Abstract
Purpose
Imaging for treatment planning shortly after hydrogel injection is optimal for practical purposes, reducing the number of appointments. The aim was to evaluate the actual difference between early and late imaging.
Patients and methods
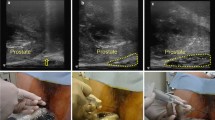
Treatment planning computed tomography (CT) was performed shortly after injection of 10 ml hydrogel (CT1) and 1–2 weeks later (CT2) for 3 patients. The hydrogel was injected via the transperineal approach after dissecting the space between the prostate and rectum with a saline/lidocaine solution of at least 20-ml. Hydrogel volume and distances between the prostate and rectal wall were compared. Intensity-modulated radiotherapy (IMRT) plans up to a dose of 78 Gy were generated (rectum V70 < 20 %, rectum V50 < 50 %; with the rectum including hydrogel volume for planning).
Results
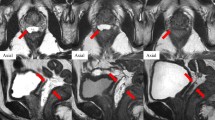
A mean planning treatment volume of 104 cm3 resulted for a prostate volume of 37 cm3. Hydrogel volumes of 30 and 10 cm3 were determined in CT1 and CT2, respectively. Distances between the prostate and rectal wall at the levels of the base, middle, and apex were 1.7 cm, 1.6 cm, 1.5 cm in CT1 and 1.3 cm, 1.2 cm, 0.8 cm in CT2, respectively, corresponding to a mean decrease of 24, 25, and 47 %. A small overlap between the PTV and the rectum was found only in 1 patient in CT2 (0.2 cm3). The resulting mean rectum (without hydrogel) V75, V70, V60, V50 increased from 0 %, 0 %, 0.6 %, 10 % in CT1 to 0.1 %, 1.2 %, 6 %, 20 % in CT2, respectively.
Conclusion
Treatment planning based on imaging shortly after hydrogel injection overestimates the actual hydrogel volume during the treatment as a result of not-yet-absorbed saline solution and air bubbles.
Zusammenfassung
Hintergrund
Die Bildgebung zur Bestrahlungsplanung kurz nach der Hydrogelinjektion ist aus praktischen Gründen wegen der geringen Terminanzahl optimal. Ziel der Studie war die Analyse von Unterschieden zwischen früher und später Bildgebung.
Fallberichte
Die Computertomographie zur Bestrahlungsplanung wurde kurz nach der Injektion von 10 ml Hydrogel (CT1) und 1–2 Wochen später (CT2) bei 3 Patienten durchgeführt. Das Hydrogel wurde transperineal nach Dissektion des Raums zwischen Prostata und Rektum mit ≥ 20 ml Kochsalz/Lidocain verabreicht. Das Hydrogelvolumen und die Abstände zwischen Prostata und Rektumwand wurden verglichen. IMRT-Pläne wurden mit einer Verschreibungsdosis von 78 Gy berechnet (Rektum V70 < 20 %, Rektum V50 < 50 %; Rektum einschließlich Hydrogel zur Planung).
Ergebnisse
Ein durchschnittliches Planungszielvolumen von 104 cm3 resultierte aus einem Prostatavolumen von 37 cm3. Hydrogelvolumina von 30 und 10 cm3 wurden in CT1 und CT2 bestimmt. Die Abstände zwischen Prostata und Rektumwand auf der Ebene von Basis, Mitte und Apex betrugen jeweils 1,7 cm/1,6 cm/1,5 cm in CT1 und 1,3 cm/1,2 cm/0,8 cm in CT2, entsprechend einer Abnahme von 24, 25 und 47 %. Eine geringe Überlappung zwischen PTV und Rektum fand sich nur bei einem einzigen Patienten in CT2 (0,2cm3). Die resultierenden durchschnittlichen Rektum-V75, -V70, -V60, -V50 (ohne Hydrogel) waren jeweils mit 0 %, 0 %, 0,6 %, 10 % in CT1 geringer als in CT2 mit jeweils 0,1 %, 1,2 %, 6 % und 20 %.
Schlussfolgerung
Die auf einer Bildgebung kurz nach der Hydrogelinjektion basierende Bestrahlungsplanung überschätzt das tatsächlich während der Behandlung vorhandene Hydrogelvolumen infolge noch nicht absorbierter Kochsalzlösung und Luftblasen.


Similar content being viewed by others
References
Pinkawa M, Piroth MD, Holy R et al (2011) Combination of dose escalation with technological advances (intensity-modulated and image-guided radiotherapy) is not associated with increased morbidity for patients with prostate cancer. Strahlenther Onkol 187:479–484
Alongi F, Fogliata A, Navarria P et al (2012) Moderate hypofractionation and simultaneous integrated boost with volumetric modulated arc therapy (RapidArc) for prostate cancer. Report of feasibility and acute toxicity. Strahlenther Onkol 188:990–996
Geier M, Astner ST, Duma MN et al (2012) Dose-escalated simultaneous integrated-boost treatment of prostate cancer patients via helical tomotherapy. Strahlenther Onkol 188:410–416
Prada PJ, Fernandez J, Martinez AA et al (2007) Transperineal injection of hyaluronic acid in anterior perirectal fat to decrease rectal toxicity from radiation delivered with intensity modulated brachytherapy or EBRT for prostate cancer patients. Int J Radiat Oncol Biol Phys 69:95–102
Prada PJ, Gonzalez H, Menendez C et al (2009) Transperineal injection of hyaluronic acid in the anterior perirectal fat to decrease rectal toxicity from radiation delivered with low-dose-rate brachytherapy for prostate cancer patients. Brachytherapy 8:210–217
Noyes WR, Hosford CC, Schultz SE (2012) Human collagen injections to reduce rectal dose during radiotherapy. Int J Radiat Oncol Biol Phys 82:1918–1922
Melchert C, Gez E, Bohlen G et al (2013) Interstitial biodegradable balloon for reduced rectal dose during prostate radiotherapy: results of a virtual planning investigation based on the pre- and post-implant imaging data of an international multicenter study. Radiother Oncol 106:210–214
Pinkawa M, Piroth MD, Holy R et al (2013) Spacer stability and prostate position variability during radiotherapy for prostate cancer applying a hydrogel to protect the rectal wall. Radiother Oncol 106:220–224
Hatiboglu G, Pinkawa M, Vallee JP et al (2012) Application technique: placement of a prostate-rectum spacer in men undergoing prostate radiation therapy. BJU Int 110:E647–E652
Pinkawa M, Piroth MD, Holy R et al (2012) Quality of life after intensity-modulated radiotherapy for prostate cancer with a hydrogel spacer. Matched-pair analysis. Strahlenther Onkol 188:917–925
Song DY, Herfarth KK, Uhl M et al (2013) A Multi-institutional clinical trial of rectal dose reduction via injected polyethylene-glycol hydrogel during intensity modulated radiation therapy for prostate cancer: analysis of dosimetric outcomes. Int J Radiat Oncol Biol Phys (in press)
Susil RC, McNutt TR, DeWeese TL et al (2010) Effects of prostate-rectum separation on rectal dose from external beam radiotherapy. Int J Radiat Oncol Biol Phys 76:1251–1258
Uhl M, Triest B van, Eble MJ et al (2013) Low rectal toxicity after dose escalated IMRT treatment of prostate cancer using an absorbable hydrogel for increasing and maintaining space between the rectum and prostate: results of a multi-institutional phase II trial. Radiother Oncol 106:215–219
Lawton CAF, Michalski J, El-Naqa I et al (2009) RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys 74:383–387
Wu Q, Mohan R, Niemierko A et al (2002) Optimization of intensity-modulated radiotherapy plans based on the equivalent uniform dose. Int J Radiat Oncol Biol Phys 52:224–235
Luxton G, Keall PJ, King CR (2008) A new formula for normal tissue complication probability (NTCP) as a function of equivalent uniform dose (EUD). Phys Med Biol 53:23–26
Burman C, Kutcher GJ, Emami B et al (1991) Fitting of normal tissue tolerance data to an analytic function. Int J Radiat Oncol Biol Phys 21:123–135
Rancati T, Fiorino C, Gagliardi G et al (2004) Fitting late rectal data using different NTCP models: results from an Italian multi-centri study (AIROPROSO101). Radiother Oncol 73:21–32
Pinkawa M, Corral NE, Caffaro M et al (2011) Application of a spacer gel to optimize three-dimensional conformal and intensity modulated radiotherapy for prostate cancer. Radiother Oncol 100:436–441
Yang Y, Ford EC, Wu B et al (2013) An overlap-volume-histogram based method for rectal dose prediction and automated treatment planning in the external beam prostate radiotherapy following hydrogel injection. Med Phys 40:011709
Conflict of interest
On behalf of all authors, the corresponding author states that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pinkawa, M., Bornemann, C., Escobar-Corral, N. et al. Treatment planning after hydrogel injection during radiotherapy of prostate cancer. Strahlenther Onkol 189, 796–800 (2013). https://doi.org/10.1007/s00066-013-0388-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-013-0388-0