Abstract
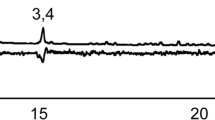
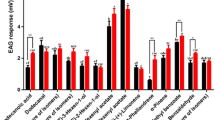
Previous studies on the legume pod borer, Maruca vitrata Fabricius (Lepidoptera: Crambidae), a serious pest of cowpea, Vigna unguiculata (L.) Walp. (Fabales: Fabaceae), in sub-Saharan Africa have focused on sex pheromones, but the role of the host plant on sexual behavior has not been explored. We investigated this interaction in the laboratory using behavioral assays and chemical analyses. We found that the presence of cowpea seedlings and a dichloromethane extract of the leaf increased coupling in the legume pod borer by 33 and 61 %, respectively, compared to the control, suggesting the involvement of both contact and olfactory cues. We used coupled gas chromatography-electroantennographic detection (GC/EAD) and GC-mass spectrometry (GC/MS) to identify compounds from the cowpea leaf extract, detected by M. vitrata antenna. We found that the antennae of the insect consistently detected four components, with 1-octen-3-ol identified as a common and dominant component in both the volatiles released by the intact cowpea plant and leaf extract. We therefore investigated its role in the coupling of M. vitrata. In dose–response assays, 1-octen-3-ol increased coupling in M. vitrata with increasing dose of the compound compared to the control. Our results suggest that the cowpea volatile 1-octen-3-ol contributes to M. vitrata sexual behavior.





Similar content being viewed by others
References
Adams RP (1995) Identification of essential oil components by gas chromotography/ mass spectrometry: Adams2 terpenoid/natural product library. Allured, Carol Stream
Adati T, Tatsuki S (1999) Identification of the female sex pheromone of the legume pod borer, Maruca vitrata and antagonistic effects of geometrical isomers. J Chem Ecol 25:105–115. doi:10.1023/A:1020841217376
Adati T, Nakamura S, Tamò M, Kawazu K (2004) Effect of temperature on development and survival of the legume pod borer, Maruca vitrata (Fabricius) (Lepidoptera: Pyralidae) reared on a semi-synthetic diet. Appl Entomol Zool 39:139–145. doi:10.1303/Aez.2004.139
Adati T, Tamo M, Sr Y, Mca D, Bb S, Hammond W (2008) Integrated pest management for cowpea-cereal cropping systems in the West African Savannah. Int J Trop Insect Sci 27(3):123. doi:10.1017/S1742758407883172
Allmann S, Späthe A, Bisch-Knaden S, Kallenbach M, Reinecke A, Sachse S, Baldwin IT, Hansson BS (2013) Feeding-induced rearrangement of green leaf volatiles reduces moth oviposition. Elife 2013:1–23. doi:10.7554/Elife.00421
Beck JJ, Higbee BS, Light DM, Gee WS, Merrill GB, Hayashi JM (2012) Hull split and damaged almond volatiles attract male and female navel orangeworm moths. J Agric Food Chem 60(33):8090–8096. doi:10.1021/Jf302658v
Bruce TJA, Wadhams LJ, Woodcock CM (2005) Insect host location: a volatile situation. Trends Plant Sci 10(6):269–274. doi:10.1016/J.Tplants.2005.04.003
Dai J, Deng J, Du J (2008) Development of bisexual attractants for diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae) based on sex pheromone and host volatiles. Appl Entom Zool 43(4):631–638. doi:10.1303/Aez.2008.631
Deng J, Hy W, Huang Y, Du J (2004) Enhancement of attraction to sex pheromones of Spodoptera exigua by volatile compounds produced by host plants. J Chem Ecol 30(10):2037–2045. doi:10.1023/B:Joec.0000045593.62422.73
Dicke M, Baldwin I (2010) The evolutionary context for herbivore-induced plant volatiles: beyond the “cry for help”. Trends Plant Sci 15:167–175. doi:10.1016/J.Tplants.2009.12.002
Downham M (2005) Implementing pheromone traps and other new technologies for control of cowpea pest in West Africa through Farmer Field Schools Crop Protection Programme. Natural Resources Institute, University Of Greenwich
Downham M (2006) Facilitating the uptake of pheromone traps for Maruca vitrata in West Africa. Final technical report Crop Protection Programme. Natural Resources Institute, University of Greenwich.
Downham MCA, Hall DR, Chamberlain DJ, Cork A, Farman DI, Tamò M, Dahounto D, Datinon B, Adetonah S (2003) Minor components in the sex pheromone of legume podborer: Maruca vitrata development of an attractive blend. J Chem Ecol 29(4):989–1011. doi:10.1023/A:1022996103147
Downham MCA, Tamò M, Dr H, Datinon B, Adetonah S, Di F (2004) Developing pheromone traps and lures for Maruca vitrata in Benin, West Africa. Entomol Exp Appl 110(2):151–158. doi:10.1111/J.0013-8703.2004.00128.X
Fürstenau B, Rosell G, Guerrero A, Quero C (2012) Electrophysiological and behavioral responses of the black-banded oak borer, Coroebus florentinus, to conspecific and host-plant volatiles. J Chem Ecol 38:378–388. doi:10.1007/S10886-012-0110-1
Gregory B (1989) Field observations of mating, oviposition, and feeding behavior of Anticarsia gemmatalis (Lepidoptera: Noctuidae) in a soybean field. J Insect Behav 2(6):761–774. doi:10.1007/Bf01049399
Hassan Mn (2007) Re-investigation of the female sex pheromone of the legume podborer, Maruca vitrata (Lepidoptera: Crambidae). Thesis, University of Greenwich
Huang C, Peng W (2001) Emergence, mating, and oviposition of the bean podborer, Maruca vitrata (F.) (Lepidoptera: Pyralidae). Formos Entomology 21:37–45
Jackai LEN (1981) Relationship between cowpea crop phenology and field infestation by the legume pod borer, Maruca testulalis. Ann Entomol Soc Am 74(4):402–408.. doi:10.1093/Aesa/74.4.402
Jackai LEN, Ochieng RS, Raulston JR (1990) Mating and oviposition behaviour in the legume pod borer, Maruca testulalis. Entomol Exp Appl 56:179–186. doi:10.1111/J.1570-7458.1990.Tb01395.X
Kudo A, Kawazu K, Adati T (2014) Age-dependent changes in feeding preference and effects of commercial semi-synthetic diets on development of the legume pod borer, Maruca vitrata (Lepidoptera: Crambidae). Appl Entomol Zool 49:459–466. doi:10.1007/S13355-014-0272-0
Kugel M (1977) The time course of the electroretinogram of compound eyes in insects and its dependence on special recording conditions. J Exp Bio 71(1):1–6
Landolt PJ, Phillips TW (1997) Host plant influences on sex pheromone behavior of phytophagous insects 1. Ann Rev Entomol 42(1):371–391. doi:10.1146/Annurev.Ento.42.1.371
Landolt PJ, Heath RR, Millar JG, Davis-Hernandez KM, Dueben BD, Ward KE (1994) Effects of host plant, Gossypium hirsutum L., on sexual attraction of cabbage looper moths, Trichoplusia ni (Hübner) (Lepidoptera: Noctuidae). J Chem Ecol 20:2959–2974. doi:10.1007/Bf02098402
Lu P, Hl Q, Wang X, Wang X, Cl L (2007) Emergence and mating rhythms of the legume pod borer, Maruca vitrata (Fabricius, 1787) (Lepidoptera: Pyralidea). Pan-Pacific Entomol 83(3):226–234. doi:10.3956/0031-0603-83.3.226
McNeil JN, Delisle J (1989a) Are host plants important in pheromone-mediated mating systems of Lepidoptera? Experientia 45:236–240. doi:10.1007/Bf01951809
McNeil JN, Delisle J (1989b) Host plant pollen influences calling behavior and ovarian development of the sunflower moth, Homoeosoma electellum. Oecologia 80:201–205. doi:10.1007/Bf00380151
National Institutes of Standards and Technology (NIST) (2005) NIST/EPA/NIH Mass Spectral Library. http://www.nist.gov
Ngumbi EN, Ngi-Song AJ, Njagi ENM, Torto R, Wadhams LJ, Birkett MA, Pickett JA, Overholt WA, Torto B (2005) Responses of the stem borer larval endoparasitoid Cotesia flavipes (Hymenoptera: Braconidae) to plant derived synomones: laboratory and field cage experiments. Bioc Sci Tech 15:271–279. doi:10.1080/09583150400016985
Okeyo-Owuor JB, Agwaro PO, Simbi COJ (1983) Studies on the legume pod-borer Maruca testulalis (Geyer)—V. Larval population. Int J Trop Insect Sci 4(1–2):75–81. doi:10.1017/S1742758400004057
Onyango FO, Ochieng’-Odero JPR (1993) Laboratory rearing of the legume pod borer, Maruca testulalis Geyer (Lepidoptera: Pyralidae) on a semi-synthetic diet. Int J Trop Insect Sci 14(5–6):719–722. doi:10.1017/S174275840001818x
Raina AK, Kingan TG, Mattoo AK (1992) Chemical signals from host plant and sexual behavior in a moth. Science (Washington) 255(5044):592–594
Reddy GVP, Guerrero A (2004) Interactions of insect pheromones and plant semiochemicals. Trends Plant Sci 9(5):253–261. doi:10.1016/J.Tplants.2004.03.009
Schläger S, Ulrichs C, Srinivasan R, Beran F, Krm B, Mewis I, Schreiner M (2012) Developing pheromone traps and lures for Maruca vitrata in Taiwan. Gesunde Pflanzen 64(4):183–186. doi:10.1007/S10343-012-0287-9
Schmidt-Büsser D, Von Arx M, Pm AG (2009) Host plant volatiles serve to increase the response of male European grape berry moths, Eupoecilia ambiguella, to their sex pheromone. J Comp Physiol A: Neuroethol, Sens, Neural, Behav Physiol 195(9):853–864. doi:10.1007/S00359-009-0464-1
Sharma HC (1998) Bionomics, host plant resistance, and management of the legume pod borer, Maruca vitrata—a review. Crop Prot 17(5):373–386. doi:10.1016/S0261-2194(98)00045-3
Srinivasan R (2012) Integrating biopesticides in pest management strategies for tropical vegetable production. J Biopest 5:36–45
Tang R, Jp Z, Zn Z (2012) Electrophysiological and behavioral responses of male fall webworm moths (Hyphantria cunea) to herbivory-induced mulberry (Morus alba) leaf volatiles. Plos One 7:1–7. doi:10.1371/Journal.Pone.0049256
Tasin M, Bäckman A, Bengtsson M, Varela N, Ioriatti C, Witzgall P (2006) Wind tunnel attraction of grapevine moth females, Lobesia botrana, to natural and artificial grape odour. Chemoecology 16(2):87–92. doi:10.1007/S00049-005-0332-6
Team R (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing ISBN 3-900051-07-0, Vienna, Austria.
Timko MP, Singh BB (2008) Cowpea, a multifunctional legume. Genomics of tropical crop plants. Springer, New York, pp 227–258
Von Arx M, Schmidt-Büsser D, Pm G (2011) Host plant volatiles induce oriented flight behaviour in male European grapevine moths, Lobesia botrana. J Ins Physiol 57(10):1323–1331. doi:10.1016/J.Jinsphys.2011.06.010
Von Arx M, Schmidt-Büsser D, Pm G (2012) Plant volatiles enhance behavioral responses of grapevine moth males, Lobesia botrana to sex pheromone. J Chem Ecol 38:222–225. doi:10.1007/S10886-012-0068-Z
Yang Z, Bengtsson M, Witzgall P (2004) Host plant volatiles synergize response to sex pheromone in codling moth, Cydia pomonella. J Chem Ecol 30(3):619–629. doi:10.1023/B:Joec.0000018633.94002.Af
Acknowledgments
This research was funded by the Federal Ministry for Economic Cooperation and Development (BMZ), Germany and supported by the International Centre of Insect Physiology and Ecology (icipe), Kenya, with collaboration from the World Vegetable Center (AVRDC), Taiwan. The study could not have been realized without the studentship support from the German Academic Exchange Service (DAAD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 34 kb)
Rights and permissions
About this article
Cite this article
Bendera, M., Ekesi, S., Ndung’u, M. et al. A major host plant volatile, 1-octen-3-ol, contributes to mating in the legume pod borer, Maruca vitrata (Fabricius) (Lepidoptera: Crambidae). Sci Nat 102, 47 (2015). https://doi.org/10.1007/s00114-015-1297-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-015-1297-0