Abstract
Behavior modification using chemical stimuli is a promising tactic for integrated management of lepidopteran pests. Therefore, this approach was evaluated for the management of the butterfly Opsiphanes cassina Felder 1862, a defoliating pest in oil palm plantations. To achieve this aim, we observed adults’ sexual behavior under laboratory conditions and identified glandular structures in the abdomen and hind wings of males that are associated with the production of sexual pheromones in taxonomically related insects. It was also determined that male calls to females occur at 18:00 h when they fill their glandular structures, tremble, and make abdominal movements while possibly releasing pheromones. The hairpencils of the hind wings are also exposed during this time. Furthermore, the analysis using Gas Chromatography-Mass Spectrometry (GC–MS) on the samples obtained by Solvent Assisted Extraction (SAE) revealed the identification of three male-specific volatile organic compounds (VOCs), including (E)-nerolidol, vanillin, and (Z)-7-heptadecene. The Electroantennographic (EAG) evaluation demonstrated that females respond to the compounds, indicating their likely involvement in intraspecific interactions of the species. However, the behavioral evaluation did not confirm their attractive effect under the conditions employed. Nevertheless, the identified male-specific semiochemicals could be an alternative for inclusion in integrated pest management (IPM) of this pest in oil palm crops. Therefore, it is recommended to evaluate them under field conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The butterfly Opsiphanes cassina Felder 1862 (Lepidoptera: Nymphalidae: Satyrinae) is a significant pest in oil palm cultivation. Its widespread distribution includes South and Central America (Mexzón and Chinchilla 2011; Sierra Márquez et al. 2017; das Chagas 2018). Larvae of this butterfly cause damage to plants by consuming up to 300 cm2 of foliage per individual (Matabanchoy et al. 2018). This factor has a direct impact on the oil palm’s growth as it relies on the continual expansion of its leaf surface. Simultaneously, declining leaf quality reduces the plant’s photosynthetic capability, which ultimately impacts crop productivity (Aldana de La Torre and Aldana de La Torre 2011; Camperos et al. 2020).
Effective and environmentally favorable integrated pest management (IPM) of O. cassina has not yet been achieved (Rodríguez et al. 2012). Therefore, further research is required to create alternative pest management strategies. Among the approaches that promote natural control mechanisms of adult lepidopterans and generate minimal alterations to agricultural ecosystems, behavior modification in response to chemical stimuli, such as pheromones, represents a promising scenario (Schulz 2004; Wyatt 2017; Guo et al. 2020).
Within Lepidoptera, moths and butterflies have undergone various evolutionary pressures that have led to morphological and reproductive behavioral differences, particularly in sexual reproduction (Rutowski 1982; Myers 1972). The shift from a nocturnal to a diurnal lifestyle, and the consequent increased reliance on visual cues for food and mate location, have led many to conclude that butterflies have significantly altered their behavior in response to chemical signals (Liénard et al. 2014). In fact, the majority of female butterflies do not produce the long-range pheromones on which moths almost exclusively depend; instead, mate-seeking is typically accomplished by waiting and/or watching for males to perform stereotypical behaviors (perching, patrolling, lekking) while emitting sex pheromones (Cannon 2020; Wickman 1985; Boggs et al. 2003; i Monteys et al. 2016; Kristensen 2012). These pheromones are involved in closer-range courtship displays that complement visual patterns in many butterfly species and may play a critical role in species recognition and mate choice by females (Vane-Wright and Boppre 1993; Birch et al. 1990). In addition, males have evolved a variety of structures that release volatile organic compounds (VOCs) that can be found throughout the body, including abdominal plates, androconia on the wings, and tufts or hairpencils (Greendfield 2002).
The great chemical diversity of volatile organic compounds (VOCs) that make up male butterfly pheromones, including alkaloid derivatives, terpenoids, aromatic compounds, and carboxylic acids, parallels the existing butterfly diversity (Liénard et al. 2014; Ehlers and Schulz 2022). Although in some cases male butterfly sex pheromone VOCs resemble Type I class sex pheromones of female moths (Schulz 2004; Wang et al. 2014; Nieberding et al. 2008), in several cases they have been suggested or shown to be more similar to those of their host plants (Pliske and Eisner 1969; Nishida et al. 1996; Andersson et al. 2007; Taylor et al. 1986). In fact, recent studies have indicated that certain butterflies can synthesize typical plant compounds (sesquiterpenes) de novo (Schulz et al. 2008; Darragh et al. 2021).
While female pheromones are widely used in lepidopteran IPM, there are considerably fewer studies evaluating the potential of male pheromones in this field (Sasaerila et al. 2003; i Monteys et al. 2012; das Chagas 2018; Hamidi and Frérot 2020). Thus, this study aimed to provide evidence for the mediation of semiochemicals in the sexual behavior of conspecific individuals in O. cassina and to identify candidate structures of the corresponding compounds as a foundation for further developing an integrated pest-management program for this pest in oil palm crops.
Materials and methods
Insects
Opsiphanes cassina prepupae and pupae were collected from commercial oil palm crops (Sepropal Plantation, 4° 16′ 02″ N71° 58′ 58″ W) in Puerto Gaitán, Meta, Colombia. The captures were made during the second semester of 2021 and the first and second semester of 2022. For collecting insects, an individual permit is required to gather wild species of biological diversity for non-commercial scientific research purposes. This permit was issued by the National Environmental License Authority of Colombia (ANLA) under Resolution No. 02431 dated December 24, 2018. First, the insects were collected manually from the palm leaves, mainly from leaves 17 and 25. Next, the prepupae and pupae were taken to the Palmar de las Corocoras Experimental Field laboratories, Paratebueno, Cundinamarca (4° 22′ 5.5″ N73° 10′ 16.9″ W). Some insects were used to establish the breeding, and the others were sent to Bogotá, Colombia. In the laboratory, adults were sexually differentiated by observation; for the separation, the characteristics of sexual dimorphism described for species of the same genus were considered (das Chagas 2018). Subsequently, the adults separated by sex were kept in cages made of muslin cloth and PVC tubes with sugar water as food and controlled conditions (13–22 °C, 46–59% RH, 12:12 h LD regime).
Mating behavior
In the field, O. cassina mating has been observed during scotophase, mainly between 18:00 and 20:00 h. To describe the mating behavior sequence of O. cassina and identify cues in mate location, insects were observed under laboratory conditions during dusk. In each experiment, insects were placed in transparent plastic containers with paper towels at the bottom and sugar water as a food source. The observations were made with natural lighting or red light to simulate natural conditions and not cause interference in the behavior of the insects. The temperature and relative humidity conditions were 13–22 °C and 46–59%, respectively.
Experiment 1: Courtship
Eight pairs of adult insects (eight virgin males and eight virgin females) aged between 24 and 120 h were used. Two groups of adults, aged 24 – 72 h were also used, one with one female and four males and the other with four females and ten males (virgins). This relationship between females and males was selected, taking into account the study of the behavior of a species of the same genus (O. invirae), where groups of insects with at least three more males than females were used. In this study, it was found that males group and fly together before beginning courtship and releasing a pheromone (das Chagas 2018). Observations were made over three days from 17:00 to 5:00 h. In addition, courtship and copulation behaviors presented by males and females were studied, recording the times and referencing the reports for species of the same genus (Srygley 1994).
Experiment 2: calling behavior
Observations were made between 17:00 and 19:00 h. Males in the process of calling were recognized by behaviors such as vibration, movement of the abdomen, and continuous flight exposing the hairpencils of the hindwings (Srygley and Penz 1999; das Chagas 2018). Males of different ages (24, 48, and 72 h) were studied, considering the variables of onset, duration, and number of calls (Andersson et al. 2007; Sandoval 2020). The experimental unit consisted of 1 male, and 19 – 28 males were evaluated by age. The males were placed individually in transparent plastic boxes using water with sugar soaked in cotton as a food source. Data were analyzed following an ANOVA model using the F statistic as a criterion for rejecting the null hypothesis (H0: no significant difference between the means of the groups evaluated).
Volatile collection from O. cassina adults by solvent assisted extraction SAE
The samples of the pheromone glands of males and cuticular sections of females were obtained from virgin adults from 24 to 72 h of age during dusk, from 18:00 – 19:00 h, which coincides with the period of the calling behavior of O. cassina identified previously (das Chagas 2018). Before the extraction, the insects were immobilized by thermal shock in a freezer at − 20 ± 2 °C for 20 min. Then the male glands and cuticular sections of females, on hindwings and abdomen, were excised and immersed in polar (dichloromethane) and nonpolar (hexane) HPLC grade solvents (Sigma-Aldrich, Darmstadt, Germany) for comparison. Each sample was extracted with 5 mL/each solvent for 5 min. The supernatant was filtered through a glass wool column, transferred to glass vials, concentrated to 50 μL with nitrogen and stored in a freezer at − 20 °C for further analysis (Nieberding et al. 2008; das Chagas 2018). Extracts with different numbers of adults (2–30) were obtained to determine how many insects were necessary for an instrumental response.
Analytical procedures
For coupled Gas Chromatography-Mass Spectrometry (GC–MS) analyses, 1 µL of the SAE extracts was injected in an Agilent 7890B GC coupled to a 5977A MS detector (Agilent Technologies, Wilmington, DE, USA). The injection port was maintained at 250 °C in 1:10 Split injection mode. Separation was performed on an HP-5 ((5%phenyl)-methylpolysiloxane) capillary column (30 m × 0.25 mm × 0.25 µm) (Agilent Technologies, Wilmington, DE, USA). The temperature program started at 50 °C, was maintained for five minutes at this temperature, and continued with a ramp of 10 °C/min until reaching 300 °C, keeping this temperature for 5 min (das Chagas 2018). Helium was used as carrier gas at 1.0 mL/min, and the detector temperature was 300 °C. For mass spectrometry analyses, ionization by electronic impact (EI) was used at 70 eV, and the acquisition mode was full scan, with a mass range between 40 − 400 m/z and a scan speed of 1.5 scans/s. Data was processed by Agilent MSD Productivity ChemStation for GC/MS Systems Data Analysis Application.
Lineal retention indexes (LRI) were calculated according to the H. van den Dool and D.J. Kratz modification with a paraffin mixture standard (C8-C40) at a concentration of 25 ppm (Stashenko and Martínez 2010). Mass spectral identification was completed by comparing with either authentic reference standards or spectra from commercial mass spectral databases (NIST Mass Spectral Library version 17). The relative amounts of individual VOCs identified for both sexes were determined based on their area ratio.
Reference compounds
A pure reference standard of vanillin was purchased from Sigma-Aldrich (Taufkirchen, Germany), the nerolidol (1:1 mixture of Z and E isomers) was kindly provided by Dr. Oscar Patiño from Universidad Nacional de Colombia (Bogotá, Colombia), (Z)-7-heptadecene was kindly supplied by Dr. Jocelyn Millar from University of California (Riverside, USA), and n-alkane mixture (C8-C40) was acquired from Merck (Darmstadt, Germany).
Evaluation of O. cassina adults response
The electrophysiological and behavioral responses of O. cassina adult females to the different olfactory stimuli were evaluated in the Entomology Laboratory of the Tibaitatá Research Center in Agrosavia—Colombian Agricultural Research Corporation. The methodology used for each case is described below.
Electroantennographic EAG studies
The antennae of O. cassina females (9 to 10 days old) were carefully removed using micro-scissors to evaluate the electrophysiological response. A Syntech brand electroantennographic detector and a silver fork-type electrode were used to perform the EAG measurements. The base of the antenna was connected to the reference electrode while the tip was connected to the working electrode, and an electrically conductive gel (Sigma Gel, Parker Laboratories, Fairfield, NJ, United States) was used to cover the ends on each electrode (das Chagas 2018). Rectangles of filter paper (0.5 × 2.0 cm) with 10 uL of the treatments were subsequently inserted into a glass Pasteur pipette. As olfactory stimuli for the EAG tests, the SAE samples were selected in addition to the individual compounds (1 μg/μL). This pipette was connected to the pulse generator system (StimulusController, Type CS-55, Syntech), where the “puff” pulse that sent the stimulus was maintained for 0.5 s, with a continuous main stream of humidified air (600 mL/min), using an electrically controlled airflow generator (CS-05, Syntech, Germany). The EAG signal recorded with the electrodes was amplified with a high input impedance preamplifier (Syntech, Hilversum, The Netherlands). Then it was analyzed by a PC-based signal processing system (IDAC-2, Syntech, Germany) and software (GC-EAD 2014 v1.2.5, Syntech, Germany) (Malo et al. 2000). To determine how long it was possible to use the live antenna, the change in the depolarization of the antennas was evaluated at 5–min intervals for 20 min, using the male-specific component identified as the majority (nerolidol) as the stimulus (Malo et al. 2000). Subsequently, the samples were evaluated on antennae of O. cassina females, where each tested antenna received stimuli from all treatments sequentially randomizing the order, leaving at least 30 s between successive stimulations. The average of the control (solvents) responses was subtracted from that of the stimuli, and the depolarization means were compared for significance using ANOVA followed by a one-tailed Paired Sample T-Test (i Monteys et al. 2016).
Wind tunnel experiments
The behavioral responses of O. cassina females to the different olfactory stimuli were evaluated using an FDR 55 Fischbach Luft- und Ventilatorentechnik GmbH (Neunkirchen, Germany) wind tunnel. The equipment consisted of an odorless acrylic tunnel (200 cm long × 65 cm wide × 65 cm high) coupled with a filter, ventilation, and extraction system. The airflow was produced by a suction pump and purified through an activated carbon filter. The speed of the airflow was fixed through an anemometer. The flight chamber was separated from the windward section by fine wire mesh with additional wire mesh at the other end. The olfactory stimuli were placed inside the wind tunnel in front of the air inlet end, while the receptor insects were placed at the end of the tunnel. When males were used as a stimulus (positive control), they were placed in the pre-chamber of the tunnel. The tests were carried out under controlled conditions of temperature (24 ± 2 °C), relative humidity (68 ± 5%), photoperiod (12:12 h LD regime), and airflow (50–55 cm/s). The insects to be evaluated were kept in an odor–free room under the previously described conditions, using sugar water as a food source. Two hours before each bioassay, they were placed in plastic boxes and deprived of food. Bioassays were performed during the first 2 – 5 h of scotophase. An attenuated red light was used to observe the experiments to not interfere with the insects’ behavior. In each test, a square of filter paper (3 cm × 3 cm) was used where the aliquot of the solutions of the compounds to be evaluated was placed. The three VOCs identified as male-specific (nerolidol (mixture of E and Z isomers), vanillin, and (Z)-7-heptadecene) at different doses (10–1000 μg) were evaluated. Before starting the test, the papers impregnated with the solutions of the compounds were allowed to evaporate for 10 s. In each evaluation, the insects were observed for a maximum of 15 min. Receptor females were between 1 and 11 days old; a total of 83 individuals were evaluated (between 5 and 7 per stimulus). Observations were categorized into four behavioral responses: (1) Do not move from the starting area; (2) Taking flight from the departure area; (3) Upwind flight toward the source of the olfactory stimulus; (4) Getting contact with the source of the olfactory stimulus (Eiras 2000; Fouad et al. 2013; Lu et al. 2022).
Results and discussion
Courtship behavior
Structures associated with chemical signal production and dissemination were identified in males (androconial glands), in addition to sexual dimorphism reported for the genus (das Chagas 2018). Two dark yellow secretory plates are located on the fourth and fifth abdominal segments, and the hindwings contain groups of hairpencils or tufts that are thought to disseminate pheromones (see Fig. 1). These features, as reported in other species of the genus Opsiphanes and the family Nymphalidae (Darragh et al. 2017; das Chagas 2018), suggest that the males of this species produce and/or emit VOCs that regulate courtship and copulation.
For couples and groups, there was a high level of activity during twilight (18:00 h), with males approaching females and initiating a stereotypical behavior pattern. This behavior was repeated consistently in all tests and included thorax and abdomen vibration, filling of glandular plates, wing movement, hair pencils display towards the female, and the release of a sweet aroma detectable by humans. Nonetheless, copulation was not observed (Fig. 2).
Similar courtship behavior has been observed in O. invirae, O. tamarindi, Caligo memmon, and C. eurilochus, all of the same genus or family as O. cassina, in which the male vibrates and moves his abdomen after filling his glandular structures to attract and copulate with the female (Benson et al. 1997; Penz & Williams 2020). Additionally, most butterflies in the Brassolini tribe (Lepidoptera: Nymphalidae: Satyrinae) have been reported to be active at twilight, with mating and oviposition occurring at dawn, dusk, or both, depending on the species (Benson et al. 1997; Penz and Williams 2020). Likewise, in some species of the genus Opsiphanes, low twilight temperatures have been found to cause males to tremble or vibrate to increase thoracic temperature in preparation for reproductive behavior (Srygley 1994). Similarly, studies in Panama and Brazil found that adult Opsiphanes males perform aerial displays using rapid flight, and some species form mating aggregations (Srygley 1994; das Chagas 2018; Penz and Williams 2020; Crees et al. 2021).
Although mating is typically considered an essential activity for the adult stage of most insects, as it provides an opportunity to spread genes to the next generation (Cannon 2020), some studies have reported that some females remain unmated even under field conditions (Rhainds 2010). For Lepidoptera, research suggests that various factors including population density, flight area, distance from host plants, and their position on the plants influence female response to chemical or visual stimuli emitted by males, ultimately impacting the species’ reproductive success (Shapiro 1970; Kvedaras et al. 2000; Solensky 2004; Cameron et al. 2005). In addition, the absence of copulation among adults may be linked to the altered environmental conditions in the laboratory as compared to collection sites (Wellington 1946; Mbata 1986; Royer and McNeil 1993; Pellegrino et al. 2013; Wu et al. 2014; Zeng et al. 2018; Miao et al. 2021). Temperature and humidity are significant environmental factors that affect the growth and development of insects, including generation time, body size, sexual behaviors, and voltinism (Mbata 1986; Wu et al. 2014; Zeng et al. 2018). Additionally, these conditions impact the insect’s ability to detect food sources or pheromones produced by their sexual partner (Royer and McNeil 1993). Furthermore, there is evidence that insects adjust their flight, mating, and foraging behaviors based on changes in barometric pressure (Wellington 1946). These conditions can affect the beginning or length of copulation and possibly even decrease occurrence percentages, especially during mating (Pellegrino et al. 2013; Miao et al. 2021).
Considering the above, although the emitting insect can produce the chemical signal in the ideal concentration and proportion, variables such as temperature, humidity, and barometric pressure, among others, can significantly impact the receiving insect, hindering successful copulation. Therefore, the next stage of O. cassina behavioral study focused on determining the calling time by the males, followed by the extraction of VOCs during the same period.
Calling behavior of O. cassina males
Reproductive success may be viewed as a primary driver of sexual behavior pattern evolution among animals, specifically those in the Insecta class (Rutowski 1982; Baker 1983; Srygley 1994; Stepien et al. 2020). For mating and reproduction to occur, communication across long distances must be achieved. The attraction and location of a suitable sexual partner are crucial events, as inaccuracies in partner finding would lead to prolonged search times, higher energy expenditure, greater predation risk, and fewer viable offspring (Cardé and Baker 1984; Cardé and Millar 2004; Saveer et al. 2014). In most species of Lepidoptera, this issue is settled through the release and detection of sexual pheromones (Wyatt 2017; Guo et al. 2020). In this scenario, it is important to note that the emission of sexual pheromones is linked to stereotyped behaviors in what is known as "calling behavior." The organism releasing the chemical signal makes movements or emits sounds to aid in the dissemination of the pheromone components and to attract the attention of the receiving organism during a specific time period (McNeil 1991; Fitzpatrick and McNeil 1988; Lee et al. 2014; Dion et al. 2019).
The calling behavior of O. cassina males was initiated at 18:00 h for adults aged 24, 48, and 72 h. For 24-h-old adults, the onset began 10 min later than for the other two ages (refer to Fig. 3). However, there were no significant differences found among the three ages assessed for the calling onset variable (p value = 0.06, 95% confidence) (see Fig. 4). Additionally, it was observed that the male does not give the call continuously. Rather, it vibrates, flies, and exposes the hairpencils with pauses. On average, five calls are given with a total duration of five minutes. Additionally, no significant differences were found in the variables of number of calls (p value = 0.26, 95% confidence) and duration of call (p value = 0.08, 95% confidence) among the three ages evaluated (see Fig. 4). Therefore, the results indicate that the removal of sexual glands from virgin males aged 24, 48, and 72 h is feasible during the assessed hours around twilight.
Extraction and identification of VOCs released by O. cassina adults
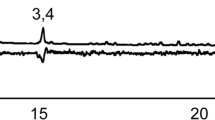
The GC–MS analysis of the samples obtained by SAE allowed the identification of six VOCs (see Table 1). Comparison of the results acquired with the two solvents used (Fig. 5) revealed the presence of (E)-β-farnesene 1, (E)-nerolidol 3, (Z)-7-heptadecene 4, palmitic acid 5, and oleic acid 6 in both the wings and abdomen extracts. Notably, male-specific compounds 1 and 4 were only detected in wings. On the other hand, the dichloromethane extracts only contain vanillin 2 and VOC 3 in male samples, both in their wings and abdomen. It is noteworthy that compound 2 is solely extracted with this solvent. The comparison of the profiles of the two solvents reveals a substantial difference in the number of components detected. Hexane samples detect a significantly higher number of components than dichloromethane samples, attributed to the nonpolar nature of most of the VOCs extracted.
Representative total ion chromatograms (TIC) of VOCs detected by SAE with hexane (left) and dichloromethane (right) in wing samples from male (♂) and female (♀) Opsiphanes cassina adults on an HP-5 column. The numbers correspond to those in Table 1
Palmitic and oleic acids were tentatively identified as compounds 5 and 6 respectively, detected in both male and female samples. These VOCs have been reported as pheromones and allelochemicals for various species of Lepidoptera. In some cases, both fatty acids have been found simultaneously as semiochemicals. Oleic acid is a constituent of the sex pheromone mixture produced by female Telchin licus Drury, 1773 (= Castnia licus) (Lepidoptera: Castniidae) according to Rebouças et al. (1999). Additionally, Phelan et al. (1991) have reported it as a kairomone in almond, pistachio, and walnut trees, attracting and stimulating the oviposition of the pest-insect Amyelois transitella Walker 1863 (Lepidoptera: Pyralidae). Palmitic acid has been found in extracts of androconial glands from males of seven species of the Amauris genus (Lepidoptera: Danaidae) in their abdomen, wings, and hairpencils, indicating its potential role as a sex pheromone (Schulz et al. 1993, 1988). In the case of Eldana saccharina Walker, 1865 (Lepidoptera: Pyralidae), both palmitic and oleic acid have been identified as constituents of the sex pheromone mixture produced by males (Rebouças et al. 1999). In a prior study, the bee moth Acherontia atropos Linnaeus, 1758 (Lepidoptera: Sphingidae) was found to utilize oleic and palmitic acid as allomones for camouflage and evade detection by worker bees when infiltrating honeycombs (Moritz et al. 1991). In the case of O. cassina, these two VOCs may act as allelochemicals facilitating communication with other species in the oil palm crop since they are present in samples from both males and females. Nevertheless, it should be noted that many insects incorporate such substances as free fatty acids, using them as precursors for other semiochemicals or merely storing them as triacylglycerols (Blaul et al. 2014; Schulz 2004; Municio et al. 1975).
Regarding the male-specific VOCs, three compounds were detectable and identifiable, possibly making up part of the sex pheromone of O. cassina: vanillin 2, (E)-nerolidol 3 (major compound), and (Z)-7-heptadecene 4. Additionally, (E)-β-farnesene 1 was detected as a male-specific VOC, but it was tentatively identified in the present study. The four compounds mentioned above have been reported to occur naturally in plants or animals.
(E)-Nerolidol 3 is a sesquiterpene present in the essential oils of several aromatic plants and displays a diverse range of biological activities (Chan et al. 2016). Four plant hosts (Lycopersicon esculentum, Capsicum annuum, Datura wrightii, Proboscidea parviflora) of Manduca sexta (Linnaeus, 1763) (Lepidoptera: Sphingidae) have presented it as a kairomone (Fraser et al. 2003). Regarding reports of lepidopteran pheromones, a study was conducted on the species O. invirae, which is a pest insect of the oil palm. The study found that (E)-nerolidol is one of the volatile organic compounds that make up the sexual pheromone produced by the male. This was determined through electroantennography tests, which detected response in the females’ antennae. Furthermore, field tests have shown that it effectively captures adults when used in combination with food baits (das Chagas 2018). It is important to note that (E)-nerolidol has two stereoisomers. Further analysis by GC on a chiral phase is required to determine which of them (if not both) is emitted by O. cassina males (Tabacchi et al. 2010).
For another palm pest, the species Paysandisia archon (Burmeister, 1880) (Lepidoptera, Castniidae), male-specific VOCs including sesquiterpenes were identified as its pheromone. The study of the P. archon pheromone identified three potential male-specific VOCs: (Z, E)-farnesal, the corresponding E, E isomer, and (E, Z)-2,13-octadecadienol, which produced significant GC-EAG responses in female antennae (i Monteys et al. 2012). Farnesals were only detected on the forewings and hindwings of males and solely elicited responses in the antennae of females. For its part, dienol was detected in glandular sections in the legs of males, but it triggered significant responses in both female and male antennae (i Monteys et al. 2012). In field evaluations, it was observed that virgin males exhibit a scratching behavior while releasing a chemical signal that attracts virgin females from long distances (Hamidi and Frérot 2020).
Vanillin 2 (4-hydroxy-3-methoxybenzaldehyde) is a crucial molecule not only in the flavor and aroma industry but also because of its biogenetic relationship with the phenylpropanoid pathway in plants and other essential physiological molecules (Walton et al. 2003). Furthermore, it has been reported to have activity as an insect attractant for orders Coleoptera, Heteroptera, Hymenoptera, and Lepidoptera. Although the structure of this compound departs from the biosynthesis routes reported for the production of volatile organic compounds by Lepidoptera, it has been identified as a pheromone for certain species of the Pyralidae (Lepidoptera) family, including Tirathaba mundella Walker, 1864, an insect that also damages oil palms. Vanillin, along with (3S,6S)-2,2,6-trimethyl-6-vinyltetrahydropyran-3-ol, was detected in samples taken directly from the male wings. In field assays, the compounds produced significant female attraction (Sasaerila et al. 2003).
The structure of (Z)-7-heptadecene 4 is similar to a type I Lepidoptera pheromone (Yew and Chung 2015). It is suggested that this compound is generated through the biosynthesis of saturated fatty acids from acetyl coenzyme A, with subsequent chain modification via desaturation and decarboxylation (Tillman et al. 1999; Foster and Anderson 2018). (Z)-7-heptadecene has been identified as a component of the volatile profile of certain marine algae species and as a pheromone of select mite species (Sugisawa et al. 1990; Sakata et al. 1995). Furthermore, it has been documented as a male-specific volatile organic compound in the study of the sex pheromone of O. invirae, a pest species that was previously mentioned to be present in oil palm plantations. When evaluated by EAG, the compound elicited a positive response in the antennae of females and resulted in high adult captures in the field when used in combination with food baits (das Chagas 2018).
For its part, with respect to reports on (E)-β-farnesene 1, this sesquiterpene has been found in the secretions of lepidopteran larvae of the Papilionidae family, acting as a defense mechanism against predators (Eisner et al. 1970). It has also been identified as a kairomone of the codling moth Cydia pomonella Linnaeus, 1758 (Lepidoptera: Tortricidae), and wind tunnel tests have shown that it enhances the response of males to the pheromone produced by the female (Schmera and Guerin 2012). Additionally, this VOC was identified as a constituent of the male-specific sex pheromone of O. invirae along with (E)-nerolidol and (Z)-7-heptadecene (das Chagas 2018).
Since compounds that differ from typical VOCs characteristics reported for the same taxonomic order have been found in insect emissions and are closer to the chemical profile of the host plants, there are ongoing discussions about the potential for insects to use plant-derived compounds as pheromone sources. It is reported that there are three ways in which diet can contribute to quantitative or qualitative differences in a chemical signal (Raoul et al. 2017). First, diet quality and the accumulation of macro or micronutrients can influence pheromone production (Kopena et al. 2011; South et al. 2011). Secondly, pheromone components that cannot be fully synthesized endogenously must be acquired from dietary sources and then either transformed or directly incorporated into the pheromone (Blaul et al. 2014; Darragh et al. 2019). Additionally, the diet can have a direct impact on the pheromone or signature blend profile, determining the VOCs present and their relative relationships. This can have consequences for recognition systems that rely on phenotype matching or self-reference mechanisms (Buczkowski et al. 2005). In the case of O. cassina VOCs, it is possible that vanillin is derived from the plant, as there are no studies showing that Lepidoptera can produce phenolic aldehydes. However, there is a possibility of endogenous biosynthesis for sesquiterpenes (Schulz et al. 2008; Darragh et al. 2021). In the study of the butterfly Heliconius melpomene (Lepidoptera: Nymphalidae) (Linnaeus, 1758), the terpene (E)-β-ocimene was found to be an anti-aphrodisiac pheromone. In the first approach, it was observed that males fed with labeled D-13C6-glucose produced 13C-labeled ocimene (Schulz et al. 2008). Glucose can be converted by butterflies through the citric acid cycle into doubly labeled acetate. This acetate can then enter the mevalonate pathway of terpene synthesis. Along with two additional unlabeled acetate units, which are present in excess in experimental butterflies, it gets converted into isopentenyl pyrophosphate building blocks containing one or two 13C atoms (Barton et al. 1999). In a later work, through linkage mapping, gene expression and functional analyses two novel terpene synthases (TPSs) were identified, one of which was able to synthesize (E)-β-ocimene in vitro. Notably, these TPSs are unrelated to previously described plant and insect TPSs, demonstrating that chemical convergence has independent evolutionary origins (Darragh et al. 2021).
EAG studies of O. cassina adults
The electrical reactions of the dendrites in olfactory cells have a negative potential that is equivalent to the depolarization of the receptor membrane. The amplitude of the response recorded in EAG, which correlates with the frequency of nerve impulses generated, increases with higher concentrations of chemical stimulus, and sensitivity is observed until saturation occurs (Roelofs 1984). To determine the length of time the O. cassina antennae could be used, we evaluated the response to the major VOC because preliminary tests showed higher depolarization in female antennae (Fig. 6). After evaluating the antennae’s response for 20 min, it was concluded that they remain viable for up to 10 min post-extraction as there were no significant differences in the recorded responses at 0, 5, and 10 min for the four evaluated antennae (see Fig. 7).
EAG response time of Opsiphanes cassina adult females to the major male-specific VOC (A = antenna, n = 4) (left) and mean of EAG responses at 0, 5, and 10 min (right). ANOVA results indicate that there are no significant differences between the means of the EAG responses for the three times evaluated with a confidence of 95% (p value = 0.92)
When evaluating the different treatments, it was found that the antennae of O. cassina females were stimulated both by the cuticular extracts and by the individual male-specific compounds (Fig. 8), which suggests that nerolidol, vanillin, and (Z)-7-heptadecene could act as semiochemicals detected by females.
Wind tunnel experiments
The behavioral evaluation results of O. cassina females for the three male-specific VOCs are presented in Table 2, displaying the percentage of females that responded in accordance with the four established behavioral categories. It was observed under the evaluated conditions that there was no response indicating attraction of females to the stimuli tested at the concentrations used, nor to the males’ call. Instead, the insects remained in or flew away from the departure zone without upwind flight towards the source of the olfactory stimulus.
The results obtained can be explained by the fact that, in butterflies, the male’s search for a mate does not always result in copulation, as it ultimately depends on the female’s preference (receptivity and choice). Male pheromones provide females with a wealth of information that they can use to determine their age, health, and sexual maturity, and thus their attractiveness. This information allows females to choose from a pool of available suitors. Therefore, to ensure a successful courtship, the female must positively respond to the chemical signals that the male emits when making the call (Cannon 2020). Additionally, while changes in concentration did not show significant differences in results, this variable still has a considerable impact. The stimulus must surpass the threshold necessary for the receiving insect to detect it and orient itself towards it (Cardé 2021). However, the response may increase proportionally with concentration until the saturation point of the olfactory receptors is reached, resulting in a lack of targeted response (Conchou et al. 2021; Renou and Anton 2020).
Additionally, the insect’s ability to locate food sources or detect pheromones produced by potential mates is significantly impacted by changes in environmental conditions. Despite adjusting the temperature and relative humidity to match their natural ecological requirements, the barometric pressure remained noticeably different. On the other hand, it is important to note that sexual behavior in Lepidoptera generally follows a distinct circadian rhythm that is influenced by the photoperiod (Kamimura and Tatsuki 1994). Changes in this factor can affect various aspects of the insect’s behavior, such as calling behavior, pheromone production, and response to stimuli (Gemeno and Haynes 2001). In the case of crepuscular insects like O. cassina, it is important to note that while ambient light levels during this time are at least two orders of magnitude lower than during the day, the presence of available light during call-driven courtship displays can still have an effect (Cannon 2020). The wind tunnel evaluation did not account for this condition, which may have influenced the observed response.
Since the proportion of VOCs in the pheromone mixture has not yet been established, the evaluation of the synergistic effect on the insect response was not conducted. However, research is currently underway to determine the proportion and concentration of pheromone components, as well as their evaluation under both wind tunnel and field conditions (Hamidi and Frérot 2020).
The interaction between plants and insects is a useful model for comprehending the organizational and functional complexity of biological systems at various levels of hierarchy. This is because there exists a wealth of information and research at both the molecular and ecosystem levels (Barah and Bones 2015). Among them, investigating the chemical signals of insects that feed on plants is fascinating because many herbivores use secondary metabolites (allelochemicals) that plants produce as anti-herbivorous defenses to regulate behaviors as important as mating (Zvereva and Kozlov 2016). This capacity of insects to exploit plant defenses (sequestered or synthesized de novo) for their own benefit is among the most captivating instances of trophic interactions in nature. It exemplifies how adaptability sparks coevolutionary progress (Beran and Petschenka 2022). In this scenario, it is important to note that sexual signaling in these arthropods demonstrates high phenotypic plasticity. This indicates that semiochemical-mediated behaviors are susceptible to changes in environmental conditions, including biotic and abiotic factors, as well as changes in geographic location or population density, resulting in interspecific variations (Groot et al. 2009; De Pasqual et al. 2021). Despite this, even when exogenous variables strongly influence it, the mixture that constitutes a pheromone is specific to the species in a given population, making it a significant force in reproductive isolation and speciation. Therefore, identifying the chemical structures and composition of pheromone mixtures is fundamental to understanding chemical communication and its role in the evolution of ecology (Nojima et al. 2018).
For the first time, this study examined sexual behavior and characterized the VOCs released by O. cassina adults. Three male-specific compounds were identified, specifically (E)-nerolidol, vanillin, and (Z)-7-heptadecene, which are part of the calling behavior during the twilight period. The EAG evaluation confirmed that these three compounds elicit an electrophysiological response in the female antennae, indicating their role in modulating the sexual behavior of the species. Our findings suggest that the assessed compounds that are unique to male attractiveness act as semiochemicals, leading to the infestation of oil palm crops by O. cassina. This vital knowledge serves as a cornerstone for designing an effective IPM plan to handle this pest in the oil palm.
Data availability
All data are available to requests to the corresponding author.
References
Aldana de La Torre RC, Aldana de La Torre JA (2011) Guía para el reconocimiento y manejo de insectos defoliadores y asociados a la Pestalotiopsis. Tecnologías para la agroindustria de la palma de aceite: guía para facilitadores. Centro de Investigación en Palma de Aceite – Cenipalma, Colombia
Andersson J, Borg-Karlson AK, Vongvanich N, Wiklund C (2007) Male sex pheromone release and female mate choice in a butterfly. J Exp Biol 210:964–970. https://doi.org/10.1242/jeb.02726
Baker R (1983) Insect territoriality. Annu Rev Entomol 28:65–89. https://doi.org/10.1146/annurev.en.28.010183.000433
Barah P, Bones AM (2015) Multidimensional approaches for studying plant defense against insects: from ecology to omics and synthetic biology. J Exp Bot 66:479–493. https://doi.org/10.1093/jxb/eru489
Barton D, Nakanishi K, Meth-Cohn O (1999) Comprehensive natural products chemistry, vol 2. Elsevier, Amsterdam
Benson WW, Marini-filho OJ, De CRM (1997) Territoriality by the dawn’s early light: the Neotropical owl butterfly Caligo idomenaeus (Nymphalidae: Brassolinae). J Res Lepid 34:14–20
Beran F, Petschenka G (2022) Sequestration of plant defense compounds by insects: from mechanisms to insect–plant coevolution. Annu Rev Entomol 67:163–180. https://doi.org/10.1146/annurev-ento-062821-062319
Birch MC, Poppy GM, Baker TC (1990) Scents and eversible scent structures of male moths. Annu Rev Entomol 35:25–58. https://doi.org/10.1146/annurev.en.35.010190.000325
Blaul B, Steinbauer R, Merkl P et al (2014) Oleic acid is a precursor of linoleic acid and the male sex pheromone in Nasonia vitripennis. Insect Biochem Mol Biol 51:33–40. https://doi.org/10.1016/j.ibmb.2014.05.007
Boggs CL, Watt WB, Ehrlich PR (2003) Butterflies: ecology and evolution taking flight. University of Chicago Press, Chicago
Buczkowski G, Kumar R, Suib SL, Silverman J (2005) Diet-related modification of cuticular hydrocarbon profiles of the argentine ant, Linepithema humile, diminishes intercolony aggression. J Chem Ecol 31:829–843. https://doi.org/10.1007/s10886-005-3547-7
Cameron PJ, Wallace AR, Madhusudhan VV et al (2005) Mating frequency in dispersing potato tuber moth, Phthorimaea operculella, and its influence on the design of refugia to manage resistance in Bt transgenic crops. Entomol Exp Appl 115:323–332. https://doi.org/10.1111/j.1570-7458.2005.00256.x
Camperos J, Barrera E, Hernandez J et al (2020) Propuesta metodológica para estimar la defoliación en la palma de aceite. XV Reunión Técnica Nacional de Palma de Aceite. https://www.researchgate.net/publication/351563403_Propuesta_metodologica_para_estimar_la_defoliacion_en_la_palma_de_aceite. Accessed 15 Mar 2022
Cannon RJC (2020) Courtship and mating in butterflies. CABI, Boston
Cardé RT (2021) Navigation along windborne plumes of pheromone and resource-linked odors. Annu Rev Entomol 66:317–336. https://doi.org/10.1146/annurev-ento-011019-024932
Cardé RT, Baker TC (1984) Sexual communication with pheromones. Chemical ecology of insects. Springer, Boston, pp 355–383
Cardé RT, Millar JG (2004) Advances in insect chemical ecology. Cambridge University Press, Cambridge
Chan WK, Tan LTH, Chan KG et al (2016) Nerolidol: a sesquiterpene alcohol with multi-faceted pharmacological and biological activities. Molecules 21:529. https://doi.org/10.3390/molecules21050529
Conchou L, Lucas P, Deisig N et al (2021) Effects of multi-component backgrounds of volatile plant compounds on moth pheromone perception. InSects 12:409. https://doi.org/10.3390/insects12050409
Crees LD, Devries P, Penz CM (2021) Do Hind Wing Eyespots of Caligo Butterflies Function in Both Mating Behavior and Antipredator Defense? (Lepidoptera, Nymphalidae). Ann Entomol Soc Am 114:329–337. https://doi.org/10.1093/aesa/saaa050
Darragh K, Vanjari S, Mann F et al (2017) Male sex pheromone components in Heliconius butterflies released by the androconia affect female choice. bioRxiv 10:033506. https://doi.org/10.1101/033506
Darragh K, Byers KJRP, Merrill RM et al (2019) Male pheromone composition depends on larval but not adult diet in Heliconius melpomene. Ecol Entomol 44:397–405. https://doi.org/10.1111/een.12716
Darragh K, Orteu A, Black D et al (2021) A novel terpene synthase controls differences in anti-aphrodisiac pheromone production between closely related Heliconius butterflies. PLoS Biol 19:e3001022. https://doi.org/10.1371/JOURNAL.PBIO.3001022
das Chagas K (2018) Estudo do feromônio de Opsiphanes invirae (Hubner, 1818) (Lepidoptera: Nymphalidae). Dissertation. Universidade Federal de Alagoas
De Pasqual C, Groot AT, Mappes J, Burdfield-Steel E (2021) Evolutionary importance of intraspecific variation in sex pheromones. Trends Ecol Evol 36:848–859. https://doi.org/10.1016/j.tree.2021.05.005
Dion E, Monteiro A, Nieberding CM (2019) The role of learning on insect and spider sexual behaviors, sexual trait evolution, and speciation. Front Ecol Evol 6:225. https://doi.org/10.3389/fevo.2018.00225
Ehlers S, Schulz S (2022) The scent chemistry of butterflies. Nat Prod Rep 40:794–818
Eiras ÁE (2000) Calling behaviour and evaluation of sex pheromone glands extract of Neoleucinodes elegantalis Guenée (Lepidoptera: Crambidae) in wind tunnel. An Da Soc Entomológica Do Bras 29:453–460. https://doi.org/10.1590/S0301-80592000000300007
Eisner T, Pliske TE, Ikeda M et al (1970) Defense mechanisms of arthropods. XXVII. osmeterial secretions of papilionid caterpillars (Baronia, Papilio, Eurytides). Ann Entomol Soc Am 63:914–915. https://doi.org/10.1093/aesa/63.3.914
Fitzpatrick SM, McNeil JN (1988) Male scent in lepidopteran communication: the role of male pheromone in mating behaviour of Pseudaletia unipuncta (Haw.) (Lepidoptera: Noctuidae). Mem Entomol Soc Canada 120:131–151. https://doi.org/10.4039/ENTM120146131-1
Foster SP, Anderson KG (2018) Differential pheromone sampling of the gland of female Heliothis Virescens moths reveals glandular differences in composition and quantity. J Chem Ecol 44:452–462. https://doi.org/10.1007/s10886-018-0954-0
Fouad HA, Faroni LRDA, Vilela EF, de Lima ER (2013) Flight responses of Sitotroga cerealella (Lepidoptera: Gelechiidae) to corn kernel volatiles in a wind tunnel. Arthropod Plant Interact 7:651–658. https://doi.org/10.1007/s11829-013-9275-y
Fraser AM, Mechaber WL, Hildebrand JG (2003) Electroantennographic and behavioral responses of the sphinx moth Manduca sexta to host plant headspace volatiles. J Chem Ecol 29:1813–1833. https://doi.org/10.1023/A:1024898127549
Gemeno C, Haynes KF (2001) Impact of photoperiod on the sexual behavior of the black cutworm moth (Lepidoptera: Noctuidae). Environ Entomol 30:189–195. https://doi.org/10.1603/0046-225X-30.2.189
Greendfield M (2002) Signalers and receiver: mechanisms and evolution of arthropod communication. Oxford University Press, Oxford
Groot AT, Inglis O, Bowdridge S et al (2009) Geographic and temporal variation in moth chemical communication. Evolution 63:1987–2003. https://doi.org/10.1111/j.1558-5646.2009.00702.x
Guo JM, Liu XL, Liu SR et al (2020) Functional characterization of sex pheromone receptors in the fall armyworm (Spodoptera frugiperda). InSects 11:193. https://doi.org/10.3390/insects11030193
Hamidi R, Frérot B (2020) Evidence for long-range mate attraction mediated by a male-produced sex pheromone in Paysandisia archon (Lepidoptera: Castniidae). J Insect Behav 332(33):97–104. https://doi.org/10.1007/S10905-020-09752-Y
i Monteys V, Acín P, Rosell G et al (2012) Moths behaving like butterflies. Evolutionary loss of long range attractant pheromones in castniid moths: a Paysandisia archon Model. PLoS ONE 7:e29282. https://doi.org/10.1371/journal.pone.0029282
i Monteys V, Quero C, Santa-Cruz MC et al (2016) Sexual communication in day-flying Lepidoptera with special reference to castniids or “butterfly-moths.” Bull Entomol Res 106:421–431. https://doi.org/10.1017/S0007485316000158
Kamimura M, Tatsuki S (1994) Effects of photoperiodic changes on calling behavior and pheromone production in the Oriental tobacco budworm moth, Helicoverpa assulta (Lepidoptera: Noctuidae). J Insect Physiol 40:731–734. https://doi.org/10.1016/0022-1910(94)90101-5
Kopena R, Martín J, López P, Herczeg G (2011) Vitamin E supplementation increases the attractiveness of males’ scent for female European green lizards. PLoS ONE 6:e19410. https://doi.org/10.1371/journal.pone.0019410
Kristensen NP (2012) Lepidoptera, moths and butterflies, vol 2. Morphology, physiology, and development. Walter de Gruyter, Berlin
Kvedaras OL, Gregg PC, Del Socorro AP (2000) Techniques used to determine the mating behaviour of Helicoverpa armigera (Hubner) (Lepidoptera: Noctuidae) in relation to host plants. Aust J Entomol 39:188–194. https://doi.org/10.1046/j.1440-6055.2000.00156.x
Lee MS, Albajes R, Eizagurre M (2014) Mating behaviour of female Tuta absoluta (Lepidoptera: Gelechiidae): polyandry increases reproductive output. J Pest Sci 873(87):429–439. https://doi.org/10.1007/s10340-014-0576-4
Liénard MA, Wang HL, Lassance JM, Löfstedt C (2014) Sex pheromone biosynthetic pathways are conserved between moths and the butterfly Bicyclus anynana. Nat Commun. https://doi.org/10.1038/ncomms4957
Lu H, Yang Y-Q, Yu J et al (2022) (Z, Z, Z)-3,6,9-nonadecadiene, a potential inhibitor of sex pheromone of Ectropis grisescens Warren (Lepidoptera: Geometridae): electroantennogram test, wind tunnel, and in silico study. bioRxiv. https://doi.org/10.1101/2022.11.02.514740
Malo EA, Renou M, Guerrero A (2000) Analytical studies of Spodoptera littoralis sex pheromone components by electroantennography and coupled gas chromatography-electroantennographic detection. Talanta 52:525–532. https://doi.org/10.1016/S0039-9140(00)00401-X
Matabanchoy JA, Raigoza D, Bustillo AE (2018) Biología de Opsiphanes cassina defoliador palma de aceite en la Zona Suroccidental palmera de Colombia. XIX Conferencia Internacional sobre Palma de Aceite. https://www.researchgate.net/publication/340794229_Biologia_de_Opsiphanes_cassina_defoliador_palma_de_aceite_en_Colombia. Accessed 21 Jun 2022
Mbata GN (1986) Combined effect of temperature and relative humidity on mating activities and commencement of oviposition in Plodia interpunctella (Hubner) (Lepidoptera: Phycitidae). Int J Trop Insect Sci 7:623–628. https://doi.org/10.1017/S1742758400011553
McNeil JN (1991) Behavioral Ecology of Pheromone-Mediated Communication in Moths and Its Importance in the Use of Pheromone Traps. Annu Rev Entomol 36:407–430. https://doi.org/10.1146/annurev.en.36.010191.002203
Mexzón RG, Chinchilla CM (2011) Opsiphanes cassina Felder (Lepidoptera: Nymphalidae), defoliator of the oil palm (Elaeis guineensis Jacquin) in Central America. ASD Oil Palm Pap 36:14–33
Miao J, Guo P, Li H et al (2021) Low barometric pressure enhances tethered-flight performance and reproductive of the oriental armyworm, Mythimna separata (Lepidoptera: Noctuidae). J Econ Entomol 114:620–626. https://doi.org/10.1093/jee/toaa291
Moritz RFA, Kirchner WH, Crewe RM (1991) Chemical camouflage of the death’s head hawkmoth (Acherontia atropos L.) in honeybee colonies. Naturwissenschaften 78:179–182. https://doi.org/10.1007/BF01136209
Municio AM, Odriozola JM, Pérez-Albarsanz MA (1975) Biochemistry of development in insects: incorporation of fatty acids into different lipid classes. Eur J Biochem 60:123–128. https://doi.org/10.1111/j.1432-1033.1975.tb20983.x
Myers J (1972) Pheromones and courtship behavior in butterflies. Integr Comp Biol 12:545–551. https://doi.org/10.1093/icb/12.3.545
Nieberding CM, de Vos H, Schneider MV et al (2008) The male sex pheromone of the butterfly Bicyclus anynana: towards an evolutionary analysis. PLoS ONE 3:e2751. https://doi.org/10.1371/journal.pone.0002751
Nishida R, Schulz S, Kim CS et al (1996) Male sex pheromone of a giant danaine butterfly, Idea leuconoe. J Chem Ecol 225(22):949–972. https://doi.org/10.1007/BF02029947
Nojima S, Classen A, Groot AT, Schal C (2018) Qualitative and quantitative analysis of chemicals emitted from the pheromone gland of individual Heliothis subflexa females. PLoS ONE 13:e0202035. https://doi.org/10.1371/journal.pone.0202035
Pellegrino AC, Gomes MF, Peñ Aflor V et al (2013) Weather forecasting by insects: modified sexual behaviour in response to atmospheric pressure changes. PLoS ONE 8:e75004. https://doi.org/10.1371/journal.pone.0075004
Penz CM, Williams SF (2020) Wing morphology and body design in Opsiphanes and Caligo butterflies match the demands of male mating displays (Lepidoptera: Nymphalidae). Ann Entomol Soc Am 113:207–215. https://doi.org/10.1093/aesa/saz073
Phelan PL, Roelofs CJ, Youngman RR, Baker TC (1991) Characterization of chemicals mediating ovipositional host-plant finding by Amyelois transitella females. J Chem Ecol 17:599–613. https://doi.org/10.1007/BF00982129
Pliske TE, Eisner T (1969) Sex pheromone of the queen butterfly: biology. Science (80-) 164:1170–1172. https://doi.org/10.1126/SCIENCE.164.3884.1170
Raoul S, Dall X, Nelson XJ et al (2017) Diet-mediated pheromones and signature mixtures can enforce signal reliability. Front Ecol Evol 4:145. https://doi.org/10.3389/fevo.2016.00145
Rebouças LMC, Caraciolo M, do SB, Sant’Ana AEG, et al (1999) Composição química da glândula abdominal da fêmea da mariposa Castnia licus (Drury) (Lepidoptera:Castniidae): possíveis feromônios e precursores. Quim Nova 22:645–648. https://doi.org/10.1590/S0100-40421999000500003
Renou M, Anton S (2020) Insect olfactory communication in a complex and changing world. Curr Opin Insect Sci 42:1–7. https://doi.org/10.1016/j.cois.2020.04.004
Rhainds M (2010) Female mating failures in insects. Entomol Exp Appl 136:211–226. https://doi.org/10.1111/j.1570-7458.2010.01032.x
Rodríguez G, Silva R, Cásares R et al (2012) Tecnología agronómica de la palma aceitera (Elaeis guineensis Jacq,) y manejo integrado de su defoliador Opsiphanes cassina Felder (Lepidoptera: Brassolidae) en plantaciones comerciales del estado Monagas, Venezuela. Rev Científica UDO Agrícola 12:584–598
Roelofs WL (1984) Electroantennogram assays: rapid and convenient screening procedures for pheromones. Springer, New York
Royer L, McNeil JN (1993) Effect of relative humidity conditions on responsiveness of European corn borer (Ostrinia nubilalis) males to female sex pheromone in a wind tunnel. J Chem Ecol 19:61–69. https://doi.org/10.1007/BF00987471
Rutowski RL (1982) Mate choice and lepidopteran mating behavior. Florida Entomol 65:72–82. https://doi.org/10.2307/3494146
Sakata T, Tagami K, Kuwahara Y (1995) Chemical ecology of oribatid mites. I. Oil gland components of Hydronothrus crispus Aoki. J Acarol Soc Japan 4:69–75. https://doi.org/10.2300/acari.4.69
Sandoval Y (2020) Comportamiento sexual y éxito reproductivo del barrenador de la caña Diatraea saccharalis (Fabricius 1794) (Lepidoptera: Crambidae) en laboratorio. Dissertation. Universidad Nacional de Colombia
Sasaerila Y, Gries R, Gries G et al (2003) Sex pheromone components of male Tirathaba mundella (Lepidoptera: Pyralidae). Chemoecology 13:89–93. https://doi.org/10.1007/s00049-003-0233-5
Saveer AM, Becher PG, Birgersson G et al (2014) Mate recognition and reproductive isolation in the sibling species Spodoptera littoralis and Spodoptera litura. Front Ecol Evol 2:18. https://doi.org/10.3389/fevo.2014.00018
Schmera D, Guerin PM (2012) Plant volatile compounds shorten reaction time and enhance attraction of the codling moth (Cydia pomonella) to codlemone. Pest Manag Sci 68:454–461. https://doi.org/10.1002/ps.2292
Schulz S (2004) The chemistry of pheromones and other semiochemicals I. Springer Science & Business Media, Berlin
Schulz S, Francke W, Boppre M (1988) Carboxylic acids from hairpencils of male Amauris butterflies (Lep.: Danainae). Biol Chem Hoppe Seyler 369:633–638. https://doi.org/10.1515/bchm3.1988.369.2.633
Schulz S, Boppre M, Vane-Wright RI (1993) Specific mixtures of secretions from male scent organs of African milkweed butterflies (Danainae). Philos Trans R Soc B Biol Sci 342:161–181. https://doi.org/10.1098/rstb.1993.0144
Schulz S, Estrada C, Yildizhan S et al (2008) An antiaphrodisiac in Heliconius melpomene butterflies. J Chem Ecol 34:82–93. https://doi.org/10.1007/s10886-007-9393-z
Shapiro AM (1970) The role of sexual behavior in density-related dispersal of pierid butterflies. Am Nat 104:367–372. https://doi.org/10.1086/282670
Sierra Márquez J, Sierra Márquez L, Olivero-Verbel J (2017) Economic potential of the oil palm (Elaeis guineensis Jacq). Agron Mesoam 28:523. https://doi.org/10.15517/ma.v28i2.25927
Solensky MJ (2004) The effect of behavior and ecology on male mating success in overwintering monarch butterflies (Danaus plexippus). J Insect Behav 17:723–743. https://doi.org/10.1023/B:JOIR.0000048985.58159.0d
South SH, House CM, Moore AJ et al (2011) Male cockroaches prefer a high carbohydrate diet that makes them more attractive to females: implications for the study of condition dependence. Evolution 65:1594–1606. https://doi.org/10.1111/j.1558-5646.2011.01233.x
Srygley RB (1994) Shivering and its cost during reproductive behaviour in neotropical owl butterflies, Caligo and Opsiphanes (Nymphalidae: Brassolinae). Anim Behav 47:23–32. https://doi.org/10.1006/anbe.1994.1004
Srygley RB, Penz CM (1999) Lekking in Neotropical Owl Butterflies, Caligo illioneus and C. oileus (Lepidoptera: Brassolinae). J Insect Behav 12:81–103. https://doi.org/10.1023/A:1020981215501
Stashenko EE, Martínez JR (2010) Algunos aspectos prácticos para la identificación de analitos por cromatografía de gases acoplada a espectrometría de masas. Sci Chromatogr 2:29–47
Stepien TL, Zmurchok C, Hengenius JB et al (2020) Moth mating: modeling female pheromone calling and male navigational strategies to optimize reproductive success. Appl Sci 10:6543. https://doi.org/10.3390/app10186543
Sugisawa H, Nakamura K, Tamura H (1990) The aroma profile of the volatiles in marine green algae (Ulva pertusa). Food Rev Int 6:573–589. https://doi.org/10.1080/87559129009540893
Tabacchi R, Saturnin C, Porret CL et al (2010) A guide to the analysis of chiral compounds by GC. Restek. https://gcms.cz/labrulez-bucket-strapi-h3hsga3/e8cc224389284d48bbd2e2652527dd95/59889.pdf. Accessed 4 Jan 2023
Taylor LR, Vane-Wright RI, Ackery PR (1986) The biology of butterflies. J Anim Ecol 55:752. https://doi.org/10.2307/4754
Tillman JA, Seybold SJ, Jurenka RA, Blomquist GJ (1999) Insect pheromones—an overview of biosynthesis and endocrine regulation. Insect Biochem Mol Biol 29:481–514. https://doi.org/10.1016/S0965-1748(99)00016-8
Vane-Wright RI, Boppre M (1993) Visual and chemical signalling in butterflies: functional and phylogenetic perspectives. Philos Trans R Soc Lond B 340:197–205. https://doi.org/10.1098/rstb.1993.0058
Walton NJ, Mayer MJ, Narbad A (2003) Vanillin. Phytochemistry 63:505–515. https://doi.org/10.1016/S0031-9422(03)00149-3
Wang H-L, Brattström O, Brakefield PM et al (2014) Identification and biosynthesis of novel male specific esters in the wings of the tropical butterfly, Bicyclus martius sanaos. J Chem Ecol 406(40):549–559. https://doi.org/10.1007/S10886-014-0452-Y
Wellington WG (1946) The effects of variations in atmospheric pressure upon insects. Can J Res 24:51–70. https://doi.org/10.1139/cjr46d-006
Wickman PC (1985) The influence of temperature on the territorial and mate locating behaviour of the small heath butterfly, Coenonympha pamphilus (L.) (Lepidoptera: Satyridae). Behav Ecol Sociobiol 16:233–238. https://doi.org/10.1007/BF00310985
Wu S, Refinetti R, Kok LT et al (2014) Photoperiod and Temperature Effects on the Adult Eclosion and Mating Rhythms in Pseudopidorus fasciata (Lepidoptera: Zygaenidae). Environ Entomol 43:1650–1655. https://doi.org/10.1603/EN14164
Wyatt TD (2017) Pheromones. Curr Biol 27:739–743. https://doi.org/10.1016/j.cub.2017.06.039
Yew JY, Chung H (2015) Insect pheromones: an overview of function, form, and discovery. Prog Lipid Res 59:88–105. https://doi.org/10.1016/j.plipres.2015.06.001
Zeng B, Zhu W, Fu Y, Zhou IdS (2018) Influence of high-temperature exposure on the mating, oviposition and thermotaxis of Bactrocera cucurbitae (Coquillet) (Diptera: Tephritidae). PLoS ONE 13:e0204065. https://doi.org/10.1371/journal.pone.0204065
Zvereva EL, Kozlov MV (2016) The costs and effectiveness of chemical defenses in herbivorous insects: a meta-analysis. Ecol Monogr 86:107–124. https://doi.org/10.1890/15-0911.1
Acknowledgements
The authors thank the Fondo de Fomento Palmero, administrated by Federación Nacional de Cultivadores de Palma (Fedepalma), Centro de Investigación en Palma de Aceite (Cenipalma), Universidad Nacional de Colombia – Sede Bogotá, and Universidad Antonio Nariño for the financial support. Likewise, they would like to thank Dr. Nancy Barreto, Associate Ph.D. Researcher at Tibaitatá Research Center in Agrosavia for allowing the use of the Entomology Laboratory to perform EAG and wind tunnel assays.
Funding
Open Access funding provided by Colombia Consortium. This research was supported by grants from the Dirección Nacional de Investigación (DIB—Universidad Nacional de Colombia—Sede Bogotá, Centro de Investigación en Palma de Aceite (Cenipalma), Fondo de Fomento Palmero—Federación Nacional de Cultivadores de Palma (Fedepalma), and Vicerrectoría de Ciencia, Tecnología e Innovación—Universidad Antonio Nariño.
Author information
Authors and Affiliations
Contributions
AR-F, CC-A, and AM supervised/designed the research. JJB-C conducted the experiments, data analysis, and original text. RAT performed insect rearing and collected insects in the countryside of Llanos Orientales, Colombia. All authors revised and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Günther Raspotnig.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bustos-Cortés, J.J., Aldana-De la Torre, R.C., Morales-Rodríguez, A. et al. Sexual behavior and chemical signaling in Opsiphanes cassina (Lepidoptera: Nymphalidae). Chemoecology 33, 161–175 (2023). https://doi.org/10.1007/s00049-023-00393-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-023-00393-z