Abstract
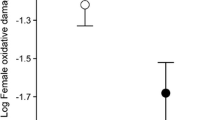
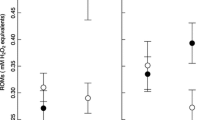
Carotenoid pigments are responsible for many of the red, yellow and orange plumage and integument traits seen in birds. One idea suggests that since carotenoids can act as antioxidants, carotenoid-mediated colouration may reveal an individual's ability to resist oxidative damage. In fact, there is currently very little information on the effects of most dietary-acquired antioxidants on oxidative stress in wild birds. Here, we assessed the impacts on oxidative damage, plasma antioxidants, growth and plumage colouration after supplementing nestling blue tits Cyanistes caeruleus with one of three diets; control, carotenoid treatment or α-tocopherol treatment. Oxidative damage was assessed by HPLC analysis of plasma levels of malondialdehyde (MDA), a by-product of lipid peroxidation. Contrary to predictions, we found no differences in oxidative damage, plumage colouration or growth rate between treatment groups. Although plasma lutein concentrations were significantly raised in carotenoid-fed chicks, α-tocopherol treatment had no effect on concentrations of plasma α-tocopherol compared with controls. Interestingly, we found that faster growing chicks had higher levels of oxidative damage than slower growing birds, independent of treatment, body mass and condition at fledging. Moreover, the chromatic signal of the chest plumage of birds was positively correlated with levels of MDA but not plasma antioxidant concentrations: more colourful nestlings had higher oxidative damage than less colourful individuals. Thus, increased carotenoid-mediated plumage does not reveal resistance to oxidative damage for nestling blue tits, but may indicate costs paid, in terms of oxidative damage. Our results indicate that the trade-offs between competing physiological systems for dietary antioxidants are likely to be complex in rapidly developing birds. Moreover, interpreting the biological relevance of different biomarkers of antioxidant status represents a challenge for evolutionary ecology.




Similar content being viewed by others
References
Adelmann R, Saul RL, Ames BN (1988) Oxidative damage to DNA: Relation to species metabolic rate and life span. Proc Natl Acad Sci USA 85:2706–2708
Aguilera E, Amat JA (2007) Carotenoids, immune response and the expression of sexual ornaments in male greenfinches (Carduelis chloris). Naturwissenschaften 94(11):895–902
Arnold KE, Ramsay SL, Donaldson C, Adam A (2007) Parental prey selection affects risk-taking behaviour and spatial learning in avian offspring. Proc R Soc Biol Sci Ser B 274(1625):2563–2569
Arnold KE, Ramsay SL, Henderson L, Larcombe SD (2010) Seasonal variation in diet quality: anti-oxidants, invertebrates and blue tits Cyanistes caeruleus. Biol J Linn Soc 99(4):708–717
Bertrand S, Faivre B, Sorci G (2006) Do carotenoid-based sexual traits signal the availability of non-pigmentary antioxidants? J Exp Biol 209(22):4414–4419
Biard C, Surai PF, Moller AP (2005) Effects of carotenoid availability during laying on reproduction in the blue tit. Oecologia 144(1):32–44
Biard C, Surai PF, Moller AP (2006) Carotenoid availability in diet and phenotype of blue and great tit nestlings. J Exp Biol 209(6):1004–1015
Biard C, Hardy C, Motreuil S, Moreau J (2009) Dynamics of PHA-induced immune response and plasma carotenoids in birds: should we have a closer look? J Exp Biol 212(9):1336–1343
Blount JD, Metcalfe NB, Birkhead TR, Surai PF (2003) Carotenoid modulation of immune function and sexual attractiveness in zebra finches. Science 300(5616):125–127
Blount JD, Houston DC, Surai PF, Moller AP (2004) Egg-laying capacity is limited by carotenoid pigment availability in wild gulls Larus fuscus. Proc R Soc Biol Sci Ser B 271:S79–S81
Bohm F, Edge R, Land EJ, McGarvey DJ, Truscott TG (1997) Carotenoids enhance vitamin E antioxidant efficiency. J Am Chem Soc 119(3):621–622
Brown WP, Eggermont P, LaRiccia V, Roth RR (2007) Are parametric models suitable for estimating avian growth rates? J Avian Biol 38(4):495–506
Burton GW, Ingold KU (1981) Autoxidation of biological molecules 1. The antioxidant activity of vitamin E and related chain breaking phenolic antioxidants in vitro. J Am Chem Soc 103(21):6472–6477
Burton GW, Joyce A, Ingold KU (1983) Is vitamin E the only lipid-soluble, chain-breaking antioxidant in human blood plasma and erythrocyte membranes? Arch Biochem Biophys 221(1):281–290
Cassey P (2009) Biological optics: seeing colours in the dark. Curr Biol 19(23):R1083–R1084
Cohen AA, McGraw KJ (2009) No simple measures for antioxidant status in birds: complexity in inter- and intraspecific correlations among circulating antioxidant types. Funct Ecol 23(2):310–320
Costantini D, Moller AP (2008) Carotenoids are minor antioxidants for birds. Funct Ecol 22(2):367–370
Costantini D, Verhulst S (2009) Does high antioxidant capacity indicate low oxidative stress? Funct Ecol 23(3):506–509
Costantini D, Coluzza C, Fanfani A, Dell’Omo G (2007a) Effects of carotenoid supplementation on colour expression, oxidative stress and body mass in rehabilitated captive adult kestrels (Falco tinnunculus). J Comp Physiol, B Biochem Syst Environ Physiol 177:723–731
Costantini D, Fanfani A, Dell’Omo G (2007b) Carotenoid availability does not limit the capability of nestling kestrels (Falco tinnunculus) to cope with oxidative stress. J Exp Biol 210(7):1238–1244
Cucco M, Guasco B, Malacarne G, Ottonelli R (2006) Effects of beta-carotene supplementation on chick growth, immune status and behaviour in the grey partridge, Perdix perdix. Behav Process 73(3):325–332
de Ayala RM, Martinelli R, Saino N (2006) Vitamin E supplementation enhances growth and condition of nestling barn swallows (Hirundo rustica). Behav Ecol Sociobiol 60(5):619–630
De Block M, Stoks R (2008) Compensatory growth and oxidative stress in a damselfly. Proc R Soc Biol Sci Ser B 275(1636):781–785
Dolnik VR, Gavrilov VM (1979) Bioenergetics of molt in the chaffinch (Fringilla coelebs). Auk 96(2):253–264
Dotan Y, Lichtenberg D, Pinchuk I (2004) Lipid peroxidation cannot be used as a universal criterion of oxidative stress. Prog Lipid Res 43(3):200–227
Fischer K, Zeilstra I, Hetz SK, Fiedler K (2004) Physiological costs of growing fast: does accelerated growth reduce pay-off in adult fitness? Evol Ecol 18(4):343–353
Fitze PS, Tschirren B (2006) No evidence for survival selection on carotenoid-based nestling coloration in great tits (Parus major). J Evol Biol 19(2):618–624
Fitze PS, Tschirren B, Richner H (2003a) Carotenoid-based colour expression is determined early in nestling life. Oecologia 137(1):148–152
Fitze PS, Kolliker M, Richner H (2003b) Effects of common origin and common environment on nestling plumage coloration in the great tit (Parus major). Evolution 57(1):144–150
Fuhrman B, Ben-Yaish L, Attias J, Hayek T, Aviram M (1997) Tomato lycopene and beta-carotene inhibit low density lipoprotein oxidation and this effect depends on the lipoprotein vitamin E content. Nutr Metab Cardiovasc Dis 7(6):433–443
Griffiths R, Double MC, Orr K, Dawson RJG (1998) A DNA test to sex most birds. Mol Ecol 7(8):1071–1075
Hadfield JD, Owens IPF (2006) Strong environmental determination of a carotenoid-based plumage trait is not mediated by carotenoid availability. J Evol Biol 19(4):1104–1114
Hall ME, Blount JD, Forbes S, Royle NJ (2010) Does oxidative stress mediate the trade-off between growth and self maintenance in structured families. Funct Ecol 24(2):365–373
Hart NS, Partridge JC, Cuthill IC, Bennett AT (2000) Visual pigments, oil droplets, ocular media and cone photoreceptor distribution in two species of passerine bird: the blue tit (Parus caeruleus L.) and the blackbird (Turdus melua L.). J Comp Physiol A 186(4):375–387
Hartley RC, Kennedy MW (2004) Are carotenoids a red herring in sexual display? Trends Ecol Evol 19(7):353–354
Hill GE (1995) Seasonal variation in circulating carotenoid pigments in the House Finch. Auk 112(4):1057–1061
Hill GE (1999) Is there an immunological cost to carotenoid-based ornamental coloration? Am Nat 154(5):589–595
Horak P, Surai PF, Ots I, Moller AP (2004) Fat soluble antioxidants in brood-rearing great tits Parus major: relations to health and appearance. J Avian Biol 35(1):63–70
Horak P, Zilmer M, Saks L, Ots I, Karu U, Zilmer K (2006) Antioxidant protection, carotenoids and the costs of immune challenge in greenfinches. J Exp Biol 209(21):4329–4338
Horak P, Saks L, Zilmer M, Karu U, Zilmer K (2007) Notes and comments—do dietary antioxidants alleviate the cost of immune activation? An experiment with greenfinches. Am Nat 170(4):625–635
Isaksson C, Andersson S (2008) Oxidative stress does not influence carotenoid mobilization and plumage pigmentation. Proc R Soc Biol Sci Ser B 275:309–314
Isaksson C, Ornborg J, Stephensen E, Andersson S (2005) Plasma glutathione and carotenoid coloration as potential biomarkers of environmental stress in great tits. EcoHealth 2(2):1612–9202
Isaksson C, McLaughlin P, Monaghan P, Andersson S (2007a) Carotenoid pigmentation does not reflect total non-enzymatic antioxidant activity in plasma of adult and nestling great tits, Parus major. Funct Ecol 21:1123–1129
Isaksson C, Von Post M, Andersson S (2007b) Sexual, seasonal, and environmental variation in plasma carotenoids in great tits, Parus major. Biol J Linn Soc 92(3):521–527
Kagan VE, Serbinova EA, Forte T, Scita G, Packer L (1992) Recycling of vitamin E in human low-density lipoproteins. J Lipid Res 33(3):385–397
Karu U, Saks L, Hõrak P (2008) Carotenoid-based plumage colouration is not affected by vitamin E supplementation in male greenfinches. Ecol Res 23:931–935
Kennedy TA, Liebler DC (1992) Peroxyl radical scavenging by beta-carotene in lipid bilayers—effect of oxygen partial-pressure. J Biol Chem 267(7):4658–4663
Krinsky NI (1989) Antioxidant functions of carotenoids. Free Radic Biol Med 7(6):617–635
Larcombe SD, Tregaskes CA, Coffey JS, Stevenson AE, Alexander L, Arnold KE (2008) The effects of short-term antioxidant supplementation on oxidative stress and flight performance in adult budgerigars Melopsittacus undulatus. J Exp Biol 211(17):2859–2864
Lozano GA (2001) Carotenoids, immunity, and sexual selection: comparing apples and oranges? Am Nat 158(2):200–203
Machlin LJ (1991). VITAMIN E. In Machlin LJ (ed) Food science and technology, 40 Handbook of vitamins, 2nd edn Xi + 595p. Marcel Dekker, Inc: New York, New York, USA; Basel, Switzerland Illus: 99–144
McGraw KJ, Ardia DR (2003) Carotenoids, immunocompetence, and the information content of sexual colors: an experimental test. Am Nat 162(6):704–712
Metcalfe NB, Monaghan P (2001) Compensation for a bad start: grow now, pay later? TREE 16(5):254–260
Olson VA, Owens IPF (1998) Costly sexual signals: are carotenoids rare, risky or required? Trends Ecol Evol 13(12):510–514
Olsson M, Wilson M, Isaksson C, Uller T, Mott B (2008) Carotenoid intake does not mediate a relationship between reactive oxygen species and bright colouration: experimental test in a lizard. J Exp Biol 211(8):1257–1261
Osorio D, Miklosi A, Gonda Z (1999) Visual ecology and perception of coloration patterns by domestic chicks. Evol Ecol 13(7–8):673–689
Pacifici RE, Davies KJA (1991) Protein, lipid and DNA repair systems in oxidative stress: the free-radical theory of aging revisited. Gerentology 37:166–180
Palozza P, Krinsky NI (1992) Beta-carotene and alpha-tocopherol are synergistic antioxidants. Arch Biochem Biophys 297(1):184–187
Parker RS (1996) Carotenoids. 4. Absorption, metabolism, and transport of carotenoids. FASEB J 10(5):542–551
Perez C, Lores M, Velando A (2008) Availability of nonpigmentary antioxidant affects red coloration in gulls. Behav Ecol 19(5):967–973
Peters A (2007) Testosterone and carotenoids: an integrated view of trade-offs between immunity and sexual signalling. BioEssays 29(5):427–430
Pike TW, Blount JD, Lindstrom J, Metcalfe NB (2007) Availability of non-carotenoid antioxidants affects the expression of a carotenoid-based sexual ornament. Biol Lett 3(4):353–356
Saks L, Ots I, Horak P (2003a) Carotenoid-based plumage coloration of male greenfinches reflects health and immunocompetence. Oecologia 134(3):301–307
Saks L, McGraw K, Horak P (2003b) How feather colour reflects its carotenoid content. Funct Ecol 17(4):555–561
Serra L, Griggio M, Licheri D, Pilastro A (2007) Moult speed constrains the expression of a carotenoid-based sexual ornament. J Evol Biol 20(5):2028–2034
Sodhi S, Sharma A, Brar APS, Brar RS (2008) Effect of alpha tocopherol and selenium on antioxidant status, lipid peroxidation and hepatopathy induced by malathion in chicks. Pestic Biochem Physiol 90(2):82–86
Surai PF (1999) Vitamin E in avian reproduction. Poult Avian Biol Rev 10(1):1–60
Surai PF, Ionov IA, Kuchmistova EF, Noble RC, Speake BK (1998) The relationship between the levels of alpha-tocopherol and carotenoids in the maternal feed, yolk and neonatal tissues: comparison between the chicken, turkey, duck and goose. J Sci Food Agric 76(4):593–598
Tanner M, Richner H (2008) Ultraviolet reflectance of plumage for parent–offspring communication in the great tit (Parus major). Behav Ecol 19(2):369–373
Toyoda Y, Thomson LR, Langer A, Craft NE, Garnett KM, Nichols CR, Cheng KM, Dorey CK (2002) Effect of dietary zeaxanthin in tissue distribution of zeaxanthin and lutein in quail. Invest Ophth Vis Sci 43:1210–1221
Tschirren B, Fitze PS, Richner H (2003) Proximate mechanisms of variation in the carotenoid-based plumage coloration of nestling great tits (Parus major L.). J Evol Biol 16(1):91–100
von Schantz T, Bensch S, Grahn M, Hasselquist D, Wittzell H (1999) Good genes, oxidative stress and condition-dependent sexual signals. Proc R Soc Biol Sci Ser B 266(1414):1–12
Woodall AA, Britton G, Jackson MJ (1996) Dietary supplementation with carotenoids: effects on alpha-tocopherol levels and susceptibility of tissues to oxidative stress. Br J Nutr 76(2):307–317
Young IS, Trimble ER (1991) Measurement of malondialdehyde in plasma by high-performance liquid-chromatography with fluorometric detection. Ann Clin Biochem 28:504–508
Acknowledgements
We would like to thank Jo Coffey at WCPN for help with MDA analysis and the many fieldworkers who assisted in the chick feeding and data collection. Caroline Isaksson, Ruedi Nager and Staffan Andersson provided useful comments on the manuscript. SDL was funded by a Biotechnology and Biological Sciences Research Council Industrial CASE studentship and KEA by a Royal Society University Research Fellowship. This research adhered to the Association for the Study of Animal Behaviour Guidelines for the Use of Animals in Research, the legal requirements of the UK and all institutional guidelines.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Larcombe, S.D., Mullen, W., Alexander, L. et al. Dietary antioxidants, lipid peroxidation and plumage colouration in nestling blue tits Cyanistes caeruleus . Naturwissenschaften 97, 903–913 (2010). https://doi.org/10.1007/s00114-010-0708-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-010-0708-5