Abstract
Key message
Genetic diversity in Canadian soybean is maintained over decades of selection in two public breeding programs. Breeders have used a portion of the genetic diversity available in germplasm collections.
Abstract
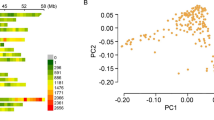
Both public and private breeding efforts have been critical for the development of soybean cultivars grown around the world. Global genetic diversity of soybean has been well characterized; however, this diversity is not well studied at the breeding program scale. The objective of this study was to characterize genetic diversity over decades of breeding in two public soybean breeding programs at the University of Guelph, Canada. To address this objective, a pedigree-related panel combining 296 soybean accessions from the Ridgetown and Guelph Campus breeding programs was studied. The accessions were genotyped using genotyping-by-sequencing, imputed using the GmHapMap reference genotypes resulting in more than 3.8M SNPs, further filtered to 77k SNPs. Population structure analysis did not identify structure between the breeding programs and historical germplasm. The linkage disequilibrium decay ranged from 400 to 600 kb on average in euchromatic regions. Nucleotide diversity over decades of breeding shows that historical accessions had the highest nucleotide diversity, with significant decreases corresponding to the initial breeding activity in Canada; however, genetic diversity has increased in the last 20 years in both breeding programs. Maturity gene E2 was nearly fixed for e2 in Ridgetown accessions, while unfixed in Guelph accessions. Comparison of the breeding programs to the USDA germplasm collection reveals that breeders have only used a portion of the available genetic diversity, allowing future breeders to exploit this untapped resource. The approach used in this study may be of interest to other breeding programs for evaluating changes in genetic diversity resulting from breeding activities.




Similar content being viewed by others
References
Bandillo N, Jarquin D, Song Q et al (2015) A population structure and genome-wide association analysis on the USDA soybean germplasm collection. Plant Genome. https://doi.org/10.3835/plantgenome2015.04.0024
Bastidas M (2008) Validation of QTLs associated with soybean cyst nematode resistance in two inter-specific soybean populations. University of Guelph, Guelph
Browning BL, Browning SR (2016) Genotype imputation with millions of reference samples. Am J Hum Genet 98:116–126. https://doi.org/10.1016/j.ajhg.2015.11.020
Bruce RW, Grainger CM, Ficht A et al (2019) Trends in soybean trait improvement over generations of selective breeding. Crop Sci 59:12. https://doi.org/10.2135/cropsci2018.11.0664
Chang CC, Chow CC, Tellier LC et al (2015) Second-generation PLINK: rising to the challenge of larger and richer datasets. GigaScience. https://doi.org/10.1186/s13742-015-0047-8
Danecek P, Auton A, Abecasis G et al (2011) The variant call format and VCFtools. Bioinformatics 27:2156–2158. https://doi.org/10.1093/bioinformatics/btr330
Deschamps S, Llaca V, May GD (2012) Genotyping-by-sequencing in plants. Biology 1:460–483. https://doi.org/10.3390/biology1030460
Elshire RJ, Glaubitz JC, Sun Q et al (2011) A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6:e19379. https://doi.org/10.1371/journal.pone.0019379
Fu Y-B (2015) Understanding crop genetic diversity under modern plant breeding. Theor Appl Genet 128:2131–2142. https://doi.org/10.1007/s00122-015-2585-y
Gizlice Z, Carter TE, Burton JW (1994) Genetic base for North American public soybean cultivars released between 1947 and 1988. Crop Sci 34:1143–1151
Grainger CM, Rajcan I (2013) Characterization of the genetic changes in a multi-generational pedigree of an elite Canadian soybean cultivar. Theor Appl Genet. https://doi.org/10.1007/s00122-013-2211-9
Grant D, Nelson RT, Cannon SB, Shoemaker RC (2010) SoyBase, the USDA-ARS soybean genetics and genomics database. Nucleic Acids Res 38:D843–D846. https://doi.org/10.1093/nar/gkp798
Huang X, Kurata N, Wei X et al (2012) A map of rice genome variation reveals the origin of cultivated rice. Nature 490:497–501. https://doi.org/10.1038/nature11532
Hyten DL, Song Q, Zhu Y et al (2006) Impacts of genetic bottlenecks on soybean genome diversity. Proc Natl Acad Sci 103:16666–16671
Hyten DL, Choi I-Y, Song Q et al (2007) Highly variable patterns of linkage disequilibrium in multiple soybean populations. Genetics 175:1937–1944. https://doi.org/10.1534/genetics.106.069740
Lam H-M, Xu X, Liu X et al (2010) Resequencing of 31 wild and cultivated soybean genomes identifies patterns of genetic diversity and selection. Nat Genet 42:1053–1059. https://doi.org/10.1038/ng.715
Langewisch T, Lenis J, Jiang G-L et al (2017) The development and use of a molecular model for soybean maturity groups. BMC Plant Biol. https://doi.org/10.1186/s12870-017-1040-4
Moore RM, Harrison AO, McAllister SM, Wommack KE (2018) Iroki: automatic customization and visualization of phylogenetic trees. bioRxiv. https://doi.org/10.1101/106138
Moose SP, Mumm RH (2008) Molecular plant breeding as the foundation for 21st century crop improvement. Plant Physiol 147:969–977. https://doi.org/10.1104/pp.108.118232
Nielsen NH, Backes G, Stougaard J et al (2014) Genetic diversity and population structure analysis of European hexaploid bread wheat (Triticum aestivum L.) varieties. PLoS ONE 9:e94000. https://doi.org/10.1371/journal.pone.0094000
Palomeque L, Li-Jun L, Li W et al (2009a) QTL in mega-environments: II. Agronomic trait QTL co-localized with seed yield QTL detected in a population derived from a cross of high-yielding adapted × high-yielding exotic soybean lines. Theor Appl Genet 119:429–436. https://doi.org/10.1007/s00122-009-1048-8
Palomeque L, Li-Jun L, Li W et al (2009b) QTL in mega-environments: I. Universal and specific seed yield QTL detected in a population derived from a cross of high-yielding adapted × high-yielding exotic soybean lines. Theor Appl Genet 119:417–427. https://doi.org/10.1007/s00122-009-1049-7
Palomeque L, Liu L-J, Li W et al (2010) Validation of mega-environment universal and specific QTL associated with seed yield and agronomic traits in soybeans. Theor Appl Genet 120:997–1003. https://doi.org/10.1007/s00122-009-1227-7
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Raj A, Stephens M, Pritchard JK (2014) fastSTRUCTURE: variational inference of population structure in large SNP data sets. Genetics 197:573–589. https://doi.org/10.1534/genetics.114.164350
Rossi ME, Orf JH, Liu L-J et al (2013) Genetic basis of soybean adaptation to North American vs. Asian mega-environments in two independent populations from Canadian-Chinese crosses. Theor Appl Genet 126:1809–1823. https://doi.org/10.1007/s00122-013-2094-9
Schmutz J, Cannon SB, Schlueter J et al (2010) Genome sequence of the palaeopolyploid soybean. Nature 463:178–183. https://doi.org/10.1038/nature08670
Sonah H, Bastien M, Iquira E et al (2013) An improved genotyping by sequencing (GBS) approach offering increased versatility and efficiency of SNP discovery and genotyping. PLoS ONE 8:e54603. https://doi.org/10.1371/journal.pone.0054603
Song Q, Hyten DL, Jia G et al (2013) Development and evaluation of SoySNP50K, a high-density genotyping array for soybean. PLoS ONE 8:e54985. https://doi.org/10.1371/journal.pone.0054985
Song Q, Hyten DL, Jia G et al (2015) Fingerprinting soybean germplasm and its utility in genomic research. G3 Genes Genomes Genet 5:1999–2006. https://doi.org/10.1534/g3.115.019000
Song Q, Jenkins J, Jia G et al (2016) Construction of high resolution genetic linkage maps to improve the soybean genome sequence assembly Glyma1.01. BMC Genom. https://doi.org/10.1186/s12864-015-2344-0
Torkamaneh D, Belzile F (2015) Scanning and filling: ultra-dense SNP genotyping combining genotyping-by-sequencing, SNP array and whole-genome resequencing data. PLoS ONE 10:e0131533. https://doi.org/10.1371/journal.pone.0131533
Torkamaneh D, Laroche J, Bastien M et al (2017) Fast-GBS: a new pipeline for the efficient and highly accurate calling of SNPs from genotyping-by-sequencing data. BMC Bioinformatics. https://doi.org/10.1186/s12859-016-1431-9
Torkamaneh D, Boyle B, Belzile F (2018a) Efficient genome-wide genotyping strategies and data integration in crop plants. Theor Appl Genet 131:499–511. https://doi.org/10.1007/s00122-018-3056-z
Torkamaneh D, Laroche J, Tardivel A et al (2018b) Comprehensive description of genomewide nucleotide and structural variation in short-season soya bean. Plant Biotechnol J 16:749–759. https://doi.org/10.1111/pbi.12825
Torkamaneh D, Laroche J, Valliyodan B et al (2019) Soybean haplotype map (GmHapMap): a universal resource for soybean translational and functional genomics. bioRxiv. https://doi.org/10.1101/534578
Valliyodan B, Qiu D, Patil G et al (2016) Landscape of genomic diversity and trait discovery in soybean. Sci Rep. https://doi.org/10.1038/srep23598
van de Wouw M, van Hintum T, Kik C et al (2010) Genetic diversity trends in twentieth century crop cultivars: a meta analysis. Theor Appl Genet 120:1241–1252. https://doi.org/10.1007/s00122-009-1252-6
Van Inghelandt D, Melchinger AE, Lebreton C, Stich B (2010) Population structure and genetic diversity in a commercial maize breeding program assessed with SSR and SNP markers. Theor Appl Genet 120:1289–1299. https://doi.org/10.1007/s00122-009-1256-2
Zhang C, Dong S-S, Xu J-Y et al (2018) PopLDdecay: a fast and effective tool for linkage disequilibrium decay analysis based on variant call format files. Bioinformatics 35(10):1786–1788
Zhou Z, Jiang Y, Wang Z et al (2015) Resequencing 302 wild and cultivated accessions identifies genes related to domestication and improvement in soybean. Nat Biotechnol 33:408–414. https://doi.org/10.1038/nbt.3096
Zhou L, Luo L, Zuo J-F et al (2016) Identification and validation of candidate genes associated with domesticated and improved traits in soybean. Plant Genome. https://doi.org/10.3835/plantgenome2015.09.0090
Acknowledgements
We would like to acknowledge support from the Natural Sciences and Engineering Research Council of Canada Collaborative Research and Development Grant program as well as our industry partners including Grain Farmers of Ontario, Secan and Huron Commodities Inc. We would also like to acknowledge sequencing support from Brian Boyle at the Genomic Analysis Platform in the Institut de Biologie Intégrative et des Systèmes (IBIS) at Université Laval. Additional thanks to the field crews at Guelph and Ridgetown Campuses for providing additional historical and pedigree data used in this study.
Funding
Funding was provided by National Sciences and Research Council of Canada (Grant No. 447948-13), Huron Commodities Inc. (Grant No. 447948-13), SeCan (Grant No. 447948-13), Grain Farmers of Ontario (Grant No. 447948-13).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by Volker Hahn.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bruce, R.W., Torkamaneh, D., Grainger, C. et al. Genome-wide genetic diversity is maintained through decades of soybean breeding in Canada. Theor Appl Genet 132, 3089–3100 (2019). https://doi.org/10.1007/s00122-019-03408-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-019-03408-y