insulin therapy yielded similar results and reduced statistical heterogeneity. Consistently, compared with placebo, more participants receiving any of the three tirzepatide doses had reductions in body weight of at least 5%, 10% or 15% (ESM Table 2). Tirzepatide induced larger reductions in body weight vs GLP-1 RAs, ranging from 1.68 kg (95% CI 0.84, 2.52 [I2 0%]) with tirzepatide 5 mg to 7.16 kg (95% CI 4.86, 9.46 [I2 72%]) with tirzepatide 15 mg (Fig. 3b). The OR for achieving a weight loss of at least 5% with tirzepatide 5 mg, 10 mg and 15 mg (vs GLP-1 RAs) was, respectively, 1.96 (95% CI 1.01, 3.80 [I2 61%]), 4.79 (95% CI 1.95, 11.73 [I2 74%]) and 4.57 (95% CI 3.38, 6.18 [I2 0%]) (ESM Table 2). All tirzepatide doses were more efficacious than GLP-1 RAs in achieving a body weight loss of at least 10% and 15% (ESM Table 2). The superiority of tirzepatide in terms of weight control was more pronounced in the comparisons vs basal insulin (ESM Fig. 2 and ESM Table 2).
Hypoglycaemia
Incidence of any hypoglycaemia (defined as plasma glucose ≤3.9 mmol/l) with tirzepatide did not differ vs placebo (ESM Fig. 3) and was lower with tirzepatide compared with basal insulin (OR ranging from 0.17 with tirzepatide 5 mg to 0.25 with tirzepatide 15 mg) (ESM Fig. 4). We did not pool data for the two studies vs GLP-1 RAs due to the different definition of hypoglycaemia used in each trial. We did not conduct meta-analyses for severe hypoglycaemia because events were rare. Across all trials, a total of 22 participants experienced severe hypoglycaemia, defined as an event requiring assistance: ten with tirzepatide (n = 4414); 1 with semaglutide (n = 469); and 11 with insulin glargine (n = 1000).
Gastrointestinal adverse events
Compared with placebo, nausea was more frequent with all tirzepatide doses, especially 15 mg (OR 5.60 [95% CI 3.12, 10.06], I2 0%) (Table 2). Tirzepatide 15 mg was also associated with higher incidence of vomiting (OR 5.50 [95% CI 2.40, 12.59], I2 0%) and diarrhoea (OR 3.31 [95% CI 1.40, 7.85], I2 52%), while more participants receiving tirzepatide 10 mg experienced vomiting (OR 2.98 [95% CI 1.13, 7.80], I2 0%) (Table 2). Odds of gastrointestinal events were similar between tirzepatide and GLP-1 RAs, except for diarrhoea with tirzepatide 10 mg (OR 1.51 [95% CI 1.07, 2.15], I2 0%) (Table 2). Compared with basal insulin, all three tirzepatide doses were associated with dose-dependent increased odds of nausea, vomiting and diarrhoea (Table 2).
Treatment discontinuation due to adverse events
Discontinuation of treatment due to adverse events did not differ between tirzepatide 5 mg and placebo (OR 1.99 [95% CI 0.83, 4.77], I2 0%). However, more participants discontinued treatment with tirzepatide 10 mg (OR 2.39 [95% CI 1.02, 5.59], I2 0%) and 15 mg (OR 3.64 [95% CI 1.51, 8.78], I2 13%) when compared with placebo (ESM Table 3). Compared with GLP-1 RAs, more participants receiving tirzepatide 15 mg discontinued treatment due to adverse events (OR 2.29 [95% CI 1.39, 3.75], I2 0%), whereas no difference was evident for tirzepatide 5 mg and 10 mg (ESM Table 3). Compared with basal insulin, both tirzepatide 5 mg and tirzepatide 15 mg were associated with increased odds of discontinuation of study medication due to adverse events (ESM Table 3).
Serious adverse events and mortality
Incidence of serious adverse events did not differ between any of the tirzepatide doses and any comparator (ESM Table 3). Across all trials, 41 deaths occurred in individuals receiving tirzepatide (n = 4573) and 39 in the comparator arms (n = 2151). Of note, 19 of the total deaths were related to Covid-19 disease. We did not pool mortality data in a meta-analysis because most deaths occurred in a single trial which recruited exclusively patients at increased cardiovascular risk (25 deaths with tirzepatide and 35 deaths with insulin glargine) [21].
Discussion
In this systematic review and meta-analysis we summarised and synthesised the most up-to-date data from RCTs of once-weekly tirzepatide in individuals with type 2 diabetes. Based on our findings, tirzepatide induced dose-dependent reductions in HbA1c that were clinically important, not only vs placebo but also when compared with once-weekly GLP-1 RAs and basal insulin regimens. Notably, this favourable glycaemic effect was not associated with increased risk for hypoglycaemia. With respect to lowering of body weight, a significant dose-dependent effect was evident with tirzepatide even when compared with the GLP-1 RAs semaglutide and dulaglutide. The incidence of gastrointestinal adverse events was similar when comparing tirzepatide with GLP-1 RAs. However, in comparison with placebo or basal insulin, tirzepatide increased odds of nausea, while the doses of 10 and 15 mg were also more likely to cause vomiting or diarrhoea. In addition, treatment with tirzepatide increased odds of discontinuation of study drug due to adverse events. In particular, tirzepatide 15 mg was associated with at least twofold higher odds of study drug discontinuation regardless of comparator. It could be speculated that this increased discontinuation rate vs all comparators, including GLP-1 RAs, may be possibly attributed to the severity of gastrointestinal adverse events experienced with tirzepatide 15 mg, considering that the incidence of gastrointestinal adverse events was similar between tirzepatide and GLP-1 RAs. Finally, tirzepatide was not associated with higher incidence of serious adverse events or all-cause mortality.
Our literature search identified one prior systematic review and meta-analysis with tirzepatide that included four RCTs (2783 participants) [26]. Important differences and methodological considerations render the findings of that meta-analysis non-comparable with our results. More specifically, Bhagavathula and colleagues pooled efficacy outcome data in the same analysis irrespective of type of comparator (placebo or GLP-1 RA) [26]. This introduces clinical heterogeneity and considerably attenuates the practical interpretation of pooled estimates, given the well-established beneficial effects of GLP-1 RAs in reducing both HbA1c and body weight, as opposed to the neutral effect of a placebo intervention [4,5,6]. Instead, we opted to produce meta-analysis estimates that are clinically relevant and meaningful by conducting separate analyses based on type of comparator (placebo, GLP-1 RAs and basal insulin) for each outcome. Moreover, we included three additional RCTs (two vs basal insulin [20, 21] and one vs placebo [19]), totalling a considerably larger number of participants (n = 6609). Additionally, we performed meta-analyses and produced comparative estimates for safety and tolerability outcomes, which are equally important to efficacy measures when deciding on optimal diabetes therapy in clinical practice.
Certain limitations should be considered when interpreting our findings. A degree of statistical heterogeneity, as measured by the I2 statistic, was present in the analyses for change in HbA1c and body weight. However, heterogeneity was considerably reduced vs placebo in a sensitivity analysis excluding one trial with short duration and one trial in which all participants were on background insulin therapy. Heterogeneity in the analyses vs active comparators could be attributed to differences in efficacy between the two GLP-1 RAs comparators (dulaglutide and semaglutide) or to differences in background glucose-lowering therapy between the two trials with basal insulin. Moreover, we assessed overall risk of bias for each trial solely for the primary outcome of change in HbA1c. As such, we did not consider open-label status as a source of bias, given that measurement of HbA1c is an objective outcome and thus is not affected by blinding status [15]. Had we assessed risk of bias for less objective outcomes, such as participant-reported gastrointestinal adverse events, overall risk of bias for such outcomes in these trials might have been deemed of some concern. Furthermore, our results can be generalised mostly to individuals with type 2 diabetes who are already on metformin-based background therapy, given that drug-naive individuals were recruited only in one study [22]. In addition, overall mean body weight of all participants was 91.5 kg and, as such, it is uncertain whether our findings are applicable to individuals with type 2 diabetes who are not overweight or obese. Notably, the effect of tirzepatide as an anti-obesity medication is being investigated in the ongoing SURMOUNT clinical trial programme, in a similar manner to the assessment of semaglutide 2.4 mg for obesity in the Semaglutide Treatment Effect in People with obesity (STEP) programme [27].
In October 2021, the drug manufacturer submitted a marketing authorisation application to the EMA and a priority review voucher to the US FDA for the regulatory approval of tirzepatide in type 2 diabetes, leading to an expected review time of 8 months from the date of submission [12]. As such, tirzepatide is anticipated to receive marketing approval by mid-to-late 2022. Our meta-analysis findings can help clinicians and other diabetes stakeholders to determine the optimal place of tirzepatide among existing medications for type 2 diabetes. We found that tirzepatide is superior in reducing HbA1c compared with other injectable therapies, in particular basal insulin and once-weekly GLP-1 RAs. In addition, tirzepatide, even at the lowest maintenance dose of 5 mg, can reduce body weight to a greater extent compared with GLP-1 RAs including subcutaneous semaglutide which, in turn, has been shown to be superior to other glucose-lowering agents [28]. Notably, head-to-head data for tirzepatide vs GLP1 RAs are available only for dulaglutide 1.5 mg and semaglutide 1 mg. Higher doses of dulaglutide (3.0 mg and 4.5 mg) [29] have also received marketing approval for treatment of type 2 diabetes, while application for a label extension of semaglutide at the dose of 2.0 mg [30] has been submitted to the US FDA and has recently received a positive recommendation by the EMA. At present, even though the comparative efficacy of tirzepatide vs these higher dosing regimens of dulaglutide and semaglutide is unknown, available data suggest that tirzepatide could be a reasonable treatment option when glycaemic control and body weight loss are therapeutic priorities. However, clinicians should also be aware that some individuals receiving tirzepatide may experience gastrointestinal adverse events, which could possibly lead to discontinuation of treatment.
Policy decisions on the reimbursement of tirzepatide in individual countries should be based on health technology assessments integrating long-term efficacy and safety clinical data with country-specific cost-utility analyses comparing tirzepatide with other glucose-lowering medications used in clinical practice. In this regard, it is still unknown whether tirzepatide can induce long-term cardiovascular benefits that are comparable to those of specific GLP-1 RAs or sodium–glucose cotransporter 2 inhibitors [6]. Of note, in the SURPASS-4 trial, adjudicated major adverse cardiovascular events (MACE) were not increased with tirzepatide compared with insulin glargine over an extended follow-up period of 52 additional weeks after the main trial period of 52 weeks [21]. However, the design of SURPASS-4 was based on the change in HbA1c at 52 weeks, and not on MACE, as the primary outcome [21]. The ongoing SURPASS-CVOT trial (ClinicalTrials.gov registration no. NCT04255433) is expected to provide definitive answers on the impact of tirzepatide on cardiovascular disease compared with dulaglutide, a long-acting GLP-1 RA that has been shown to be cardioprotective in individuals with type 2 diabetes at increased cardiovascular risk [31]. Finally, ongoing or recently completed, yet unpublished, RCTs are expected to provide additional information on the comparative effects of tirzepatide vs other glucose-lowering agents, including dulaglutide (ClinicalTrials.gov registration no. NCT03861052), insulin glargine (ClinicalTrials.gov registration no. NCT04093752) and insulin lispro (ClinicalTrials.gov registration no. NCT04537923).
Conclusions
The findings of this meta-analysis of seven RCTs (6609 participants) suggest a dose-dependent superiority of all three tirzepatide maintenance doses on glycaemic control, not only vs placebo but also vs long-acting GLP-1 RAs and basal insulin regimens. All tirzepatide doses were superior to all comparators in terms of reducing body weight. Treatment with tirzepatide did not increase the odds of hypoglycaemia but was associated with increased incidence of gastrointestinal adverse events, mainly nausea. The dose of 15 mg also increased the odds of discontinuation due to adverse events by at least twofold regardless of comparator. These findings are mostly applicable to individuals on metformin-based background therapy, while further trial data are required to determine whether the salutary metabolic effects of tirzepatide translate to long-term cardiovascular benefits.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- EMA:
-
European Medicines Agency
- FDA:
-
Food and Drug Administration
- GIP:
-
Glucose-dependent insulinotropic peptide
- GLP-1 RA:
-
Glucagon-like peptide-1 receptor agonist
- MACE:
-
Major adverse cardiovascular events
References
Nauck MA, Meier JJ (2018) Incretin hormones: their role in health and disease. Diabetes Obes Metab 20:5–21. https://doi.org/10.1111/DOM.13129
Baggio LL, Drucker DJ (2007) Biology of incretins: GLP-1 and GIP. Gastroenterology 132(6):2131–2157. https://doi.org/10.1053/J.GASTRO.2007.03.054
Nauck M, Stöckmann F, Ebert R, Creutzfeldt W (1986) Reduced incretin effect in type 2 (non-insulin-dependent) diabetes. Diabetologia 29(1):46–52. https://doi.org/10.1007/BF02427280
Tsapas A, Avgerinos I, Karagiannis T et al (2020) Comparative effectiveness of glucose-lowering drugs for type 2 diabetes: a systematic review and network meta-analysis. Ann Intern Med 173(4):278–286. https://doi.org/10.7326/M20-0864
Alexander JT, Staab EM, Wan W et al (2021) The longer-term benefits and harms of glucagon-like peptide-1 receptor agonists: a systematic review and meta-analysis. J Gen Intern Med 37(2):415–438. https://doi.org/10.1007/S11606-021-07105-9
American Diabetes Association (2021) 9. Pharmacologic approaches to glycemic treatment: standards of medical Care in diabetes—2021. Diabetes Care 44(Supplement 1):S111–S124. https://doi.org/10.2337/DC21-S009
Bastin M, Andreelli F (2019) Dual GIP-GLP1-receptor agonists in the treatment of type 2 diabetes: a short review on emerging data and therapeutic potential. Diabetes Metab Syndr Obes 12:1973–1985. https://doi.org/10.2147/DMSO.S191438
Asmar M, Simonsen L, Asmar A, Holst JJ, Dela F, Bülow J (2016) Insulin plays a permissive role for the vasoactive effect of GIP regulating adipose tissue metabolism in humans. J Clin Endocrinol Metab 101(8):3155–3162. https://doi.org/10.1210/JC.2016-1933
Coskun T, Sloop KW, Loghin C et al (2018) LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: from discovery to clinical proof of concept. Mol Metab 18:3–14. https://doi.org/10.1016/J.MOLMET.2018.09.009
Thomas MK, Nikooienejad A, Bray R et al (2021) Dual GIP and GLP-1 receptor agonist tirzepatide improves beta-cell function and insulin sensitivity in type 2 diabetes. J Clin Endocrinol Metab 106(2):388–396. https://doi.org/10.1210/CLINEM/DGAA863
Min T, Bain SC (2021) The role of tirzepatide, dual GIP and GLP-1 receptor agonist, in the management of Type 2 diabetes: the SURPASS clinical trials. Diabetes Ther 12(1):143–157. https://doi.org/10.1007/S13300-020-00981-0
Eli Lilly and Company (2021) Lilly Reports Robust Third-Quarter 2021 Financial Results as Pipeline Success Strengthens Future Growth Potential. https://investor.lilly.com/news-releases/news-release-details/lilly-reports-robust-third-quarter-2021-financial-results. Accessed 20 Nov 2021
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/BMJ.N71
Lawrance R, Degtyarev E, Griffiths P et al (2020) What is an estimand & how does it relate to quantifying the effect of treatment on patient-reported quality of life outcomes in clinical trials? J Patient Reported Outcomes 4(1):68. https://doi.org/10.1186/S41687-020-00218-5
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898. https://doi.org/10.1136/BMJ.L4898
Debray TPA, Moons KGM, Riley RD (2018) Detecting small-study effects and funnel plot asymmetry in meta-analysis of survival data: a comparison of new and existing tests. Res Synth Methods 9(1):41. https://doi.org/10.1002/JRSM.1266
Veroniki AA, Jackson D, Viechtbauer W et al (2016) Methods to estimate the between-study variance and its uncertainty in meta-analysis. Res Synth Methods 7(1):55–79. https://doi.org/10.1002/JRSM.1164
Frias JP, Nauck MA, Van J et al (2020) Efficacy and tolerability of tirzepatide, a dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist in patients with type 2 diabetes: a 12-week, randomized, double-blind, placebo-controlled study to evaluate different dose-escalation regimens. Diabetes Obes Metab 22(6):938–946. https://doi.org/10.1111/dom.13979
Dahl D, Onishi Y, Norwood P et al (2022) Effect of subcutaneous tirzepatide vs placebo added to titrated insulin glargine on glycemic control in patients with type 2 diabetes: the SURPASS-5 randomized clinical trial. JAMA 327(6):534–545. https://doi.org/10.1001/JAMA.2022.0078
Ludvik B, Giorgino F, Jódar E et al (2021) Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet 398(10300):583–598. https://doi.org/10.1016/S0140-6736(21)01443-4
Del Prato S, Kahn SE, Pavo I et al (2021) Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet 398(10313):1811–1824. https://doi.org/10.1016/S0140-6736(21)02188-7
Rosenstock J, Wysham C, Frías JP et al (2021) Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet 398(10295):143–155. https://doi.org/10.1016/S0140-6736(21)01324-6
Frias JP, Nauck MA, Van J et al (2018) Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet 392(10160):2180–2193. https://doi.org/10.1016/S0140-6736(18)32260-8
Frias JP, Davies MJ, Rosenstock J et al (2021) Efficacy and safety of tirzepatide vs. semaglutide once-weekly as add-on therapy to metformin in people with type 2 diabetes (SURPASS-2). Diabetes 70(Supplement_1):84–LB. https://doi.org/10.2337/db21-84-LB
Frías JP, Davies MJ, Rosenstock J et al (2021) Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med 385(6):503–515. https://doi.org/10.1056/NEJMoa2107519
Bhagavathula AS, Vidyasagar K, Tesfaye W (2021) Efficacy and safety of tirzepatide in patients with type 2 diabetes mellitus: a systematic review and meta-analysis of randomized phase II/III trials. Pharmaceuticals (Basel) 14(10):991. https://doi.org/10.3390/PH14100991
Kushner RF, Calanna S, Davies M et al (2020) Semaglutide 2.4 mg for the treatment of obesity: key elements of the STEP trials 1 to 5. Obesity (Silver Spring) 28(6):1050–1061. https://doi.org/10.1002/OBY.22794
Tsapas A, Karagiannis T, Kakotrichi P et al (2021) Comparative efficacy of glucose-lowering medications on body weight and blood pressure in patients with type 2 diabetes: a systematic review and network meta-analysis. Diabetes Obes Metab 23(9):2116–2124. https://doi.org/10.1111/DOM.14451
Frias JP, Bonora E, Ruiz LN et al (2021) Efficacy and safety of dulaglutide 3.0 mg and 4.5 mg versus dulaglutide 1.5 mg in metformin-treated patients with type 2 diabetes in a randomized controlled trial (AWARD-11). Diabetes Care 44(3):765–773. https://doi.org/10.2337/DC20-1473
Frías JP, Auerbach P, Bajaj HS et al (2021) Efficacy and safety of once-weekly semaglutide 2.0 mg versus 1.0 mg in patients with type 2 diabetes (SUSTAIN FORTE): a double-blind, randomised, phase 3B trial. Lancet Diabetes Endocrinol 9(9):563–574. https://doi.org/10.1016/S2213-8587(21)00174-1
Gerstein HC, Colhoun HM, Dagenais GR et al (2019) Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 394(10193):121–130. https://doi.org/10.1016/S0140-6736(19)31149-3
Authors’ relationships and activities
SDP declares grants from AstraZeneca and Boehringer Ingelheim, consulting fees from Applied Therapeutics, AstraZeneca, Boehringer Ingelheim, Eli Lilly and Company, Merck Sharpe and Dohme, Novartis Pharmaceuticals, Novo Nordisk and Sanofi, and honoraria for lectures from AstraZeneca, Boehringer Ingelheim, Eli Lilly and Company, Merck Sharpe and Dohme, Novartis Pharmaceuticals, Novo Nordisk and Sanofi. DRM reports receiving research support from Janssen, serving on advisory boards and as a consultant for Novo Nordisk, Novartis, Eli Lilly, Sanofi-Aventis, Janssen and Servier, and giving lectures for Novo Nordisk, Servier, Sanofi-Aventis, Eli Lilly, Novartis and Janssen. AT has received research support from Boehringer Ingelheim and Eli Lilly and has served on advisory boards for Boehringer Ingelheim and Novo Nordisk. EB has received research support and served on an advisory board for Novo Nordisk. All other authors declare that there are no relationships or activities that might bias, or be perceived to bias, their work.
Contribution statement
All authors significantly contributed to the manuscript and approved the final version for publication. TK, AT and EB conceived and designed the study. TK, IA and AL contributed to data acquisition. TK and IA carried out the statistical analysis. All authors contributed to interpretation of data. TK wrote the first manuscript draft, which was critically revised by IA, AL and AT. SDP, DRM and EB edited the final manuscript draft for important intellectual content. TK is the guarantor of this work.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM
(PDF 443 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Karagiannis, T., Avgerinos, I., Liakos, A. et al. Management of type 2 diabetes with the dual GIP/GLP-1 receptor agonist tirzepatide: a systematic review and meta-analysis. Diabetologia 65, 1251–1261 (2022). https://doi.org/10.1007/s00125-022-05715-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-022-05715-4