Abstract
Objectives
We investigated different computed tomography (CT) features between Omicron-variant and original-strain SARS-CoV‑2 pneumonia to facilitate the clinical management.
Materials and methods
Medical records were retrospectively reviewed to select patients with original-strain SARS-CoV‑2 pneumonia from February 22 to April 22, 2020, or Omicron-variant SARS-CoV‑2 pneumonia from March 26 to May 31, 2022. Data on the demographics, comorbidities, symptoms, clinical types, and CT features were compared between the two groups.
Results
There were 62 and 78 patients with original-strain or Omicron-variant SARS-CoV‑2 pneumonia, respectively. There were no differences between the two groups in terms of age, sex, clinical types, symptoms, and comorbidities. The main CT features differed between the two groups (p = 0.003). There were 37 (59.7%) and 20 (25.6%) patients with ground-glass opacities (GGO) in the original-strain and Omicron-variant pneumonia, respectively. A consolidation pattern was more frequently observed in the Omicron-variant than original-strain pneumonia (62.8% vs. 24.2%). There was no difference in crazy-paving pattern between the original-strain and Omicron-variant pneumonia (16.1% vs. 11.6%). Pleural effusion was observed more often in Omicron-variant pneumonia, while subpleural lesions were more common in the original-strain pneumonia. The CT score in the Omicron-variant group was higher than that in the original-strain group for critical-type (17.00, 16.00–18.00 vs. 16.00, 14.00–17.00, p = 0.031) and for severe-type (13.00, 12.00–14.00 vs 12.00, 10.75–13.00, p = 0.027) pneumonia.
Conclusion
The main CT finding of the Omicron-variant SARS-CoV‑2 pneumonia included consolidations and pleural effusion. By contrast, CT findings of original-strain SARS-CoV‑2 pneumonia showed frequent GGO and subpleural lesions, but without pleural effusion. The CT scores were also higher in the critical and severe types of Omicron-variant than original-strain pneumonia.
Zusammenfassung
Ziel
Verschiedene Computertomographie(CT)-Merkmale wurden untersucht und zwischen einer Pneumonie durch die Omikron-Variante und einer Pneumonie durch den Original-SARS-CoV-2-Stamm verglichen, mit dem Ziel, die klinische Behandlung zu erleichtern.
Material und Methoden
Retrospektiv wurden Krankenunterlagen ausgewertet, um aus dem Zeitraum vom 22. Februar bis 22. April 2020 Patienten mit einer Pneumonie durch den Original-SARS-CoV-2-Stamm und aus dem Zeitraum vom 26. März bis 31. Mai 2022 Patienten mit einer Pneumonie durch die SARS-CoV-2-Omikron-Variante herauszusuchen. Zwischen den beiden Gruppen wurden Daten zu Demografie, Komorbiditäten, Symptomen, klinischen Typen und CT-Merkmalen verglichen.
Ergebnisse
Es fanden sich 62 Patienten mit einer SARS-CoV-2-Pneumonie durch den Originalstamm und 78 Patienten mit einer SARS-CoV-2-Pneumonie durch die Omikron-Variante. Zwischen den beiden Gruppen bestanden keine Unterschiede in Bezug auf Alter, Geschlecht, klinische Typen, Symptome und Komorbiditäten. Die Hauptmerkmale in der CT unterschieden sich jedoch zwischen den beiden Gruppen (p = 0,003). Es gab 37 (59,7 %) bzw. 20 (25,6 %) Patienten mit milchglasartiger Trübung bei der Originalstamm- bzw. bei der Omikron-Varianten-Pneumonie. Ein Konsolidierungsmuster wurde häufiger bei der Omikron-Varianten- als bei der Originalstammpneumonie festgestellt (62,8 vs. 24,2 %). Kein Unterschied bestand hinsichtlich eines Mosaikpflastermusters zwischen der Originalstamm- und der Omikron-Varianten-Pneumonie (16,1 vs. 11,6 %). Ein Pleuraerguss wurde häufiger bei der Omikron-Varianten-Pneumonie beobachtet, während subpleurale Läsionen häufiger bei der Originalstammpneumonie vorkamen. Der CT-Score in der Omikron-Varianten-Gruppe war höher als in der Originalstammgruppe für die Pneumonie vom kritischen Typ (17,00; 16,00–18,00 vs. 16,00; 14,00–17,00; p = 0,031) und vom schweren Typ (13,00; 12,00–14,00 vs. 12,00; 10,75–13,00; p = 0,027).
Schlussfolgerung
Zu den Hauptmerkmalen der Omikron-Varianten-SARS-CoV-2-Pneumonie in der CT gehörten Konsolidierung und Pleuraerguss. Dagegen zeigten sich als CT-Befunde der Originalstamm-SARS-CoV-2-Pneumonie häufig milchglasartige Trübungen und subpleurale Läsionen, aber ohne Pleuraerguss. Auch waren die CT-Scores bei dem kritischen und dem schweren Typ der Omikron-Varianten-Pneumonie höher als bei der Originalstammpneumonie.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The coronavirus disease 2019 (COVID-19), caused by severe acute respiratory disease coronavirus 2 (SARS-CoV-2), was first reported in December 2019 and has infected and caused the deaths of millions of people worldwide [1, 2]. SARS-CoV‑2 variants are continuously detected as the number of infected individuals grow [3]. Different variants have spread globally and have caused asynchronously occurring periodic waves of infection in different regions in the world. From October to December 2020, Beta variants (b1.351 and b1.617.2) were identified in South Africa [4]. The Delta variant was first detected in India, while its outbreak started in South Africa in May 2021. In November 2021, the genome surveillance team in South Africa and Botswana identified another SARS-CoV‑2 variant, Omicron (B.1.529), which was associated with the rapid infection recurrence in South Africa [5, 6]. However, compared with the original strain, the virulence of the variants has gradually decreased with a lower rate of severe cases of disease, hospitalizations, and deaths; however, their transmissibility increased. Patients with COVID-19, regardless of the causative variant, can have asymptomatic disease or present with symptoms such as fever, cough, sore throat, musculoskeletal pain, and shortness of breath [7, 8]. Some patients with COVID-19 have characteristic computed tomography (CT) findings, which mainly include pulmonary ground-glass opacities (GGO), mostly located in the periphery of the lung or under the pleura, without or with a small amount of pleural effusion [9,10,11]. During the early stage of the pandemic, CT was used as one of the COVID-19 diagnostic methods in Hubei, China, due to the shortage in availability of reverse-transcription polymerase chain reaction (RT-PCR) kits and the possibility of false-negative results from respiratory tract sampling. With the more prevalent Omicron variant, some studies have reported different CT findings between Omicron-variant pneumonia and Delta-variant pneumonia [12,13,14]. However, there has been no study comparing the chest CT images between Omicron variants and the original strain (the strain of SARS-CoV‑2 epidemic in the city of Wuhan, China, during 2019–2020). This study aimed to reveal differences in chest CT findings of COVID-19 pneumonia between the original strain of SARS-CoV-2and Omicron variants.
Materials and methods
Study design and participants
We performed a retrospective study and reviewed patients with COVID in Hospital A from January 20 to February 22, 2020, and in Hospital B and Hospital C from April 26 to May 31, 2022. The Institutional Research and Ethics Committee of the hospitals approved this study. All procedures involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Participant selection criteria were patients with confirmed original-strain SARS-CoV‑2 pneumonia from January 20 to February 22, 2020, or confirmed Omicron-variant SARS-CoV‑2 pneumonia from April 26 to May 31, 2022, when an Omicron variant epidemic was seen in some Chinese cities.
All patients underwent pulmonary CT evaluations. The diagnostic criteria for SARS-CoV‑2 pneumonia were based on the guideline from the National Health Commission of the People’s Republic of China [15].
The inclusion criteria were: (1) positive RT-PCR test result from nasal or throat swabs; (2) evidence of pneumonia on a thin-section chest CT scan; and (3) patients admitted for treatment. The exclusion criteria were: (1) age ≤ 17 years; (2) incomplete clinical records; (3) and/or pneumonia caused by other pathogens.
Data collection
Medical records were reviewed to collect demographic information, underlying comorbidities, and clinical symptoms. The severity of COVID-19 infection was classified into four clinical types as mild, moderate, severe, and critical, based on the published guideline ([15]; Table 1).
CT instruments and examination
All patients in this study underwent non-contrast-enhanced thin-section chest CT examination in the supine position during end-inspiration per standard protocol using a 64-row multidetector CT scanner (SOMATOM Perspective; Siemens, Munich, Germany). The scan ranged from the apex to the base of the lungs with the CT scanning parameter of 120 kV, automatic mAs, 1.0 mm slice thickness, 0.90 pitch, and field of view of 30–40 cm. Lung window imaging was reconstructed with a high-frequency algorithm (width, 1400–1600 HU; level, −400 to −600 HU).
Image analysis
The CT results were evaluated by experienced CT radiologists in a blinded fashion. Definitions of radiological terms such as GGO, crazy-paving pattern (GGO with superimposed inter- and intra-lobular septal thickening), and pulmonary consolidation were based on the standard glossary for thoracic imaging reported by the Fleischner Society [16]. In all cases, a semiquantitative CT severity scoring was proposed based on a previous publication [9, 17]. Each of the five lung lobes was assessed for the degree of involvement and classified as none (0%), minimal (1–25%), mild (26–50%), moderate (51–75%), or severe (76–100%). No involvement corresponded to a lobe score of 0; minimal, to a lobe score of 1; mild, to a lobe score of 2; moderate, to a lobe score of 3; and severe, to a lobe score of 4. An overall lung “total severity score” was obtained by summing the five lobe scores (range of scores, 0–20). Features such as pleural effusion, fibrosis, emphysema, subpleural lines, reversed “halo sign,” nodules, lymphadenopathy, and airway abnormalities were also described.
Statistical analysis
Continuous variables are presented as mean ± standard deviation or medians (interquartile range), depending on the normality test results. Categorical variables are presented as numbers with percentages. The Student t test, Mann–Whitney test, or Kruskal–Wallis test was performed for intergroup differences in continuous variables. The chi-square test was used to assess statistically significant differences between categorical variables. A value of p < 0.05 was considered statistically significant.
Results
Baseline characteristics of study participants
There were 62 and 78 patients with confirmed original-strain or Omicron-variant SARS-CoV‑2 pneumonia, respectively. Their age, sex distribution, COVID-19 types, vaccination status, clinical symptoms, comorbidities, and the time period from the symptom onset to the time point of the CT scan are shown in Table 2. There were no statistically significant differences between the two groups, except for the COVID-19 vaccination status. In addition, the majority of patients in both groups with the severe clinical type and critical clinical type were adults older than 75 years. In the original-strain SARS-CoV‑2 pneumonia group, the median age of patients with the severe type and the critical type was 56 years (65–68) and 76 years (73–82), respectively, while the median age of patients with the moderate type was only 52 years (44–60; p < 0.001). In the Omicron-variant SARS-CoV‑2 pneumonia group, the median age of patients with the moderate type, severe type, and critical type was 52 years (43–56), 73 years (65–76), and 76 years (74–86), respectively (p < 0.001). The median age of patients clinically classified as the severe type and the critical type was significantly higher than that of patients with the moderate type of disease in both groups (p = 0.000). The age of patients clinically classified as the severe type was significantly higher in the Omicron-variant pneumonia group than in the original-strain pneumonia group (P = 0.036) (Fig. 1).
Age differences in different clinical types between the two groups. The median age of patients clinically classified as the severe type and the critical type was significantly higher than that of patients with the moderate type of disease in both groups (p = 0.000). The age of patients clinically classified as the severe type was significantly higher in the Omicron-variant pneumonia group than in the original-strain pneumonia group (P = 0.036)
CT features
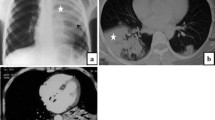
As shown in Table 3, the main imaging features differed between the two groups (p = 0.003). The GGO pattern was the most common characteristic in the original-strain pneumonia group, as it was present in 37 of 62 (59.7%) patients, compared with 26 of 78 (33.1%) patients in the Omicron-variant pneumonia group. The consolidation pattern was significantly more common in the Omicron-variant pneumonia group (55.1% vs. 24.2%). There was no significant difference in crazy-paving pattern between the two groups (original-strain pneumonia group 16.1% vs. Omicron-variant pneumonia group 11.6%). Most of the patients with Omicron-variant pneumonia had pleural effusion, significantly more than the patients in the original-strain SARS-CoV‑2 pneumonia group (41.0% vs. 11.9%). Subpleural lines were more common in the original-strain SARS-CoV‑2 pneumonia patients than in Omicron-variant pneumonia patients (46.8% vs. 15.4%). Fibrosis, emphysema, and lymphadenopathy did not differ significantly between the two groups. Reversed halo sign, nodules, or airway abnormalities were found in a few cases in both groups (Fig. 2).
Chest compute tomography findings on axial images of COVID-19 pneumonia between the original strain and the Omicron variant of SARS-CoV‑2. Original-strain SARS-CoV‑2 pneumonia: a subpleural lines and ground-glass opacities (GGO, arrows); b crazy-paving pattern (arrows); e subpleural lines and GGO (arrow); f GGO and consolidation (arrows). Omicron-variant SARS-CoV‑2 pneumonia: c consolidation (arrow); d consolidation, GGO, and pleural effusion (arrows); g consolidation, air bronchogram, and pleural effusion (arrows); h severe consolidation and pleural effusion (arrows)
In original-strain SARS-CoV‑2 pneumonia, the main CT pattern differed among the three clinical types (p = 0.006). Specifically, GGO was the main CT pattern in the moderate type (79.3%, 23/29) and consolidation was the main CT pattern in the critical type (60%, 9/15). By contrast, in Omicron-variant SARS-CoV‑2 pneumonia, the main CT pattern did not differ among the three clinical types (p = 0.653), with consolidation as the main CT pattern in all three types (Table 4).
Score of CT image
In the original-strain SARS-CoV‑2 pneumonia group, the chest CT semiquantitative score in the critical type (15.00, 14.00–18.75) was higher than that in the severe type (12.00, 11.00–13.00, p < 0.001) and the moderate type (3.00, 2.00–4.00, p < 0.001). The CT score was also significantly higher in the severe type than in the moderate type (p < 0.001). Figure 3 shows the chest CT semiquantitative score images compared between the severe and the moderate types.
Chest computed tomography (CT) from a 68-year-old woman with original-strain SARS-CoV‑2 pneumonia. Typical CT appearance: subpleural lines and ground-glass opacities. a Day 1 of admission: critical-type pneumonia, CT score of 18; b Day 10 of admission: moderate-type pneumonia, with significant improvement of CT imaging findings (CT score of 4). White arrows ground-glass opacities, red arrows subpleural lines
In the Omicron-variant SARS-CoV‑2 pneumonia group, the chest CT semiquantitative score in the critical type (17.50, 16.75–19.00) was higher than that in the severe type (14.00, 12.50–15.00, p < 0.001) and the moderate type (3.00, 3.00–4.00, p < 0.001). Moreover, the CT score was significantly higher in the severe type than in the moderate type (p < 0.001). Figure 4 shows the chest CT semiquantitative score images compared between the severe and the critical types.
Chest computed tomography (CT) from a 72-year-old man with Omicron-variant COVID-19 pneumonia. a Day 1 of admission: severe-type pneumonia, CT score of 12, and CT imaging shows consolidation, pleural effusion, and ground-glass opacities (GGO). b Day 3 of admission: critical-type pneumonia and CT imaging deterioration, CT score of 17, and CT imaging shows consolidation, pleural effusion, air bronchogram, and GGO. Red arrows consolidation
Comparisons between the two groups showed that the CT score in the Omicron-variant SARS-CoV‑2 pneumonia group was higher than that in the original-strain SARS-CoV‑2 pneumonia group in the critical clinical type (17.50, 16.75–19.00 vs. 15.00, 14.00–18.75, p = 0.033) and in the severe clinical type (14.00, 12.50–15.0 vs. 12.00, 11.00–13.00, p = 0.035). The CT score in the moderate clinical type was not significantly different between the two groups (p = 0.455; Fig. 5).
Comparison of computed tomography (CT) scores between original-strain and Omicron-variant SARS-CoV‑2 pneumonia in different clinical types. The CT score in the Omicron-variant SARS-CoV‑2 pneumonia group was higher than that in the original-strain SARS-CoV‑2 pneumonia group in patients with the critical clinical type (17.00, 16.00–18.00 vs. 16.00, 14.00–17.00, p = 0.031) and the severe clinical type (13.00, 12.00–14.00 vs. 12.00, 10.75–13.00, p = 0.027), while the CT score in patients with the moderate clinical type was not significantly different between the two groups (p = 0.377)
Discussion
In this study, we found that the clinical symptoms in both groups were mainly manifested as cough, fever, sore-throat, musculoskeletal symptoms, and dyspnea, while the symptoms of diarrhea and headache were less common Most of the patients in the two groups had one or more comorbidities. We also found that most of the patients with the severe type and the critical type of SARS-CoV‑2 pneumonia in the Omicron-variant group had not been vaccinated or had only received one dose of vaccine.
The risk of severe outcomes following Omicron-variant SARS-CoV‑2 infection was substantially lower. However, some studies found that age correlated with the severity of SARS-CoV‑2 pneumonia [18,19,20]. Most of the patients with the severe type or the critical type of COVID-19 pneumonia were older adults. A previous study in the United States showed that the mortality from COVID-19 varied markedly by age, ranging from 0.03% among patients aged 5–17 years to even 30.5% among patients aged 85 years or older [21]. Brian et al. [22] also reported that age was an independent risk factor for the development of pneumonia in patients infected by the Omicron variant. A cohort study suggested that Omicron variant-infected cases had a 59% lower risk of hospitalization, a 44% lower risk of any hospital attendance, and a 69% lower risk of death than Delta variant-infected individuals, with strong evidence for age dependence in the magnitude of the risk reduction [23]. According to Joseph et al. [24], Omicron-variant pneumonia was still associated with a high mortality rate at older ages and in patients with underlying diseases. In our study, the median age of the patients clinically classified as the severe type and the critical type of pneumonia was significantly higher than that of moderate-type patients in both groups. Although there was no difference in the median age between the two groups, the age of patients clinically classified as the severe type was significantly higher in the Omicron-variant pneumonia group than in the original-strain pneumonia group.
Some studies have shown that the majority of COVID-19 pneumonia cases have similar features on CT images, such as GGO or mixed GGO and consolidation [25,26,27], with peripheral distributions or bilateral, multifocal lower lung involvement [28]. Guan et al. [8] found that the most common patterns on chest CT were GGO (56.4%) and bilateral patchy shadows (51.8%). An initial prospective analysis showed that 40 of 41 (98%) infected patients had bilateral lung opacities. The lobular and subsegmental consolidation were the most typical findings on chest CT in Wuhan [29]. Chung et al. examined chest CT images of 21 infected patients and found a high rate of GGO and consolidation, sometimes with a rounded morphology and a peripheral lung distribution [30]. According to Kanne et al., patients with the severe type and critical type of pneumonia were more likely to have larger areas of bilateral consolidation on CT scans, whereas patients with the moderate type were more likely to have GGO and small areas of consolidation [31]. In our study, in the original-strain SARS-CoV‑2 pneumonia group, the GGO pattern was the most common characteristic, and it was present in 37 of 62 (59.7%) patients. Consolidation as the main CT pattern was found in 15 of 62 (24.2%) patients, while the crazy-paving pattern was observed in 10 of 62 (16.1%) patients. The most obvious related features were subpleural lines, which were found in 29 of 62 (46.8%) patients. The number of patients with pleural effusion was very small, only eight of 62 (12.9%). The results are consistent with those from previous studies [8, 28, 29].
SARS-CoV‑2 infection causes COVID-19. SARS-CoV‑2 spreads rapidly and mutates constantly, thereby giving rise to various variants, with different infectivity and lethality. Omicron is the most mutated SARS-CoV‑2 variant. According to current research, this variant is highly contagious, but causes mild clinical symptoms [32, 33]. Tsakok et al. found that Omicron-variant infection was associated with a lower CT score compared with the Delta-variant infection, and bronchial wall thickening was more common with the Omicron variant than with the Delta variant [14]. Askani et al. found that the Delta variant caused significantly more extensive lung involvement and showed changes classified as “typical” more often than the Omicron variant, while the Omicron variant was more likely associated with the absence of pulmonary changes on CT [13]. Soon Ho Yoon et al. found that Omicron was associated with less frequent, typical peripheral bilateral GGOs than the Delta variant [34]. On the other hand, another study showed that the GGO rate was similar between the previous non-Omicron wave and cluster-like GGO along the bronchi on chest CT was more frequently observed during the Omicron wave [35]. However, these studies did not consider patient age and the different clinical types. In this study, we found that the Omicron variant caused more severe lung imaging changes than the original strain of SARS-CoV‑2 in patients with the severe type and the critical type of pneumonia. The CT score in the Omicron-variant SARS-CoV‑2 pneumonia group was higher than that in the original-strain SARS-CoV‑2 pneumonia group in patients with the critical type of pneumonia, while the CT score in patients with the moderate type differed significantly between the two groups. The main CT pattern in the Omicron-variant SARS-CoV‑2 pneumonia group was different from that in the original-strain SARS-CoV‑2 pneumonia group. Consolidation was the most common CT pattern (49/78, 62.8%), while the GGO pattern was found in 20 of 78 (25.6%) cases and subpleural lines were found only in 11 of 78 (14.1%) cases. Many patients had pleural effusion (32/78, 41.0%). By contrast, in the original-strain SARS-CoV‑2 pneumonia group, GGO was the main CT pattern (79.3% in the moderate type and 61.1% in the severe type), while consolidation was the main CT pattern (60%) in the critical type, which was different from Omicron-variant SARS-CoV‑2 pneumonia, where consolidation was the main CT pattern in all three clinical types. The CT images indicating serious disease in Omicron-variant SARS-CoV‑2 pneumonia might be related to the fact that the patients in our study were older and had not received the COVID-19 vaccine. In addition, it is not clear whether there was a correlation between the different CT manifestations and the time point from symptom onset to the CT scan, although there was no statistical significance in the time point from symptom onset to the CT scan between the two groups.
Our study had several limitations. First, we had a relatively small number of patients. Second, since the condition of the patients often progressed very rapidly, most of them did not have repeat CT images and therefore there are no imaging data during the progression of the disease for some of the severe and critically ill patients, although certain imaging changes can be obtained from the bedside chest X‑ray. Third, because the data were collected from different periods and different hospitals in this study, it is unclear whether there are factors such as climate and treatment methods that may affect the patients’ condition. Fourth, the retrospective design of the study could introduce biases into our analysis. Larger clinical studies are needed to confirm our findings and further clarify CT differences between various strains of SARS-CoV‑2.
Conclusion
Most patients with original-strain SARS-CoV‑2 pneumonia had characteristic computed tomography (CT) findings, including frequent ground-glass opacity located in the peripheral lung areas or under the pleura, without or with a small amount of pleural effusion. Omicron-variant pneumonia lacked typical CT findings: The main CT finding was the lung consolidation with pleural effusion, which was different from the CT findings of original-strain pneumonia. In both original-strain and Omicron-variant pneumonia, most patients with the severe type and critical type were older adults and had one or more underlying diseases, with high CT scores.
Abbreviations
- COPD:
-
Chronic obstructive pulmonary disease
- COVID-19:
-
Coronavirus disease 2019
- CT:
-
Computed tomography
- GGO:
-
Ground-glass opacity
- SARS-CoV‑2:
-
Severe acute respiratory disease coronavirus 2
References
Zhou P, Yang X‑L, Wang X‑G et al (2020) A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579(7798):270–273
Zhu N, Dingyu Z, Wenling W et al (2020) A novel Coronavirus from patients with pneumonia in China, 2019. N Engl J Med 382(8):727–733. https://doi.org/10.1056/NEJMoa2001017
WHO. World Health Organization (2020) WHO Coronavirus disease (COVID-19) dashboard. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it
Al Hasan SM, Saulam J, Mikami F et al (2022) COVID-19 outbreak trends in South Africa: A comparison of Omicron (B.1.1.529), Delta (B.1.617.2), and Beta (B.1.351) variants outbreak periods. J Infect Public Health 15(7):726–733. https://doi.org/10.1016/j.jiph.2022.05.011
WHO. World Health Organization (2021) Classification of omicron (B.1.1.529): SARS-coV‑2 variant of concern. https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern
Hojjat Jodaylami M, Djaïleb A, Ricard P et al (2021) Cross-reactivity of antibodies from non-hospitalized COVID-19 positive individuals against the native, B.1.351, B.1.617.2, and P.1 SARS-CoV‑2 spike proteins. Sci Rep. https://doi.org/10.1038/s41598-021-00844-z
Struyf T, Deeks JJ, Dinnes J et al (2020) Cochrane COVID-19 diagnostic test accuracy group. 2020. Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19 disease. Cochrane Database Syst Rev 7:CD13665. https://doi.org/10.1002/14651858.CD013665
Guan W, Ni Z, Hu Y et al (2020) Clinical characteristics of Coronavirus disease 2019 in China. NEJM 6:1708–1720. https://doi.org/10.1056/NEJMoa2002032
Francone M, Iafrate F, Masci GM et al (2020) Chest CT score in COVID-19 patients: correlation with disease severity and short-term prognosis. Eur Radiol 30:6808–6817. https://doi.org/10.1007/s00330-020-07033-y
Grassi R, Belfore MP, Montanelli A et al (2021) COVID-19 pneumonia: computer-aided quantifcation of healthy lung parenchyma, emphysema, ground glass and consolidation on chest computed tomography (CT). Radiol med 126:553–560. https://doi.org/10.1007/s11547-020-01305-9
Chung M, Bernheim A, Xueyan M et al (2020) CT imaging features of 2019 novel Coronavirus (2019-ncoV). Radiology 295:202–207. https://doi.org/10.1148/radiol.2020200230
Maggialetti N, Villanova I, Castrì A et al COVID-19 in Italy: comparison of CT findings from time zero to the delta variant. Microorganisms. https://doi.org/10.3390/microorganisms10040796
Askani E, Mueller-Peltzer K, Madrid J et al (2022) Computed tomographic imaging features of COVID-19 pneumonia caused by the delta (B.1.617.2) and omicron (B.1.1.529) variant in a German nested cohort pilot study group. Tomography 8(5):2435–2449. https://doi.org/10.3390/tomography8050202
Tsakok MT, Watson RA, Saujani SJ et al (2022) Reduction in chest CT severity and improved hospital outcomes in SARS-coV‑2 omicron compared with delta variant infection. Radiology. https://doi.org/10.1148/radiol.220533
Lin L (2020) Interpretation of guidelines for diagnosis and treatment of novel coronavirus (2019-ncoV) infection by the national health commission (trail version 8). Zhonghua Yi Xue Za Zhi 100:6
Hansell DM, Bankier AA, MacMahon H, McLoud TC, Müller NL, Remy J (2008) Fleischner Society: glossary of terms for thoracic imaging. Radiology 246:697–722
Ye Z, Zhang Y, Wang Y, Huang Z et al (2020) Chest CT manifestations of new coronavirus disease 2019 (COVID-19): a pictorial review. Eur Radiol 30:4381–4389. https://doi.org/10.1007/s00330-020-06801-0
Chaomin W, Xiaoyan C, Yanping C (2020) Risk factors associated with acute respiratory distress syndrome and death in patients with Coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med 180(7):1–11. https://doi.org/10.1001/jamainternmed.2020.0994
Zhou F, Yu T, Du R et al (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395(10229):1054–1062. https://doi.org/10.1016/S0140-6736(20)30566-3
Xuecheng T, Zeyu H, Xiujun Z et al (2022) Old age is an independent risk factor for pneumonia development in patients with SARS-CoV‑2 omicron variant infection and a history of inactivated vaccine injection. Infect Drug Resist 15:5567–5573. https://doi.org/10.2147/IDR.S380005
Wiersinga WJ, Rhodes A, Cheng AC et al (2020) Pathophysiology, transmission, diagnosis, and treatment of Coronavirus disease 2019 (COVID-19). JAMA 324(8):782–793. https://doi.org/10.1001/jama.2020.12839
Garibaldi BT, Fiksel J, Muschelli J et al (2020) Patient trajectories among persons hospitalized for COVID-19. Ann Intern Med 22:M20–3905. https://doi.org/10.7326/M20-3905
Nyberg T, Ferguson NM, Nash SG et al (2022) Comparative analysis of the risks of hospitalisation and death associated with SARS-CoV‑2 omicron (B.1.1.529) and delta (B.1.617.2) variants in England: a cohort study. Lancet 399(10332):462–467. https://doi.org/10.1016/S0140-6736
Lewnard JA, Vennis XH, Patel MM et al (2022) Clinical outcomes associated with SARS-CoV‑2 Omicron (B.1.1.529) variant and BA.1/BA.1.1 or BA.2 subvariant infection in southern California. Nat Med 28(9):1933–1943. https://doi.org/10.1038/s41591-022-01887-z
Kong W, Agarwal P (2020) Chest imaging appearance of COVID-19 infection. Radiol Cardiothorac Imaging 2(1):e200028. https://doi.org/10.1148/ryct.2020200028
Bernheim A, Xueyan M, Mingqian H (2020) Chest CT findings in Coronavirus disease-19 (COVID-19): relationship to duration of infection. Radiology 295(3):200463. https://doi.org/10.1148/radiol.2020200463
Wu Y, Xie Y, Wang X (2020) Longitudinal CT findings in COVID-19 pneumonia: case presenting organizing pneumonia pattern. Radiol Cardiothorac Imaging. https://doi.org/10.1148/ryct.2020200031
Ng M, Lee E, Yang J et al (2020) Imaging profile of the COVID-19 infection: radiologic findings and literature review. Radiol Cardiothorac Imaging. https://doi.org/10.1148/ryct.2020200034
Huang C, Wang Y, Li X et al (2020) Clinical features of patients infected with 2019 novel Coronavirus in Wuhan, China. Lancet 20:30183–30185. https://doi.org/10.1016/S0140-6736(20)30183-5
Chung M, Bernheim A, Mei X et al (2020) CT imaging features of 2019 novel coronavirus (2019-nCoV). Radiology 295(1):202–207. https://doi.org/10.1148/radiol.2020200230
Kanne JP (2020) Chest CT findings in 2019 novel Coronavirus (2019-ncoV) infections from Wuhan, China: key points for the radiologist. Radiology 295(1):16–17. https://doi.org/10.1148/radiol.2020200241
Khandia R, Singhal S, Alqahtani T et al (2022) Emergence of SARS-CoV‑2 Omicron (B.1.1.529) variant, salient features, high global health concerns and strategies to counter it amid ongoing COVID-19 pandemic. Environ Res 209:112816. https://doi.org/10.1016/j.envres.2022.112816
Wang L, Berger NA, Kaelber DC et al (2022) Comparison of outcomes from COVID infection in pediatric and adult patients before and after the emergence of omicron. medRxiv. https://doi.org/10.1101/2021.12.30
Soon Ho Y, Jong Hyuk L, Baek-Nam K (2023) Chest CT findings in hospitalized patients with SARS-coV-2: delta versus omicron variants. Radiology 306(1):252–260. https://doi.org/10.1148/radiol.220676
Ito N, Kitahara Y, Miwata K, Okimoto M, Takafuta T (2022) Comparison of COVID-19 pneumonia during the SARS-coV‑2 omicron wave and the previous non-omicron wave in a single facility. Respir Investig 60(6):772–778. https://doi.org/10.1016/j.resinv.2022.08.001
Acknowledgements
We thank LetPub (www.letpub.com) for linguistic assistance and pre-submission expert review.
Funding
This study was sponsored by the Research Project of Science and Technology Department of Fujian Province, China (Grant 2021J01123491).
Availability of data and materials
The data that support the findings of this study are available from the corresponding author on reasonable request, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available.
Author information
Authors and Affiliations
Contributions
T. Guoliang contributed to the conception of the study; Z. Pingping performed the data analyses and wrote the manuscript; Z. Yanyu contributed significantly to analysis and manuscript preparation; H. Qiming performed the analysis of CT images; W. Yi performed the analysis of CT images.
Corresponding author
Ethics declarations
Conflict of interest
Z. pingping, Z. Yanyu, S. Xuri, H. Qiming, W. Yi and T. Guoliang declare that they have no competing interests.
The Institutional Research and Ethics Committee of the Hospital approved this study. All procedures involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
The supplement containing this article is not sponsored by industry.
Additional information
Zeng pingping, Zhou Yanyu, Tan Guoliang have contributed equally to the work.

Scan QR code & read article online
Rights and permissions
About this article
Cite this article
pingping, Z., Yanyu, Z., Xuri, S. et al. Comparison between original SARS-CoV-2 strain and omicron variant on thin-section chest CT imaging of COVID-19 pneumonia. Radiologie 63 (Suppl 2), 55–63 (2023). https://doi.org/10.1007/s00117-023-01147-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00117-023-01147-2